Abstract
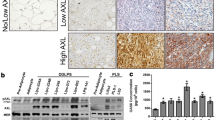
The Akt/mTOR and MAPK pathways play important roles in modulating cellular function in response to extracellular signals, and they are known to be activated in certain kinds of sarcomas. Few investigations have examined these pathways in dedifferentiated liposarcoma (DDLS), in relation to clinicopathological features. Clinicopathological and immunohistochemical analyses were conducted using 99 DDLS specimens. An in vitro study was also conducted to examine the antitumor effects of an mTOR inhibitor and a MEK inhibitor on two DDLS cell lines. The clinicopathological analyses revealed that the AJCC staging was a significant prognostic factor for overall survival and that the tumor size, depth, and location were significant prognostic factors for event-free survival. Phosphorylated Akt (pAkt), pmTOR, pS6RP, p4E-BP1, pMEK, and pERK expressions were positive in 57.4, 52.4, 71.4, 57.1, 84.1, and 50.8 % of the dedifferentiated component of the 63 primary DDLSs. Positive staining for pmTOR was significantly more frequent in the dedifferentiated component than the well-differentiated component. A univariate prognostic analysis revealed that pmTOR expression was associated with poor prognosis in the tumors in the retroperitoneum/ventral body cavity. The mTOR and MEK inhibitors dose-dependently inhibited the cell proliferation of both DDLS cell lines and decreased the expression of downstream pS6RP and pERK, respectively. The combined use of the two inhibitors enhanced antiproliferative activity. In conclusion, the Akt/mTOR and MAPK pathways were activated in DDLS specimens, and the inhibition of these pathways decreased cell proliferation in DDLS cell lines. Our findings suggest that these pathways could be a therapeutic target for patients with DDLS.



Similar content being viewed by others
References
Dei Tos J. Dedifferentiated liposarcoma. In: Fletcher CDM, Bridge JA, Hogendoorn PCW, Mertens F, editors. WHO classification of tumours of soft tissue and bone. Lyon: International Agency for Research on Cancer (IRAC); 2013. p. 37–8.
Pedeutour F, Forus A, Coindre JM, Berner JM, Nicolo G, Michiels JF, et al. Structure of the supernumerary ring and giant rod chromosomes in adipose tissue tumors. Genes Chromosom Cancer. 1999;24:30–41.
Mertens F, Fletcher CD, Dal Cin P, De Wever I, Mandahl N, Mitelman F, et al. Cytogenetic analysis of 46 pleomorphic soft tissue sarcomas and correlation with morphologic and clinical features: a report of the CHAMP Study Group. Chromosomes and MorPhology. Genes Chromosom Cancer. 1998;22:16–25.
Dalal KM, Kattan MW, Antonescu CR, Brennan MF, Singer S. Subtype specific prognostic nomogram for patients with primary liposarcoma of the retroperitoneum, extremity, or trunk. Ann Surg. 2006;244:381–91.
Italiano A, Toulmonde M, Cioffi A, Penel N, Isambert N, Bompas E, et al. Advanced well-differentiated/dedifferentiated liposarcomas: role of chemotherapy and survival. Ann Oncol. 2012;23:1601–7.
Jones RL, Fisher C, Al-Muderis O, Judson IR. Differential sensitivity of liposarcoma subtypes to chemotherapy. Eur J Cancer. 2005;41:2853–60.
Coindre JM, Pedeutour F, Aurias A. Well-differentiated and dedifferentiated liposarcomas. Virchows Arch. 2010;456:167–79.
Linehan DC, Lewis JJ, Leung D, Brennan MF. Influence of biologic factors and anatomic site in completely resected liposarcoma. J Clin Oncol. 2000;18:1637–43.
Altomare DA, Testa JR. Perturbations of the AKT signaling pathway in human cancer. Oncogene. 2005;24:7455–64.
Vakiani E, Solit DB. KRAS and BRAF: drug targets and predictive biomarkers. J Pathol. 2011;223:220–30.
Hsieh AC, Liu Y, Edlind MP, Ingolia NT, Janes MR, Sher A, et al. The translational landscape of mTOR signalling steers cancer initiation and metastasis. Nature. 2013;485:55–61.
Dasari A, Messersmith WA. New strategies in colorectal cancer: biomarkers of response to epidermal growth factor receptor monoclonal antibodies and potential therapeutic targets in phosphoinositide 3-kinase and mitogen-activated protein kinase pathways. Clin Cancer Res. 2010;16:3811–8.
Mendoza MC, Er EE, Blenis J. The Ras-ERK and PI3K-mTOR pathways: cross-talk and compensation. Trends Biochem Sci. 2011;36:320–8.
Wang C, Cigliano A, Delogu S, Armbruster J, Dombrowski F, Evert M, et al. Functional crosstalk between AKT/mTOR and Ras/MAPK pathways in hepatocarcinogenesis: implications for the treatment of human liver cancer. Cell Cycle. 2013;12:1999–2010.
Dobashi Y, Suzuki S, Sato E, Hamada Y, Yanagawa T, Ooi A. EGFR-dependent and independent activation of Akt/mTOR cascade in bone and soft tissue tumors. Mod Pathol. 2009;22:1328–40.
Dobashi Y, Suzuki S, Sugawara H, Ooi A. Involvement of epidermal growth factor receptor and downstream molecules in bone and soft tissue tumors. Hum Pathol. 2007;38:914–25.
Yamada Y, Kohashi K, Fushimi F, Takahashi Y, Setsu N, Endo M, et al. Activation of the Akt-mTOR pathway and receptor tyrosine kinase in patients with solitary fibrous tumors. Cancer. 2014;120:864–76.
