Abstract
Telomerase was initially considered as a relevant factor distinguishing cancer from normal cells. During detailed studies, it appeared that its expression and activity is not only limited to cancer cells however, but in this particular cells, the telomerase is much more abundant. Thus, it has become a very promising target for an anticancer therapy. It was revealed in many studies that regulation of telomerase is a multifactorial process in mammalian cells, involving regulation of expression of telomerase subunits coding genes, post-translational protein–protein interactions, and protein phosphorylation. Numerous proto-oncogenes and tumor suppressor genes are engaged in this mechanism, and the complexity of telomerase control is studied in the context of tumor development as well as aging. Additionally, since numerous studies reveal a correlation between short telomeres and increased genome instability or cell mortality, the telomerase control appears to be one of the crucial factors to study in order to improve the cancer diagnostics and therapy or prevention. Interestingly, almost 100 % of adenocarcinoma, including breast cancer cells, expresses telomerase which makes it a good target for telomerase-related therapy. Additionally, telomerase is also supposed to be associated with drug resistance. Thus, targeting the enzyme might result in attenuation of this phenomenon. Moreover, since stem cells existence was reported, it must be considered whether targeting telomerase can bring some serious side effects and result in stem cells viability or their regenerative potential decrease. Thus, we review some molecular mechanisms engaged in therapy based on targeting telomerase in breast cancer cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Telomeres are guanine-rich repeated sequences located at the ends of chromosomes. They function as a biological clock limiting the cell proliferation potential with every next cell division (Hayflick limit) [1]. This phenomenon is accompanied by cell senescence, mitotic crisis, and/or apoptosis [2, 3]. However, most cancer cells reveal a telomere length maintenance mechanism which is responsible for telomeres renewal during cell proliferation. Majority of cancer cells utilize telomerase for this purpose, and the enzyme contains its integral RNA molecule which constitutes the template for reverse transcription of new telomeric DNA [4]. Telomerase has been recognized as a relevant factor distinguishing cancer from normal cells since it is either undetectable or has low level of activity in normal somatic cells [5]. Thus, it has become a very promising target for an anticancer therapy. It was revealed in many studies that regulation of telomerase in mammalian cells is a multifactorial process, involving expression of telomerase subunits coding genes, post-translational protein–protein interactions, and protein phosphorylation [6, 7]. Numerous proto-oncogenes and tumor suppressor genes are engaged in this mechanism. Thus the complexity of telomerase control is studied in the context of stem cell renewal, tumor development, as well as aging [8–14]. Thus, it is of great interest to identify the enzyme regulators (tumor suppressors or oncogenes, etc.) The studies across many tumor types have shown that the vast majority of tumors (~85 %) express telomerase [15, 16] and hence, are able to maintain a stable and homogenous telomere length. Consequently, they can avoid replicative senescence. Only just about 15 % of tumors either do not maintain telomere length or activate alternative lengthening of telomeres (ALT) [17]. Interestingly, almost 100 % of adenocarcinoma, including breast cancer cells, express telomerase [18]. Till now, several strategies have been proposed to control telomerase in cancer cells: antisense technology against telomerase RNA component (TR) and telomerase reverse transcriptase (TERT), ribozymes against TERT, antiestrogens, progesterone, vitamin D, retinoic acid, quadruplex stabilizers, telomere and telomerase-targeting agents, modulation of interaction with other proteins involved in the regulation of telomerase and telomeres, etc. [19–25]. However, the transcription control of key telomerase subunits seems to play the crucial role in whole complexes activity and cancer cells immortality. Interesting results were shown by Blackburn et al. [26]. In Saccharomyces cerevisiae, the telomerase deletion response (TDR), that occurs when telomeres can no longer be maintained by telomerase, resulted in upregulation of energy production genes, accompanied by a proliferation of mitochondria. Finally, a discrete set of genes, the “telomerase deletion signature”, is uniquely upregulated in the TDR but not under other conditions of stress and DNA damage. The telomerase deletion signature genes define new candidates for involvement in cellular responses to altered telomere structure or function that might mediate cell death induction. Thus, it was suggested in numerous studies, that telomerase silencing is accompanied by abundant genes expression modulation which implicates that cell death following telomerase downregulation is not always related to telomere shortening [26–28].
Telomerase
Human telomerase is a multisubunit ribonucleoprotein that belongs to the reverse transcriptase (RT) family containing RT-like motifs. It is composed of seven conservative motifs and telomerase-specific T motif [29, 30]. The catalytic telomerase protein subunit (TERT) is encoded by gene located on chromosome 5p15.33 [29]. A number of telomere binding proteins in human were identified [31–33] and shown to play crucial role in telomere protection. They allow to distinguish telomeres from damaged DNA by forming telomere structure with D- and T-loops and therefore prevent them from degradation and fusion. Additionally, the complex is safeguarded by shelterin that enables recognition of the natural chromosome ends from DNA breaks. It also regulates telomerase-based telomere maintenance. Shelterin, a very dynamic structure, is implicated in the generation of T-loops, and control of the telomeric DNA synthesis [34]. All six shelterin subunits (TRF1, TRF2, Rap1, TIN2, TPP1, and POT1) can be found in a single complex in fractionated nuclear extracts [35, 36]. The components specifically localize to telomeres. They are abundant at telomeres throughout the cell cycle and they do not function elsewhere in the nucleus. In addition, telomeres contain a large number of nonshelterin proteins which unlike the subunits of shelterin have other nontelomeric functions [34].
Regulation of telomerase activity and expression occurs at many levels, including transcription, alternative splicing, chaperone-mediated folding, phosphorylation, nuclear translocation of each subunit, assembly of the telomerase complex, and its accessibility to telomeres [37, 38]. However, as reported so far, increased telomerase activity is always correlated with upregulation of the TERT gene and an increased level of TERT mRNA. The Nobel Prize in Physiology or Medicine awarded in 2009 to Elizabeth H. Blackburn, Carol W. Greider, and Jack W. Szostak “for the discovery of how chromosomes are protected by telomeres and the enzyme telomerase” highlighted the important role of telomerase and telomeres in genome stability, immortality, aging, and cancer.
