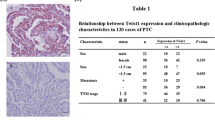
Abstract
EphB4 tyrosine kinase receptor has been involved in various physiologic and pathologic processes, and the role of the EphB4 in tumorigenesis has recently attracted much interest. However, its function in papillary thyroid carcinoma remains poorly understood. In this study, we explored the function of EphB4 in papillary thyroid carcinoma. We found that the expression of EphB4 was significantly upregulated in clinical samples. Overexpression of EphB4 in papillary thyroid carcinoma cell lines accelerated cell migration. In contrast, downregulation of EphB4 inhibited cell migration and suppressed in vivo tumor metastasis. Furthermore, we showed that EphB4 promoted cell migration by inhibiting the phosphorylation of FAK and paxillin. Moreover, EphB4 promoted cell migration in a kinase-independent manner. Taken together, our findings suggest that EphB4 plays an important role in the progression of papillary thyroid carcinoma by stimulating cell migration and EphB4 might be a potential therapeutic target in papillary thyroid carcinoma.





Similar content being viewed by others
References
Hundahl SA, Fleming ID, Fremgen AM, et al. A National Cancer Data Base report on 53,856 cases of thyroid carcinoma treated in the US, 1985–1995 [see comments] (translated from English). Cancer. 1998;83(12):2638–48.
Liu S, Semenciw R, Ugnat AM, et al. Increasing thyroid cancer incidence in Canada, 1970–1996: time trends and age-period-cohort effects. Br J Cancer. 2001;85(9):1335–9.
Asmis LM, Gerber H, Kaempf J, et al. Epidermal growth factor stimulates cell proliferation and inhibits iodide uptake of FRTL-5 cells in vitro. J Endocrinol. 1995;145(3):513–20.
Farid NR. P53 mutations in thyroid carcinoma: tidings from an old foe. J Endocrinol Investig. 2001;24(7):536–45.
Prante O, et al. Regulation of uptake of 18F-FDG by a follicular human thyroid cancer cell line with mutation-activated K-ras. J Nucl Med. 2009;50(8):1364–70.
Christine J, et al. BRAFV600E mutation is associated with an increased risk of nodal recurrence requiring reoperative surgery in patients with papillary thyroid cancer. Surgery. 2010;148(6):1139–46.
McIver B, Grebe SK, Eberhardt NL. The PAX8/PPAR gamma fusion oncogene as a potential therapeutic target in follicular thyroid carcinoma. Curr Drug Targets Immune Endocr Metab Disord. 2004;4(3):221–34.
Anna G, et al. Prevalence of RET/PTC rearrangement in benign and malignant thyroid nodules and its clinical application. Endocr J. 2011;58(1):31–8.
Wybieralska E, et al. Role of ephrins in regulation of tumor cell migration. Postepy Biochem. 2009;55:153–62.
Kullander K, Klein R. Mechanisms and functions of Eph and ephrin signalling. Nat Rev Mol Cell Biol. 2002;3:475–86.
Kumar SR, Singh J, Xia G, et al. Receptor tyrosine kinase EphB4 is a survival factor in breast cancer. Am J Pathol. 2006;169:279–93.
Xia G, Kumar SR, Masood R, et al. EphB4 expression and biological significance in prostate cancer. Cancer Res. 2005;65:4623–32.
Nakada M, Drake KL, Nakada S, et al. Ephrin-B3 ligand promotes glioma invasion through activation of Rac1. Cancer Res. 2006;66:8492–500.
Vogt T, Stolz W, Welsh J, et al. Overexpression of Lerk-5/Eplg5 messenger RNA: a novel marker for increased tumorigenicity and metastatic potential in human malignant melanomas. Clin Cancer Res. 1998;4:791–7.
Brantley-Sieders DM, Zhuang G, Hicks D, et al. The receptor tyrosine kinase EphA2 promotes mammary adenocarcinoma tumorigenesis and metastatic progression in mice by amplifying ErbB2 signaling. J Clin Invest. 2008;118:64–78.
Noren NK, Pasquale EB. Paradoxes of the EphB4 receptor in cancer. Cancer Res. 2007;67:3994–7.
Batlle E, Bacani J, Begthel H, et al. EphB receptor activity suppresses colorectal cancer progression. Nature. 2005;435:1126–30.
Cortina C, Palomo-Ponce S, Iglesias M, et al. EphB-ephrin-B interactions suppress colorectal cancer progression by compartmentalizing tumor cells. Nat Genet. 2007;39:1376–83.
Noren NK, Lu M, Freeman AL, et al. Interplay between EphB4 on tumor cells and vascular ephrin-B2 regulates tumor growth. Proc Natl Acad Sci USA. 2004;101:5583–8.
Yang NY, Pasquale EB, Owen LB, et al. The EphB4 receptor-tyrosine kinase promotes the migration of melanoma cells through rho-mediated actin cytoskeleton reorganization. J Biol Chem. 2006;281:32574–86.
Noren NK, Foos G, Hauser CA, et al. The EphB4 receptor suppresses breast cancer cell tumorigenicity through an Abl-Crk pathway. Nat Cell Biol. 2006;8:815–25.
Liu LY, Han YC, Wu SH, et al. Expression of connective tissue growth factor in tumor tissues is an independent predictor of poor prognosis in patients with gastric cancer (translated from English). World J Gastroenterol. 2008;14(13):2110–4 (in English).
Clevers H, Batlle E. EphB/EphrinB receptors and Wnt signaling in colorectal cancer. Cancer Res. 2006;66:2–5.
Andres AC, Ziemiecki A. Eph and ephrin signaling in mammary gland morphogenesis and cancer. J Mammary Gland Biol Neoplasia. 2003;8:475–85.
Kumar SR, Masood R, Spannuth WA. The receptor tyrosine kinase EphB4 is overexpressed in ovarian cancer, provides survival signals and predicts poor outcome. Br J Cancer. 2007;96:1083–91.
Yang N-Y, Pasquale EB, Owen LB, Ethell IM. The EphB4 receptor-tyrosine kinase promotes the migration of melanoma cells through rho-mediated actin cytoskeleton reorganization. J Biol Chem. 2006;43:32574–86.
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xuqing, W., Lei, C., Zhengfa, M. et al. EphB4 is overexpressed in papillary thyroid carcinoma and promotes the migration of papillary thyroid cancer cells. Tumor Biol. 33, 1419–1427 (2012). https://doi.org/10.1007/s13277-012-0392-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-012-0392-5