Abstract
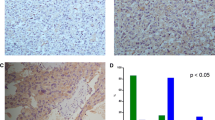
This study aims to investigate the effect of heat shock protein 90 (HSP90) inhibitor 17-allylamino-17-demethoxygeldanamycin (17-AAG) in the malignant pheochromocytoma using a xenograft mouse model. Treatment with 17-AAG induced a marked reduction in the volume and weight of PC12 pheochromocytoma cell tumor xenografts in mice. Furthermore, 17-AAG also significantly inhibited the expression of HSP90 and its client proteins. Our results validated HSP90 as an important target in pheochromocytoma and provided rationale for the testing of HSP90 inhibitors as a promising therapeutic agent in the antitumor therapy of pheochromocytoma.





Similar content being viewed by others
References
Adler JT, Meyer-Rochow GY, Chen H, Benn DE, Robinson BG, Sippel RS, et al. Pheochromocytoma: current approaches and future directions. Oncologist. 2008;13(7):779–93.
Ayala-Ramirez M, Feng L, Johnson MM, Ejaz S, Habra MA, Rich T, et al. Clinical risk factors for malignancy and overall survival in patients with pheochromocytomas and sympathetic paragangliomas: primary tumor size and primary tumor location as prognostic indicators. J Clin Endocrinol Metab. 2011;96:717–25.
Sanderson S, Valenti M, Gowan S, et al. Benzoquinone ansamycin heat shock protein 90 inhibitors modulate multiple functions required for tumor angiogenesis. Mol Cancer Ther. 2006;5(3):522–32.
Pacey S, Gore M, Chao D, et al. A Phase II trial of 17-allylamino, 17-demethoxygeldanamycin (17-AAG, tanespimycin) in patients with metastatic melanoma. Investig New Drugs. 2012;30(1):341–9.
Iyer G, Morris MJ, Rathkopf D, Slovin SF, Steers M, Larson SM, et al. A phase I trial of docetaxel and pulse-dose 17-allylamino-17-demethoxygeldanamycin in adult patients with solid tumors. Cancer Chemother Pharmacol. 2012;69(4):1089–97.
Lang SA, Klein D, Moser C, Gaumann A, Glockzin G, Dahlke MH, et al. Inhibition of heat shock protein 90 impairs epidermal growth factor-mediated signaling in gastric cancer cells and reduces tumor growth and vascularization in vivo. Mol Cancer Ther. 2007;6(3):1123–32.
Xu Y, Zhang C, Chen D, Zhao J, Shen Z, Wu Y, et al. Effect of HSP90 inhibitor in pheochromocytoma PC12 cells: an experimental investigation. Tumour Biol. 2013;34(6):4065–71.
Powers MV, Workman P. Targeting of multiple signalling pathways by heat shock protein 90 molecular chaperone inhibitors. Endocr Relat Cancer. 2006;13 Suppl 1:S125–35.
Trepel J, Mollapour M, Giaccone G, et al. Targeting the dynamic HSP90 complex in cancer. Nat Rev Cancer. 2010;10:537–49.
Whitesell L, Lindquist SL. HSP90 and the chaperoning of cancer. Nat Rev Cancer. 2005;5:761–72.
Gartner EM, Silverman P, Simon M, Flaherty L, Abrams J, Ivy P, et al. A phase II study of 17-allylamino-17-demethoxygeldanamycin in metastatic or locally advanced, unresectable breast cancer. Breast Cancer Res Treat. 2012;131:933–7.
Eiseman JL et al. Pharmacokinetics and pharmacodynamics of 17-demethoxy 17-[[(2-dimethylamino)ethyl]amino]geldanamycin (17DMAG, NSC 707545) in C.B-17 SCID mice bearing MDA-MB-231 human breast cancer xenografts. Cancer Chemother Pharmacol. 2005;55:21–32.
Moulick K, Ahn JH, Zong H, Rodina A, Cerchietti L, Gomes DaGama EM, et al. Affinity-based proteomics reveal cancer-specific networks coordinated by Hsp90. Nat Chem Biol. 2011;7:818–26.
Sanderson S, Valenti M, Gowan S, Patterson L, Ahmad Z, Workman P, et al. Benzoquinone ansamycin heat shock protein 90 inhibitors modulate multiple functions required for tumor angiogenesis. Mol Cancer Ther. 2006;5(3):522–32.
Kamal A, Thao L, Sensintaffar J, Zhang L, Boehm MF, Fritz LC, et al. A high-affinity conformation of Hsp90 confers tumour selectivity on Hsp90 inhibitors. Nature. 2003;425(6956):407–10.
Giubellino A, Sourbier C, Lee MJ, Scroggins B, Bullova P, Landau M, et al. Targeting heat shock protein 90 for the treatment of malignant pheochromocytoma. PLoS One. 2013;8(2):e56083.
Khong T, Spencer A. Targeting HSP90 induces apoptosis and inhibits critical survival and proliferation pathways in multiple myeloma. Mol Cancer Ther. 2011;10:1909–17.
Acknowledgments
This study was supported by the grants from the National Natural Science Foundation of China (no. 81272936) and Shanghai Municipal Natural Science Foundation (no. 134119a2700).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Yunze Xu and Qi Zhu share the first authorship of this article.
Rights and permissions
About this article
Cite this article
Xu, Y., Zhu, Q., Chen, D. et al. The HSP90 inhibitor 17-AAG exhibits potent antitumor activity for pheochromocytoma in a xenograft model. Tumor Biol. 36, 5103–5108 (2015). https://doi.org/10.1007/s13277-015-3162-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3162-3