Telomerase regulation in oncology
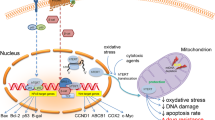
Because of significant role of telomerase in tumor development as well aging, it is of great interest to identify the enzyme expression/activity regulators. Moreover, since high telomerase expression is characteristic for stem cells but not for differentiated cells, the study of regulatory pathways that control telomerase expression may be critical for maintenance of the stem cell status. Thus, some potential side effect must be considered (Fig. 1). We do believe in significant beneficial effects of telomerase elimination in cancer cells that should be accompanied by induction of senescence, mitotic crisis, and apoptosis or autophagy. However, we are aware of the protection system of those cells which is manifested by induction of ALT.
Transactivation of telomerase, essential for cells immortalization, is supposed to be one of the reasons for cancer transformation. There are many studies describing exogenous factors influencing TERT, e.g., several viruses are known to be involved in tumorigenesis of infected tissues which was already widely discussed [5, 39, 40]. Thus, due to the complexity of the telomerase regulation, there is a great challenge on one hand, but on the other hand, there are a lot of potential ways to target and thus, stop the “immortality enzyme.”
Regulation of telomerase transcription in breast cancer
Transcriptional mechanism of telomerase regulation (Table 1) was revealed in numerous studies and abundance of transcription factors sites in TERT promoter was localized [29]. For example, involvement of c-myc in telomerase regulation was widely investigated, and its role in TERT gene expression induction was confirmed in many studies [12, 41, 42]. Additionally, both, human epidermal growth factor-2 (Her2) and ER81 (transcription factor involved in ontogenesis and breast tumor formation) [43], have been suggested to cause a synergistic increase in the transcriptional induction of TERT in breast cancer patients [44]. It was suggested that TERT gene expression could be induced by activating mutations of oncogenes such as HER2/Neu, Ras, and Raf [45]. Consequently, it was shown that HER2/Neu might become another target for a combined anticancer therapy in the context of telomerase elimination. Among other factors that contribute to telomerase expression induction, leptin (significant breast cancer risk factor) [46], epidermal growth factor receptor (EGFR) [47], and estrogen receptors ERa and ERb [48] were found. These receptors are ligand-dependent transcription factors capable of direct interaction between the hormone–receptor complex and estrogen responsive elements [49]. Their status significantly contributes to breast cancer development, diagnosis, and prognosis [50]. Estrogens were shown to activate telomerase via direct and indirect effects on the TERT promoter and suggested hormonal control of telomerase activity, cellular senescence and aging as well as estrogen-induced carcinogenesis [51]. Additionally, latest reports show that anticarcinogenic plant compound, indole-3-carbinol, that can modulate steroid hormone-mediated processes, can also trigger TERT downregulation in breast cancer MCF7 cells [52]. As suggested, this compound might induce TERT inhibition-related cell cycle arrest.
There are also some other factors that may repress telomerase in breast cancer cells. BRCA1, playing a role in maintaining genomic stability and acting as a tumor suppressor, was found to interact with c-myc (TERT inducer). It was indicated that overexpression of the BRCA1 gene inhibits TERT expression and telomerase enzymatic activity in various cell types via the inhibition of c-myc-mediated transactivation of the TERT promoter [53, 54]. Furthermore, it is suggested that BRCA1 and Nmi (N-myc and c-myc interacting protein) can form a complex with c-myc in vitro and in vivo [55]. BRCA1 is also involved in telomere shortening independently of telomerase [54, 56]. BRCA1 interacts with TRF1 and TRF2, and it is suggested that it regulates the length of the 3′ G-rich overhang by being recruited to the telomere in a Rad50-dependent manner [56]. It was demonstrated that p53 repressed telomerase activity through downregulation of TERT transcription and that interaction of p53 with Sp1 or other transcription factors may be involved in this regulation [57, 58]. The p73 protein, structural and functional homologue of p53, encoded by TP73 gene was also suggested to contribute to cell cycle regulation, apoptosis, and TERT promoter repression [59, 60]. Additionally, dexamethasone was reported to inhibit telomerase expression similarly to a “stress hormone”—cortisol [61]. Similarly, genistein (phytoestrogen) was shown to cause similar effect [62]. Among other factors that contribute to telomerase downregulation in breast cancer, raloxifene [63] and melatonin [64] were found.
Telomerase and cancer treatment at the molecular level
It is suggested that the loss of proliferative potential observed in human cells lacking telomerase may be considered as a potential mechanism helping to avoid cancer. This is hypothesized since cancer arises after multiple genetic mutations that altogether cause the cell escape from replication, migration, and growth control. Thus, a lack of telomerase would limit the growth of tumors by causing continually dividing cells to shorten their telomeres and to die before starting to spread. This is why telomerase inhibitors seem to be promising factors eliminating cancer cells (Fig. 2).
Recently, several natural-derived compounds were found to exhibit antitumor activity in the context of telomerase regulation. One of them is butein (3,4,2′,4′-tetrahydroxychalcone), a polyphenolic compound causing downregulation of telomerase through suppression of TERT gene expression. Moreover, it was demonstrated that this suppression is c-myc-dependent, because butein prevents c-myc binding to TERT promoter [65] which is followed by TERT expression decrease. Comparing telomerase-positive and ALT cells, it was found that c-myc activity was lower in ALT cells but there was no difference in the gene expression. Interestingly, it was demonstrated that leptin activated telomerase in a dose-dependent manner, upregulated the expression of TERT at mRNA and protein levels. On the other hand, the blockade of signal transducer and activator of transcription 3 (STAT3) phosphorylation significantly counteracted leptin-induced TERT transcription and protein expression. Thus, a new mechanism of the proliferative effect of leptin on breast cancer cells was suggested, and a new explanation of obesity-related breast cancer was hypothesized [46].
