Abstract
Background
Postoperative cognitive dysfunction (POCD) is a central nervous system (CNS) complication and occurs quite frequently after anesthesia, especially in elderly patients. DNA (cytosine-5)-methyltransferase 3-like (DNMT3L) is an enzymatically inactive regulatory factor that can modulate gene expression via methylation. Few studies specifically focused on the role of DNMT3L in sevoflurane-induced POCD.
Objective
The cognitive dysfunction was determined by Morris water maze assay. Hippocampal neuron apoptosis was measured by TdT-mediated dUTP nick-end labeling (TUNEL) assay. Neuron proliferation and apoptosis were measured by cell counting kit-8 (CCK-8) and flow cytometry assay. The expression levels of DNMT3L and postsynaptic density protein-95 (PSD95) were measured by qRT-PCR and western blot.
Results
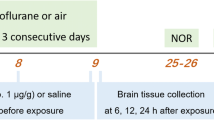
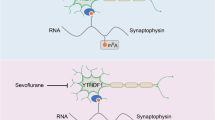
The sevoflurane-induced POCD aging rat model was established for in vivo study. Suppression of DNA methylation by 5-Aza-2′-deoxycytidine (5-AZA; a widely used methylation inhibitor) reversed sevoflurane-induced cognitive dysfunction and hippocampal neuron apoptosis in rats. Moreover, sevoflurane enhanced DNMT3L expression and induced methylation of PSD95 promoter in vivo. In vitro, sevoflurane induced neuronal apoptosis and DNMT3L expression to promote PSD95 methylation. Finally, rescue experiments indicated that sevoflurane induced neuron apoptosis by increasing DNMT3L expression and promoting methylation of PDS95 in POCD.
Conclusion
Sevoflurane induced neuronal apoptosis by increasing DNMT3L expression and promoting methylation of PDS95 in vivo and in vitro, providing a novel therapeutic strategy for POCD.





Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Aapola U et al (2000) Isolation and initial characterization of a novel zinc finger gene, DNMT3L, on 21q22.3, related to the cytosine-5-methyltransferase 3 gene family. Genomics 65:293–298. https://doi.org/10.1006/geno.2000.6168
Aapola U, Lyle R, Krohn K, Antonarakis SE, Peterson P (2001) Isolation and initial characterization of the mouse Dnmt3l gene. Cytogenet Cell Genet 92:122–126. https://doi.org/10.1159/000056881
Arora SS, Gooch JL, Garcia PS (2014) Postoperative cognitive dysfunction, Alzheimer’s disease, and anesthesia. Int J Neurosci 124:236–242. https://doi.org/10.3109/00207454.2013.833919
Bai D et al (2017) Antioxidative and anti-apoptotic roles of silibinin in reversing learning and memory deficits in APP/PS1 Mice. Neurochem Res 42:3439–3445. https://doi.org/10.1007/s11064-017-2389-3
Berger M et al (2015) Postoperative cognitive dysfunction: minding the gaps in our knowledge of a common postoperative complication in the elderly. Anesthesiol Clin 33:517–550. https://doi.org/10.1016/j.anclin.2015.05.008
Bi C et al (2018) Sevoflurane induces neurotoxicity in the developing rat hippocampus by upregulatingconnexin 43 via the JNK/c-Jun/AP-1 pathway. Biomed Pharmacother = Biomedecine & Pharmacotherapie 108:1469–1476. https://doi.org/10.1016/j.biopha.2018.09.111
Chen G, Gong M, Yan M, Zhang X (2013) Sevoflurane induces endoplasmic reticulum stress mediated apoptosis in hippocampal neurons of aging rats. PLoS ONE 8:e57870. https://doi.org/10.1371/journal.pone.0057870
Coley AA, Gao WJ (2018) PSD95: a synaptic protein implicated in schizophrenia or autism? Prog Neuropsychopharmacol Biol Psychiatry 82:187–194. https://doi.org/10.1016/j.pnpbp.2017.11.016