Takahashi Y, Kohashi K, Yamada Y, Endo M, Setsu N, Ishii T, et al. Activation of the Akt/mammalian target of rapamycin pathway in myxofibrosarcomas. Hum Pathol. 2014;45:984–93.
Tomita Y, Morooka T, Hoshida Y, Zhang B, Qiu Y, Nakamichi I, et al. Prognostic significance of activated AKT expression in soft-tissue sarcoma. Clin Cancer Res. 2006;12:3070–7.
Setsu N, Kohashi K, Fushimi F, Endo M, Yamamoto H, Takahashi Y, et al. Prognostic impact of the activation status of the Akt/mTOR pathway in synovial sarcoma. Cancer. 2013;119:3504–13.
Endo M, Yamamoto H, Setsu N, Kohashi K, Takahashi Y, Ishii T, et al. Prognostic significance of AKT/mTOR and MAPK pathways and antitumor effect of mTOR inhibitor in NF1-related and sporadic malignant peripheral nerve sheath tumors. Clin Cancer Res. 2013;19:450–61.
Setsu N, Yamamoto H, Kohashi K, Endo M, Matsuda S, Yokoyama R, et al. The Akt/mammalian target of rapamycin pathway is activated and associated with adverse prognosis in soft tissue leiomyosarcomas. Cancer. 2012;118:1637–48.
Gutierrez A, Snyder EL, Marino-Enriquez A, Zhang YX, Sioletic S, Kozakewich E, et al. Aberrant AKT activation drives well-differentiated liposarcoma. Proc Natl Acad Sci U S A. 2011;108(39):16386–91.
Trojani M, Contesso G, Coindre JM, Rouesse J, Bui NB, de Mascarel A, et al. Soft-tissue sarcomas of adults; study of pathological prognostic variables and definition of a histopathological grading system. Int J Cancer. 1984;33:37–42.
Pollock RE, Baker LH, Brennan MF, Coombes K, Kattan M, Kneisl JS, et al. Soft tissue sarcoma. In: Edge SB, Byrd DR, Compton CC, Fritz AG, Greene FL, Trotti A, editors. AJCC cancer staging manual 7th edn. American Joint Committee on Cancer, Springer: Chicago; 2010. pp 291–298.
Nishio J, Iwasaki H, Ishiguro M, Ohjimi Y, Fujita C, Ikegami H, et al. Establishment of a novel human dedifferentiated liposarcoma cell line, FU-DDLS-1: conventional and molecular cytogenetic characterization. Int J Oncol. 2003;22:535–42.
Ariizumi T, Ogose A, Kawashima H, Hotta T, Li G, Xu Y, et al. Establishment and characterization of a novel dedifferentiated liposarcoma cell line, NDDLS-1. Pathol Int. 2011;61:461–8.
Jour G, Gullet A, Liu M, Hoch BL. Prognostic relevance of Fédération Nationale des Centres de Lutte Contre le Cancer grade and MDM2 amplification levels in dedifferentiated liposarcoma: a study of 50 cases. Mod Pathol. 2015;28:37–47.
Gronchi A, Collini P, Miceli R, Valeri B, Renne SL, Dagrada G, et al. Myogenic differentiation and histological grading are major prognostic determinants in retroperitoneal liposarcoma. Am J Surg Pathol. 2015;39:383–93.
Guo S, Lopez-Marquez H, Fan KC, Choy E, Cote G, Harmon D, et al. Synergistic effects of targeted PI3K signaling inhibition and chemotherapy in liposarcoma. PLoS One. 2014;9:e93996.
Demetri GD, Chwla SP, Ray-Coquard I, Le Cesne A, Staddon AP, Milhem MM, et al. Results of an international randomized phase III trial of the mammalian target of rapamycin inhibitor ridaforolimus versus placebo to control metastatic sarcomas in patients after benefit from prior chemotherapy. J Clin Oncol. 2013;31:2485–92.
Kinkade CW, Castillo-Martin M, Puzio-Kuter A, Yan J, Foster TH, Gao H, et al. Targeting AKT/mTOR and ERK MAPK signaling inhibits hormone-refractory prostate cancer in a preclinical mouse model. J Clin Invest. 2008;118:3051–64.
Engelman JA, Chen L, Tan X, Crosby K, Guimaraes AR, Upadhyay R, et al. Effective use of PI3K and MEK inhibitors to treat mutant Kras G12D and PIK3CA H1047R murine lung cancers. Nat Med. 2008;14:1351–6.
Chawla SP, Staddon AP, Baker LH, Schuetze SM, Tolcher AW, D’Amato GZ, et al. Phase II study of the mammalian target of rapamycin inhibitor ridaforolimus in patients with advanced bone and soft tissue sarcomas. J Clin Oncol. 2012;30:78–84.
Li S, Kong Y, Si L, Chi Z, Cui C, Sheng X, et al. Phosphorylation of mTOR and S6RP predicts the efficacy of everolimus in patients with metastatic renal cell carcinoma. BMC Cancer. 2014;14:376.
Acknowledgments
This work was supported by JSPS KAKEN Grant Number 25293088. The English used in this article was revised by KN International (http://www.kninter.com). We would like to express our sincere appreciation to Prof. Masao Tanaka, Department of Surgery and Oncology, Prof. Yoshihiko Maehara, Department of Surgery and Science and Prof. Seiji Naito, Department of urology, Graduate School of Medical Sciences, Kyushu University for kind support for our research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Rights and permissions
About this article
Cite this article
Ishii, T., Kohashi, K., Iura, K. et al. Activation of the Akt-mTOR and MAPK pathways in dedifferentiated liposarcomas. Tumor Biol. 37, 4767–4776 (2016). https://doi.org/10.1007/s13277-015-4232-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-4232-2