Another compound, pectenotoxin-2 (PTX2), most toxic compound from the group of pectenotoxins (found in marine sponges and shellfish) was shown to downregulate telomerase activity through suppression of TERT gene expression at the transcriptional level. It was shown that PTX2-dependent attenuation of TERT expression was mediated through suppression of c-myc- and Sp1-binding on the regulatory regions of TERT [66]. Both factors influenced telomerase also on posttranscriptional level by attenuation of Akt phosphorylation, thereby reducing the phosphorylation and nuclear translocation of TERT [65, 66]. The investigation of the effects of PTX on human breast cancer cells (MDA-MB-231 and MCF7) revealed a significant suppression of cell proliferation and induction of G2/M phase arrest through downregulation of cyclin B1 and cdc2 expression, as well as through phosphorylation of cdc25C. The increased phosphorylation of ATM and Chk1/2 in a PTX2 dose-dependent manner was also found. Furthermore, treatment with PTX2 increased H2O2 generation with correlated G2/M arrest revealing that PTX2-induced cell cycle arrest was not restricted to p53 status in human breast cancer cells [67]. As demonstrated by Heaphy et al. [68], telomeres were shorter in more aggressive subtypes, such as luminal B, HER-2-positive and triple-negative tumors, suggesting that tumor telomere length may have utility as a prognostic and/or risk marker for breast cancer. Joseph et al. [69] revealed that when breast and pancreatic cancer cell lines were treated with potent telomerase inhibitor, imetelstat (oligonucleotide, direct enzyme inhibitor) in vitro, telomerase activity in the bulk tumor cells and cancer stem cells subpopulations was inhibited. In vitro treatment with imetelstat, but not control oligonucleotides, also reduced the proliferation and self-renewal potential of MCF7 mammospheres and resulted in cell death after <4 weeks of treatment.
As reported by Moon et al. [65], gefitinib downregulated the activation of Akt and subsequent TERT phosphorylation and translocation into the nucleus in MDA-MB-231 cells. These results indicate that gefitinib induces loss of telomerase activity through dephosphorylation of EGFR in MDA-MB-231 cells showing high expression of EGFR. Similarly, Papanikolaou et al. [70] showed that silencing of TERT and HER2 achieved by small interfering RNA technology increased radiosensitivity of cells. Knockdown of HER2 also led to increased radiosensitivity and downregulation of TERT/telomerase. It was confirmed that c-myc and mad1 regulated TERT expression in all irradiated breast cancer cells. Additionally, it was shown that circulating TERT DNA had a better diagnostic value than carbohydrate antigen CA 15.3 in early breast cancer disease and could be a possible candidate as a tumor marker in patients with infiltrating ductal carcinoma positive to steroid hormonal receptor and with amplification of HER-2/Neu [71]. Another natural compound, genistein, was reported as one of the most promising natural compounds targeting telomerase. This natural isoflavone found in soybean products has been reported to downregulate telomerase activity, to prevent cancer and to contribute to the apoptosis of cancer cells. It was shown that genistein inhibited the transcription of TERT in breast MCF10AT benign and MCF7 cancer cells in a time- and dose-dependent manner. Three major DNA methyltransferases (DNMTs; 1, 3a, and 3b) were decreased in genistein-treated breast cancer cells, suggesting that this compound might repress TERT by impacting epigenetic pathways. Thus, it was concluded that genistein might work, at least in part, through epigenetic mechanisms of telomerase inhibition and might facilitate approaches to breast cancer prevention and treatment in a combined therapy. Another compound showing potent telomerase inhibition is all-trans retinoic acid (ATRA) which was reported to have significant effect on the morphology and proliferation rate of the breast cancer SK-BR-3 cells [72]. It was shown to decrease the cancer cell growth and lower telomerase activity which was associated with a rapid decrease in histone H3-lysine 9 acetylation (H3-K9-Ac) of the TERT promoter. Further ATRA treatment was shown to induce apoptosis in estrogen receptor-negative breast cancer cells [72]. Another telomerase-inhibiting natural compound, sulforaphane (SFN), an isothiocyanate found in cruciferous vegetables is a common dietary component that reveals histone deacetylase inhibition activity and significant potential in cancer prevention. The mechanism of action is supposed to act through the decrease of DNMTs, especially DNMT1 and DNMT3a, suggesting that SFN may repress TERT by impacting epigenetic pathways [73]. Similarly, trichostatin A, an inhibitor of histone deacetylase, is a well-known antitumor agent that effectively and selectively induces tumor growth arrest and apoptosis.
Telomerase, drug resistance, and therapy
Resistance to chemotherapy is one of the principal causes of cancer mortality and generally appears in a late tumor progression process. Cellular models of drug resistance have been useful in identifying the molecular mechanism of this phenomenon and most of them are derived from a late stage of cancer. As suggested, the ability to acquire multidrug resistance (MDR) can arise before the malignant transformation stage and is preceded by expression of telomerase and inactivation of p53 and pRb. Thus, the pathways inactivated during tumorigenesis confer the ability to acquire drug resistance [74]. Telomerase catalytic subunit expression shows correlations with MDR- and apoptosis-related genes and is of prognostic significance [75, 76]. The contribution of telomerase to multidrug resistance was suggested to result from increased chromosome stability and a strong expression of both ABCB1 and ABCC genes [77]. As reported by Ji et al. [78], TR antisense oligonucleotides could downregulate the telomerase activity, increase radiation-induced DNA damage, and reduce the subsequent repair. Furthermore, it could inhibit the proliferation and increase cancer cell radiosensitivity; however, without telomere shortening. Similarly, Cerone et al. [18] demonstrated that mutated TR coding subunit resulted in an increased sensitivity of breast cancer cells to anticancer drugs. It was shown in cells with different initial telomere lengths and different mechanisms of telomere maintenance. Authors suggested that dysfunctional TR coding gene affects the binding of the shelterin complex, disturbs telomere capping, and induces a DNA damage response. It has been proposed that the mutant TR might interfere with alternative functions of telomerase [79] especially since the ability to synthesize new telomeric repeats is essential for the mutant TR-dependent effects.
Furthermore, Massard et al. [80] demonstrated that TERT repression may constitute a promising strategy for facilitating the induction of the mitochondrial pathway of apoptosis. Other researchers showed that BIBR1532 (2-[(E)-3-naphtalen-2-yl-but-2-enoylamino]-benzoic acid)-treated cells show progressive telomere shortening, decreased proliferative capacity, and sensitization to chemotherapeutic treatment [81]. It was postulated to be provoked by potent, selective, and reversible inhibitory action on human telomerase activity. This suggested that pharmacological telomerase inhibition in combination therapy may be a valid strategy for the treatment of both drug-sensitive and drug-resistant breast cancers. Interestingly, the combined effect of low-dose doxorubicin and siRNA inhibition of telomerase on breast cancer cells caused a rapid and potent apoptosis induction in breast cancer cells. The effect of doxorubicin was potentiated by the RNA interference directed against telomerase subunits [82]. Although numerous telomerase inhibition strategies cause cancer cells to undergo apoptosis or senescence, there is often a lag period between the beginning of the treatment regimen and a biological effect. Thus, much better perspectives are expected when applying effective telomerase inhibitors together with standard chemotherapeutic agents, Doxorubicin/Adriamycin or Taxol. Such attitude is supposed to provoke an increased sensitization and efficacy for triggering senescence and/or apoptosis in cultures of breast cancer cells while reducing toxicity [83].