Cui D, Xu X (2018) DNA methyltransferases, DNA methylation, and age-associated cognitive function. Int J Mol Sci. https://doi.org/10.3390/ijms19051315
de Bartolomeis A, Latte G, Tomasetti C, Iasevoli F (2014) Glutamatergic postsynaptic density protein dysfunctions in synaptic plasticity and dendritic spines morphology: relevance to schizophrenia and other behavioral disorders pathophysiology, and implications for novel therapeutic approaches. Mol Neurobiol 49:484–511. https://doi.org/10.1007/s12035-013-8534-3
Esper T, Wehner M, Meinecke CD, Rueffert H (2015) Blood/gas partition coefficients for isoflurane, sevoflurane, and desflurane in a clinically relevant patient population. Anesth Analg 120:45–50. https://doi.org/10.1213/ANE.0000000000000516
Goeldner C, Reiss D, Kieffer BL, Ouagazzal AM (2010) Endogenous nociceptin/orphanin-FQ in the dorsal hippocampus facilitates despair-related behavior. Hippocampus 20:911–916. https://doi.org/10.1002/hipo.20760
Guo S et al (2018) Repeated exposure to sevoflurane impairs the learning and memory of older male rats. Life Sci 192:75–83. https://doi.org/10.1016/j.lfs.2017.11.025
Hackett JA, Surani MA (2013) DNA methylation dynamics during the mammalian life cycle. Philos Trans R Soc Lond B BiolSci 368:20110328. https://doi.org/10.1098/rstb.2011.0328
Jevtovic-Todorovic V et al (2013) Anaesthetic neurotoxicity and neuroplasticity: an expert group report and statement based on the BJA Salzburg seminar. Br J Anaesth 111:143–151. https://doi.org/10.1093/bja/aet177
Jia D, Jurkowska RZ, Zhang X, Jeltsch A, Cheng X (2007) Structure of Dnmt3a bound to Dnmt3L suggests a model for de novo DNA methylation. Nature 449:248–251. https://doi.org/10.1038/nature06146
Kim E, Sheng M (2004) PDZ domain proteins of synapses. Nat Rev Neurosci 5:771–781. https://doi.org/10.1038/nrn1517
Ling YZ, Ma W, Yu L, Zhang Y, Liang QS (2015) Decreased PSD95 expression in medial prefrontal cortex (mPFC) was associated with cognitive impairment induced by sevoflurane anesthesia. J Zhejiang Univ Sci B 16:763–771. https://doi.org/10.1631/jzus.B1500006
Liu Q et al (2019) MiR-190a potentially ameliorates postoperative cognitive dysfunction by regulating Tiam1. BMC Genom 20:670. https://doi.org/10.1186/s12864-019-6035-0
Lu J et al (2016) Global hypermethylation in fetal cortex of down syndrome due to DNMT3L overexpression. Hum Mol Genet 25:1714–1727. https://doi.org/10.1093/hmg/ddw043
Monk TG, Price CC (2011) Postoperative cognitive disorders. Curr Opin Crit Care 17:376–381. https://doi.org/10.1097/MCC.0b013e328348bece
Patron E, MesserottiBenvenuti S, Zanatta P, Polesel E, Palomba D (2013) Preexisting depressive symptoms are associated with long-term cognitive decline in patients after cardiac surgery. General Hosp Psychiatry 35:472–479. https://doi.org/10.1016/j.genhosppsych.2013.05.004
Shoair OA et al (2015) Incidence and risk factors for postoperative cognitive dysfunction in older adults undergoing major noncardiac surgery: a prospective study. J Anaesthesiol Clin Pharmacol 31:30–36. https://doi.org/10.4103/0970-9185.150530
Tian Y, Guo S, Guo Y, Jian L (2015a) Anestheticpropofol attenuates apoptosis, abeta accumulation, and inflammation induced by sevoflurane through NF-kappaB pathway in human neuroglioma cells. Cell Mol Neurobiol 35:891–898. https://doi.org/10.1007/s10571-015-0184-8
Tian Y, Guo S, Wu X, Ma L, Zhao X (2015b) Minocycline alleviates sevoflurane-induced cognitive impairment in aged rats. Cell Mol Neurobiol 35:585–594. https://doi.org/10.1007/s10571-014-0154-6