First trials with oligonucleotides against telomerase showed that methoxyethyl oligomers (2′-O-methoxyethyl oligonucleotide) directed against the template region of telomerase are potent agents and that significant antiproliferative effects can be observed after 2–3 weeks of treatment. Reduced cell proliferation and tumor growth support the hypothesis that telomerase inhibition can make a useful contribution to chemotherapy and should encourage broad testing of telomerase inhibitors [84]. One of the most advanced telomerase-targeting strategies against cancer are represented by Geron (Menlo Park, Ca, USA). The technology being at the clinical trials at the moment is based on the oligonucleotides, called imetelstat sodium (originally known as GRN163L). GRN163L, a telomerase template antagonist, was effective in inhibiting telomerase activity and shortening telomeres in HER2-positive breast cancer cells [85]. It acts synergistically with trastuzumab in inhibiting HER2-positive MDA-MB-231 breast cancer cell growth. More importantly, it was shown that GRN163L can restore the sensitivity of therapy-resistant breast cancer cells to trastuzumab. These findings implicate that telomerase template antagonists might have a potential use in the treatment of cancers that have developed resistance to traditional cancer therapy [86]. It was also reported that breast cancer cells treated with GRN163L exhibited significantly diminished invasive potential [87]. As shown, long-term continuous treatment of cells with GRN163L results in progressive telomere shortening. It demonstrated that transient inhibition of telomerase activity by the specific inhibitor, GRN163L, increased the cytotoxicity of some, but not all, DNA-damaging agents. These results suggest that the protective role of telomerase in cell cycle-restricted DNA damage repair could be exploited for combined anticancer chemotherapy [88]. Recently, it was reported that Phase I clinical trials in breast cancer patients treated with GRN163L were successfully completed and the agent was selected for Phase II clinical trials. As it was demonstrated, the compound is characterized by promising bioavailability and pharmacokinetics. Phase II tests are currently ongoing and Imetelstat is tested in a combination with standard chemotherapeutic drugs (bevacizumab and paclitaxel) in a group of patients with locally relapsing or metastatic breast cancer [89].
Another group of telomerase-targeting drugs demonstrated by Geron are telomerase cancer vaccines—GRNVAC1 and GRNVAC2. They are autologous products consisting of mature dendritic cells (antigen-presenting cells) pulsed with RNA for the protein component of human telomerase (TERT) and a portion of a lysosomal targeting signal (LAMP). GRNVAC1 is injected into the patient’s skin. From there, the dendritic cells travel to the lymph nodes and instruct cytotoxic T cells to kill tumor cells that express telomerase. Alternatively, Geron proposed another solution (GRN1005) based on peptide technology to facilitate the transfer of anticancer compounds across the blood–brain barrier (BBB) to enable the treatment of primary brain cancers and cancers that have metastasized to the brain. It is not connected with telomerase directly; however, due to the connection of telomerase with multidrug resistance, this mechanism of action cannot be totally excluded [85]. It is known that telomerase can function in a number of alternative signaling mechanisms and some of them could be involved in chemotherapeutic resistance, including DNA damage response, and DNA repair [90]. Thus, it is suggested that upregulation of telomerase itself, independent of its role in telomere maintenance, is capable of providing an alternative pathway to drug resistance that does not require elongating telomere lengths [83]. In the light of carcinogenesis complexity and the contribution of mutations in many genes to the breast cancer development [91], it should be emphasized that monofunctional-targeted drugs will not cure most of the patients with common carcinomas. Thus, the multifunctional drugs or alternative treatment regimens are needed. Possibly telomerase inhibitors could be administered as adjuvant therapy in combination with conventional drugs to improve the efficiency of chemotherapeutic agents used in the low doses.
Summary and conclusions—prospects for cancer therapy
The expression of telomerase in various human cancers and its diminished expression in most normal cells suggest that the enzyme might serve as a good target for anticancer drugs. Due to telomerase inhibition, activity, or expression, these drugs might kill tumor cells by allowing telomeres to shrink or by provoking apoptosis. First of all, this process might have a chance to be cell-specific without serious side effects (Fig. 1). In contrast, most existing anticancer therapies disturb normal cells as well as malignant ones and often show some serious toxicity. Since telomerase seems to be a universal cancer marker, the agents targeting the enzyme might work against a broad spectrum of tumors. Nevertheless, a number of questions must be answered. For instance, researchers need to determine which normal cells (beyond the few already identified) reveal telomerase expression/activity and in what extent. They also need to assess the importance of the enzyme to those cells. Telomerase-inhibiting agents might cause cancer cells to lose their telomeres and die well before normal cells, with their much longer telomeres, lose enough of their telomeres to suffer any ill effects. It should be unquestionably demonstrated that inhibition of telomerase can destroy telomerase-producing tumors as expected. Another problem might be the delivery of telomerase-targeting agents in a way that would provide high efficiency and specificity. Therapeutic potential of vaccination has been explored in many clinical trials involving patients with breast cancer. A large variety of cancer immunogens have been tested. The majority of clinical vaccination studies have been carried out in patients with metastatic breast cancer, characterized by extremely aggressive malignant tumors, resistant to all standard cytotoxic treatments, and with longest-lasting disease. However, it is unclear whether therapeutic vaccines for cancer prolong survival. Data of clinical activity have been observed by using vaccines targeting HER-2/neu protein, human telomerase reverse transcriptase, carcinoembryonic antigen, and carbohydrate antigen given after stem cell rescue [92].
Intensive studies of telomerase functioning in human cells gave new perspectives on the mechanism of senescence, stem cells, and cancer therapy. The studies show that numerous enzymes are required for telomerase functioning that facilitate new approaches for inhibiting telomerase in treating cancer. Probably there are still numerous unrevealed proteins that contribute to regulation of such a dynamic complex. Anyway, it is still supposed to use both, activity and expression regulation methods, as adjuvant therapies similarly to G-quadruplex stabilization. Understanding of telomerase activity may then bring a new insight into many serious clinical problems that we have to face in aging societies especially since telomerase might be also considered as a predictive marker maybe even in malignant transformation. It is still unclear whether telomerase inhibitors will become a treatment option against cancer. There is concern about the alternative mechanisms of telomere maintenance and whether there will be side effects on normal, hematopoietic, and germline cells. These questions, however, can be only answered if those drugs are moved into animal and human clinical trials.
References
Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585–621.
Dimri GP. What has senescence got to do with cancer? Cancer Cell. 2005;7:505–12.
Narita M, Lowe SW. Senescence comes of age. Nat Med. 2005;11:920–2.
Uziel O, Beery E, Dronichev V, Samocha K, et al. Telomere shortening sensitizes cancer cells to selected cytotoxic agents: in vitro and in vivo studies and putative mechanisms. PLoS One. 2010;5:e9132.
Horikawa I, Barrett JC. Transcriptional regulation of the telomerase hTERT gene as a target for cellular and viral oncogenic mechanisms. Carcinogenesis. 2003;24:1167–76.
Papanikolaou V, Athanassiou E, Dubos S, Dimou I, et al. hTERT regulation by NF-kappaB and c-myc in irradiated HER2-positive breast cancer cells. Int J Radiat Biol. 2011;87:609–21.
Li AY, Lin HH, Kuo CY, Shih HM, et al. High-mobility group A2 protein modulates hTERT transcription to promote tumorigenesis. Mol Cell Biol. 2011;31:2605–17.
Donate LE, Blasco MA. Telomeres in cancer and ageing. Philos Trans R Soc Lond B Biol Sci. 2011;366:76–84.
Park KD, Seong SK, Park YM, Choi Y, et al. Telomerase reverse transcriptase related with telomerase activity regulates tumorigenic potential of mouse embryonic stem cells. Stem Cells Dev. 2011;20:149–57.
Sahin E, Depinho RA. Linking functional decline of telomeres, mitochondria and stem cells during ageing. Nature. 2010;464:520–8.
Wang X, Hao MW, Dong K, Lin F, et al. Apoptosis induction effects of EGCG in laryngeal squamous cell carcinoma cells through telomerase repression. Arch Pharm Res. 2009;32:1263–9.
Li X, Liu Y, Wen Z, Li C, et al. Potent anti-tumor effects of a dual specific oncolytic adenovirus expressing apoptin in vitro and in vivo. Mol Cancer. 2010;9:10.
Rahman R, Grundy R. Histone deacetylase inhibition as an anticancer telomerase-targeting strategy. Int J Cancer. 2011;12:2765–74.
Lu Y, Gu J, Jin D, Gao Y, et al. Inhibition of telomerase activity by HDV ribozyme in cancers. J Exp Clin Cancer Res. 2011;30:1.
Kim NW. Clinical implications of telomerase in cancer. Eur J Cancer. 1997;33:781–6.
Shay JW, Bacchetti S. A survey of telomerase activity in human cancer. Eur J Cancer. 1997;33:787–91.
Bryan TM, Englezou A, Gupta J, Bacchetti S, et al. Telomere elongation in immortal human cells without detectable telomerase activity. EMBO J. 1995;14:4240–8.
Cerone MA, Londono-Vallejo JA, Autexier C. Mutated telomeres sensitize tumor cells to anticancer drugs independently of telomere shortening and mechanisms of telomere maintenance. Oncogene. 2006;25:7411–20.
Wang Z, Kyo S, Takakura M, Tanaka M, et al. Progesterone regulates human telomerase reverse transcriptase gene expression via activation of mitogen-activated protein kinase signaling pathway. Cancer Res. 2000;60:5376–81.
Neidle S. Therapeutic Application of Quadruplex Nucleic Acids. Elsevier. 2012
Rubis B, Kaczmarek M, Szymanowska N, Galezowska E, et al. The biological activity of G-quadruplex DNA binding papaverine-derived ligand in breast cancer cells. Invest New Drugs. 2009;27:289–96.
Kasiappan R, Shen Z, Tse AK, Jinwal U, et al. 1,25-Dihydroxyvitamin D3 suppresses telomerase expression and human cancer growth through microRNA-498. J Biol Chem. 2012;287:41297–309.
Xiao X, Sidorov IA, Gee J, Lempicki RA, et al. Retinoic acid-induced downmodulation of telomerase activity in human cancer cells. Exp Mol Pathol. 2005;79:108–17.
Purev E, Soprano DR, Soprano KJ. Effect of all-trans retinoic acid on telomerase activity in ovarian cancer cells. J Exp Clin Cancer Res. 2004;23:309–16.
Nanni S, Narducci M, Della Pietra L, Moretti F, et al. Signaling through estrogen receptors modulates telomerase activity in human prostate cancer. J Clin Invest. 2002;110:219–27.
Nautiyal S, DeRisi JL, Blackburn EH. The genome-wide expression response to telomerase deletion in Saccharomyces cerevisiae. Proc Natl Acad Sci U S A. 2002;99:9316–21.
Elmore LW, Rehder CW, Di X, McChesney PA, et al. Adriamycin-induced senescence in breast tumor cells involves functional p53 and telomere dysfunction. J Biol Chem. 2002;277:35509–15.
Shammas MA, Koley H, Batchu RB, Bertheau RC, et al. Telomerase inhibition by siRNA causes senescence and apoptosis in Barrett’s adenocarcinoma cells: mechanism and therapeutic potential. Mol Cancer. 2005;4:24.
Meyerson M, Counter CM, Eaton EN, Ellisen LW, et al. hEST2, the putative human telomerase catalytic subunit gene, is up-regulated in tumor cells and during immortalization. Cell. 1997;90:785–95.
Nakamura TM, Morin GB, Chapman KB, Weinrich SL, et al. Telomerase catalytic subunit homologs from fission yeast and human. Science. 1997;277:955–9.