Vacas S, Degos V, Feng X, Maze M (2013) The neuroinflammatory response of postoperative cognitive decline. Br Med Bull 106:161–178. https://doi.org/10.1093/bmb/ldt006
Veland N et al (2019) DNMT3L facilitates DNA methylation partly by maintaining DNMT3A stability in mouse embryonic stem cells. Nucleic Acids Res 47:152–167. https://doi.org/10.1093/nar/gky947
Wang Z et al (2019) Treatment of secondary brain injury by perturbing postsynaptic density protein-95-NMDA receptor interaction after intracerebral hemorrhage in rats. J Cereb Blood Flow Metab 39:1588–1601. https://doi.org/10.1177/0271678X18762637
Wu BW, Wu MS, Guo JD (2018) Effects of microRNA-10a on synapse remodeling in hippocampal neurons and neuronal cell proliferation and apoptosis through the BDNF-TrkB signaling pathway in a rat model of Alzheimer’s disease. J Cell Physiol 233:5281–5292. https://doi.org/10.1002/jcp.26328
Wu Y et al (2019) Histone deacetylase inhibitor MS-275 alleviates postoperative cognitive dysfunction in rats by inhibiting hippocampal neuroinflammation. Neuroscience 417:70–80. https://doi.org/10.1016/j.neuroscience.2019.08.020
Xie H, She GM, Wang C, Zhang LY, Liu CF (2015) The gender difference in effect of sevoflurane exposure on cognitive function and hippocampus neuronal apoptosis in rats. Eur Rev Med Pharmacol Sci 19:647–657
Zhang Y et al (2018) The effect of dexmedetomidine on cognitive function and protein expression of Abeta, p-Tau, and PSD95 after extracorporeal circulation operation in aged rats. Biomed Res Int 2018:4014021. https://doi.org/10.1155/2018/4014021
Zhu Y et al (2017) Enhanced neuroinflammation mediated by DNA methylation of the glucocorticoid receptor triggers cognitive dysfunction after sevoflurane anesthesia in adult rats subjected to maternal separation during the neonatal period. J Neuroinflamm 14:6. https://doi.org/10.1186/s12974-016-0782-5
Funding
This work was supported by Natural Science Research Project of Anhui Educational Committee (Grant No. KJ2019A0330).
Author information
Authors and Affiliations
Contributions
YL and XL designed the study, supervised data collection, LY analyzed the data, interpreted the data, YS, DY and ZL prepare the manuscript for publication and reviewed the draft of the manuscript. All authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Yunzhi Ling states that there are no conflicts of interest to disclose. Xiaohong Li states that there are no conflicts of interest to disclose. Li Yu states that there are no conflicts of interest to disclose. Yiyun Sun states that there are no conflicts of interest to disclose. Dongdong Yang states that there are no conflicts of interest to disclose. Zhiyi Li states that there are no conflicts of interest to disclose.
Ethical approval
All animal experiments were approved by the Experimental Animal Management and Ethics Committee of Bengbu Medical College for the use of animals and conducted in accordance with the National Institutes of Health Laboratory Animal Care and Use Guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ling, Y., Li, X., Yu, L. et al. Sevoflurane induces neuronal apoptosis via enhancing DNMT3L expression and promoting methylation of PSD95 promoter in postoperative cognitive dysfunction. Mol. Cell. Toxicol. 17, 287–295 (2021). https://doi.org/10.1007/s13273-021-00135-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-021-00135-8