Giannone RJ, McDonald HW, Hurst GB, Shen RF, et al. The protein network surrounding the human telomere repeat binding factors TRF1, TRF2, and POT1. PLoS One. 2010;5:e12407.
de Lange T. Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev. 2005;19:2100–10.
Chong L, van Steensel B, Broccoli D, Erdjument-Bromage H, et al. A human telomeric protein. Science. 1995;270:1663–7.
Palm W, de Lange T. How shelterin protects mammalian telomeres. Annu Rev Genet. 2008;42:301–34.
Ye JZ, Donigian JR, van Overbeek M, Loayza D, et al. TIN2 binds TRF1 and TRF2 simultaneously and stabilizes the TRF2 complex on telomeres. J Biol Chem. 2004;279:47264–71.
Wang F, Podell ER, Zaug AJ, Yang Y, et al. The POT1-TPP1 telomere complex is a telomerase processivity factor. Nature. 2007;445:506–10.
Cong YS, Wen J, Bacchetti S. The human telomerase catalytic subunit hTERT: organization of the gene and characterization of the promoter. Hum Mol Genet. 1999;8:137–42.
Wojtyla A, Gladych M, Rubis B. Human telomerase activity regulation. Mol Biol Rep. 2010;38:3339–49.
Bellon M, Nicot C. Regulation of telomerase and telomeres: human tumor viruses take control. J Natl Cancer Inst. 2008;100:98–108.
Straat K, Liu C, Rahbar A, Zhu Q, et al. Activation of telomerase by human cytomegalovirus. J Natl Cancer Inst. 2009;101:488–97.
Fujiki T, Udono M, Kadooka K, Yamashita S, et al. Regulatory mechanisms of human and mouse telomerase reverse transcriptase gene transcription: distinct dependency on c-Myc. Cytotechnology. 2010;62:333–9.
Akiyama M, Kawano T, Mikami-Terao Y, Agawa-Ohta M, et al. Erythropoietin activates telomerase through transcriptional and posttranscriptional regulation in human erythroleukemic JAS-REN-A cells. Leuk Res. 2010;35:416–8.
Bosc DG, Janknecht R. Regulation of Her2/neu promoter activity by the ETS transcription factor, ER81. J Cell Biochem. 2002;86:174–83.
Vageli D, Ioannou MG, Koukoulis GK. Transcriptional activation of hTERT in breast carcinomas by the Her2-ER81-related pathway. Oncol Res. 2009;17:413–23.
Goueli BS, Janknecht R. Upregulation of the catalytic telomerase subunit by the transcription factor ER81 and oncogenic HER2/Neu, Ras, or Raf. Mol Cell Biol. 2004;24:25–35.
Ren H, Zhao T, Wang X, Gao C, et al. Leptin upregulates telomerase activity and transcription of human telomerase reverse transcriptase in MCF-7 breast cancer cells. Biochem Biophys Res Commun. 2010;394:59–63.
Kunimura C, Kikuchi K, Ahmed N, Shimizu A, et al. Telomerase activity in a specific cell subset co-expressing integrinbeta1/EGFR but not p75NGFR/bcl2/integrin beta4 in normal human epithelial cells. Oncogene. 1998;17:187–97.
Misiti S, Nanni S, Fontemaggi G, Cong YS, et al. Induction of hTERT expression and telomerase activity by estrogens in human ovary epithelium cells. Mol Cell Biol. 2000;20:3764–71.
Beato M, Sanchez-Pacheco A. Interaction of steroid hormone receptors with the transcription initiation complex. Endocr Rev. 1996;17:587–609.
Gladych M, Wojtyla A, Rubis B. Human telomerase expression regulation. Biochem Cell Biol. 2011;89:359–76.
Kyo S, Takakura M, Kanaya T, Zhuo W, et al. Estrogen activates telomerase. Cancer Res. 1999;59:5917–21.
Marconett CN, Sundar SN, Tseng M, Tin AS, et al. Indole-3-carbinol downregulation of telomerase gene expression requires the inhibition of estrogen receptor-alpha and Sp1 transcription factor interactions within the hTERT promoter and mediates the G1 cell cycle arrest of human breast cancer cells. Carcinogenesis. 2011;32:1315–23.
Zhou C, Liu J. Inhibition of human telomerase reverse transcriptase gene expression by BRCA1 in human ovarian cancer cells. Biochem Biophys Res Commun. 2003;303:130–6.
Xiong J, Fan S, Meng Q, Schramm L, et al. BRCA1 inhibition of telomerase activity in cultured cells. Mol Cell Biol. 2003;23:8668–90.
Li H, Lee TH, Avraham H. A novel tricomplex of BRCA1, Nmi, and c-Myc inhibits c-Myc-induced human telomerase reverse transcriptase gene (hTERT) promoter activity in breast cancer. J Biol Chem. 2002;277:20965–73.
Ballal RD, Saha T, Fan S, Haddad BR, et al. BRCA1 localization to the telomere and its loss from the telomere in response to DNA damage. J Biol Chem. 2009;284:36083–98.
Kanaya T, Kyo S, Hamada K, Takakura M, et al. Adenoviral expression of p53 represses telomerase activity through down-regulation of human telomerase reverse transcriptase transcription. Clin Cancer Res. 2000;6:1239–47.
Toh WH, Kyo S, Sabapathy K. Relief of p53-mediated telomerase suppression by p73. J Biol Chem. 2005;280:17329–38.
Jost CA, Marin MC, Kaelin Jr WG. p73 is a simian [correction of human] p53-related protein that can induce apoptosis. Nature. 1997;389:191–4.
Kaghad M, Bonnet H, Yang A, Creancier L, et al. Monoallelically expressed gene related to p53 at 1p36, a region frequently deleted in neuroblastoma and other human cancers. Cell. 1997;90:809–19.
Akiyama M, Hideshima T, Hayashi T, Tai YT, et al. Cytokines modulate telomerase activity in a human multiple myeloma cell line. Cancer Res. 2002;62:3876–82.
Li Y, Liu L, Andrews LG, Tollefsbol TO. Genistein depletes telomerase activity through cross-talk between genetic and epigenetic mechanisms. Int J Cancer. 2009;125:286–96.
Kawagoe J, Ohmichi M, Takahashi T, Ohshima C, et al. Raloxifene inhibits estrogen-induced up-regulation of telomerase activity in a human breast cancer cell line. J Biol Chem. 2003;278:43363–72.
Martinez-Campa CM, Alonso-Gonzalez C, Mediavilla MD, Cos S, et al. Melatonin down-regulates hTERT expression induced by either natural estrogens (17beta-estradiol) or metalloestrogens (cadmium) in MCF-7 human breast cancer cells. Cancer Lett. 2008;268:272–7.
Moon DO, Kim MO, Heo MS, Lee JD, et al. Gefitinib induces apoptosis and decreases telomerase activity in MDA-MB-231 human breast cancer cells. Arch Pharm Res. 2009;32:1351–60.
Kim MO, Moon DO, Kang SH, Heo MS, et al. Pectenotoxin-2 represses telomerase activity in human leukemia cells through suppression of hTERT gene expression and Akt-dependent hTERT phosphorylation. FEBS Lett. 2008;582:3263–9.
Moon DO, Kim MO, Nam TJ, Kim SK, et al. Pectenotoxin-2 induces G2/M phase cell cycle arrest in human breast cancer cells via ATM and Chk1/2-mediated phosphorylation of cdc25C. Oncol Rep. 2010;24:271–6.
Heaphy CM, Subhawong AP, Gross AL, Konishi Y, et al. Shorter telomeres in luminal B, HER-2 and triple-negative breast cancer subtypes. Mod Pathol. 2011;24:194–200.
Joseph I, Tressler R, Bassett E, Harley C, et al. The telomerase inhibitor imetelstat depletes cancer stem cells in breast and pancreatic cancer cell lines. Cancer Res. 2010;70:9494–504.
Papanikolaou V, Iliopoulos D, Dimou I, Dubos S, et al. The involvement of HER2 and p53 status in the regulation of telomerase in irradiated breast cancer cells. Int J Oncol. 2009;35:1141–9.
Divella R, Tommasi S, Lacalamita R, Daniele A, et al. Circulating hTERT DNA in early breast cancer. Anticancer Res. 2009;29:2845–9.
Phipps SM, Love WK, White T, Andrews LG, et al. Retinoid-induced histone deacetylation inhibits telomerase activity in estrogen receptor-negative breast cancer cells. Anticancer Res. 2009;29:4959–64.
Meeran SM, Patel SN, Tollefsbol TO. Sulforaphane causes epigenetic repression of hTERT expression in human breast cancer cell lines. PLoS One. 2010;5:e11457.
Yague E, Arance A, Kubitza L, O'Hare M, et al. Ability to acquire drug resistance arises early during the tumorigenesis process. Cancer Res. 2007;67:1130–7.
Wang J, Liu X, Fang J. Expression and clinical significance of telomerase catalytic subunit gene in lung cancer and its correlations with genes related to drug resistance and apoptosis. Zhonghua Zhong Liu Za Zhi. 1999;21:350–3.
Siva AC, Raval-Fernandes S, Stephen AG, LaFemina MJ, et al. Up-regulation of vaults may be necessary but not sufficient for multidrug resistance. Int J Cancer. 2001;92:195–202.
Kuranaga N, Shinomiya N, Mochizuki H. Long-term cultivation of colorectal carcinoma cells with anti-cancer drugs induces drug resistance and telomere elongation: an in vitro study. BMC Cancer. 2001;1:10.
Ji XM, Xie CH, Fang MH, Zhou FX, et al. Efficient inhibition of human telomerase activity by antisense oligonucleotides sensitizes cancer cells to radiotherapy. Acta Pharmacol Sin. 2006;27:1185–91.
Chung HK, Cheong C, Song J, Lee HW. Extratelomeric functions of telomerase. Curr Mol Med. 2005;5:233–41.
Massard C, Zermati Y, Pauleau AL, Larochette N, et al. hTERT: a novel endogenous inhibitor of the mitochondrial cell death pathway. Oncogene. 2006;25:4505–14.
Ward RJ, Autexier C. Pharmacological telomerase inhibition can sensitize drug-resistant and drug-sensitive cells to chemotherapeutic treatment. Mol Pharmacol. 2005;68:779–86.
Dong X, Liu A, Zer C, Feng J, et al. siRNA inhibition of telomerase enhances the anti-cancer effect of doxorubicin in breast cancer cells. BMC Cancer. 2009;9:133.
Poynter KR, Sachs PC, Bright AT, Breed MS, et al. Genetic inhibition of telomerase results in sensitization and recovery of breast tumor cells. Mol Cancer Ther. 2009;8:1319–27.
Chen Z, Koeneman KS, Corey DR. Consequences of telomerase inhibition and combination treatments for the proliferation of cancer cells. Cancer Res. 2003;63:5917–25.
Goldblatt EM, Gentry ER, Fox MJ, Gryaznov SM, et al. The telomerase template antagonist GRN163L alters MDA-MB-231 breast cancer cell morphology, inhibits growth, and augments the effects of paclitaxel. Mol Cancer Ther. 2009;8:2027–35.
Gellert GC, Dikmen ZG, Wright WE, Gryaznov S, et al. Effects of a novel telomerase inhibitor, GRN163L, in human breast cancer. Breast Cancer Res Treat. 2006;96:73–81.
Tamakawa RA, Fleisig HB, Wong JM. Telomerase inhibition potentiates the effects of genotoxic agents in breast and colorectal cancer cells in a cell cycle-specific manner. Cancer Res. 2010;70:8684–94.
Buseman CM, Wright WE, Shay JW. Is telomerase a viable target in cancer? Mutat Res. 2012;730:90–7.
Cong Y, Shay JW. Actions of human telomerase beyond telomeres. Cell Res. 2008;18:725–32.
Sjoblom T, Jones S, Wood LD, Parsons DW, et al. The consensus coding sequences of human breast and colorectal cancers. Science. 2006;314:268–74.
Curigliano G, Locatelli M, Fumagalli L, Goldhirsch A. Immunizing against breast cancer: a new swing for an old sword. Breast. 2009;18 Suppl 3:S51–4.
Gaudray G, Gachon F, Basbous J, Biard-Piechaczyk M, et al. The complementary strand of the human T-cell leukemia virus type 1 RNA genome encodes a bZIP transcription factor that down-regulates viral transcription. J Virol. 2002;76:12813–22.
Knight JS, Cotter 2nd MA, Robertson ES. The latency-associated nuclear antigen of Kaposi’s sarcoma-associated herpesvirus transactivates the telomerase reverse transcriptase promoter. J Biol Chem. 2001;276:22971–8.
Furuya M, Tsuji N, Kobayashi D, Watanabe N. Interaction between survivin and aurora-B kinase plays an important role in survivin-mediated up-regulation of human telomerase reverse transcriptase expression. Int J Oncol. 2009;34:1061–8.
Xu D, Wang Q, Gruber A, Bjorkholm M, et al. Downregulation of telomerase reverse transcriptase mRNA expression by wild type p53 in human tumor cells. Oncogene. 2000;19:5123–33.
Beitzinger M, Oswald C, Beinoraviciute-Kellner R, Stiewe T. Regulation of telomerase activity by the p53 family member p73. Oncogene. 2006;25:813–26.
Wang J, Xie LY, Allan S, Beach D, et al. Myc activates telomerase. Genes Dev. 1998;12:1769–74.
Wu KJ, Grandori C, Amacker M, Simon-Vermot N, et al. Direct activation of TERT transcription by c-MYC. Nat Genet. 1999;21:220–4.
Dwyer J, Li H, Xu D, Liu JP. Transcriptional regulation of telomerase activity: roles of the Ets transcription factor family. Ann N Y Acad Sci. 2007;1114:36–47.
Block TM, Mehta AS, Fimmel CJ, Jordan R. Molecular viral oncology of hepatocellular carcinoma. Oncogene. 2003;22:5093–107.
Konnikova L, Simeone MC, Kruger MM, Kotecki M, et al. Signal transducer and activator of transcription 3 (STAT3) regulates human telomerase reverse transcriptase (hTERT) expression in human cancer and primary cells. Cancer Res. 2005;65:6516–20.
Oh ST, Kyo S, Laimins LA. Telomerase activation by human papillomavirus type 16 E6 protein: induction of human telomerase reverse transcriptase expression through Myc and GC-rich Sp1 binding sites. J Virol. 2001;75:5559–66.
Hiyama E, Hiyama K. Clinical utility of telomerase in cancer. Oncogene. 2002;21:643–9.
Kim NW, Piatyszek MA, Prowse KR, Harley CB, et al. Specific association of human telomerase activity with immortal cells and cancer. Science. 1994;266:2011–5.
Oh S, Song Y, Yim J, Kim TK. The Wilms’ tumor 1 tumor suppressor gene represses transcription of the human telomerase reverse transcriptase gene. J Biol Chem. 1999;274:37473–8.
Lin SY, Elledge SJ. Multiple tumor suppressor pathways negatively regulate telomerase. Cell. 2003;113:881–9.
Zhou C, Bae-Jump VL, Whang YE, Gehrig PA, et al. The PTEN tumor suppressor inhibits telomerase activity in endometrial cancer cells by decreasing hTERT mRNA levels. Gynecol Oncol. 2006;101:305–10.
McMurray HR, McCance DJ. Human papillomavirus type 16 E6 activates TERT gene transcription through induction of c-Myc and release of USF-mediated repression. J Virol. 2003;77:9852–61.
Jagadeesh S, Banerjee PP. Inositol hexaphosphate represses telomerase activity and translocates TERT from the nucleus in mouse and human prostate cancer cells via the deactivation of Akt and PKCalpha. Biochem Biophys Res Commun. 2006;349:1361–7.
Goueli BS, Janknecht R. Regulation of telomerase reverse transcriptase gene activity by upstream stimulatory factor. Oncogene. 2003;22:8042–7.
Uziel O, Fenig E, Nordenberg J, Beery E, et al. Imatinib mesylate (Gleevec) downregulates telomerase activity and inhibits proliferation in telomerase-expressing cell lines. Br J Cancer. 2005;92:1881–91.
Fujimoto K, Kyo S, Takakura M, Kanaya T, et al. Identification and characterization of negative regulatory elements of the human telomerase catalytic subunit (hTERT) gene promoter: possible role of MZF-2 in transcriptional repression of hTERT. Nucleic Acids Res. 2000;28:2557–62.
Yang Y, Cai S, Yang L, Yu S, et al. Targeting eradication of malignant cells derived from human bone marrow mesenchymal stromal cells. Exp Cell Res. 2010;316:3329–41.
Akutagawa O, Nishi H, Kyo S, Terauchi F, et al. Early growth response-1 mediates downregulation of telomerase in cervical cancer. Cancer Sci. 2008;99:1401–6.
Racek T, Mise N, Li Z, Stoll A, et al. C-terminal p73 isoforms repress transcriptional activity of the human telomerase reverse transcriptase (hTERT) promoter. J Biol Chem. 2005;280:40402–5.
Gewin L, Myers H, Kiyono T, Galloway DA. Identification of a novel telomerase repressor that interacts with the human papillomavirus type-16 E6/E6-AP complex. Genes Dev. 2004;18:2269–82.
Lee SH, Kim JW, Oh SH, Kim YJ, et al. IFN-gamma/IRF-1-induced p27kip1 down-regulates telomerase activity and human telomerase reverse transcriptase expression in human cervical cancer. FEBS Lett. 2005;579:1027–33.
Renaud S, Loukinov D, Bosman FT, Lobanenkov V, et al. CTCF binds the proximal exonic region of hTERT and inhibits its transcription. Nucleic Acids Res. 2005;33:6850–60.
Renaud S, Loukinov D, Abdullaev Z, Guilleret I, et al. Dual role of DNA methylation inside and outside of CTCF-binding regions in the transcriptional regulation of the telomerase hTERT gene. Nucleic Acids Res. 2007;35:1245–56.
Ogawa D, Nomiyama T, Nakamachi T, Heywood EB, et al. Activation of peroxisome proliferator-activated receptor gamma suppresses telomerase activity in vascular smooth muscle cells. Circ Res. 2006;98:e50–9.
Acknowledgments
The present review was supported by 2011/03/B/NZ7/00512 research grant.
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Holysz, H., Lipinska, N., Paszel-Jaworska, A. et al. Telomerase as a useful target in cancer fighting—the breast cancer case. Tumor Biol. 34, 1371–1380 (2013). https://doi.org/10.1007/s13277-013-0757-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-013-0757-4