Abstract
Background
Deltamethrin, a type II pyrethroid insecticide, has been widely used due to its high efficacy against insects and low toxicity to mammals. However, recently, potential adverse health effects during deltamethrin exposure have been reported.
Methods
A proteomic analysis using two-dimensional electrophoresis with mass spectrometry was performed, and the proteomic data were validated by western blotting. Antioxidant enzyme activity was also measured by spectrophotometric analysis.
Results
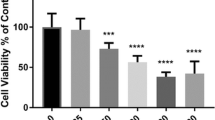
The 13 differentially expressed proteins, of which 8 were up-regulated and 5 down-regulated, are involved in a variety of cellular functions including protein folding, cytoskeleton regulation, and splicing process. The activities of antioxidant enzymes and acetylcholinesterase were also altered by deltamethrin exposure.
Conclusion
Differential protein expression caused by deltamethrin exposure in neuroblastoma cell lines could allow us to develop biomarkers for deltamethrin toxicity, and pursue future studies to assess the molecular mechanisms of deltamethrin toxicity.




Similar content being viewed by others
References
Baggerman G, Vierstraete E, De Loof A, Schoofs L (2005) Gel-based versus gel-free proteomics: a review. Comb Chem High Throughput Screen 8:669–677
Bai B et al (2019) Peroxiredoxin2 downregulation enhances hepatocellular carcinoma proliferation and migration, and is associated with unfavorable prognosis in patients. Oncol Rep 41:1539–1548
Caballero JP et al (2019) Nanoencapsulated deltamethrin as synergistic agent potentiates insecticide effect of indoxacarb through an unusual neuronal calcium-dependent mechanism. Pestic Biochem Physiol 157:1–12
Cabello G et al (2001) A rat mammary tumor model induced by the organophosphorous pesticides parathion and malathion, possibly through acetylcholinesterase inhibition. Environ Health Perspect 109:471–479
Cao W et al (2018) Determination of deltamethrin and its toxicity biomarkers in rabbit urine by high performance liquid chromatography-tandem mass spectrometry. Se Pu 36:523–530
Chauvin S, Sobel A (2015) Neuronal stathmins: a family of phosphoproteins cooperating for neuronal development, plasticity and regeneration. Prog Neurobiol 126:1–18
Chen M et al (2019) Molecular evidence of sequential evolution of DDT-and pyrethroid-resistant sodium channel in aedes aegypti. PLoS Negl Trop Dis 13:e0007432
Chrustek A et al (2018) Current research on the safety of pyrethroids used as insecticides. Medicina (Kaunas) 54:61
Cook JA, Mitchell JB (1989) Viability measurements in mammalian cell systems. Anal Biochem 179:1–7
Danh HC, Benedetti MS, Dostert P (1983) Differential changes in superoxide dismutase activity in brain and liver of old rats and mice. J Neurochem 40:1003–1007
Ding R et al (2017) The implication of p66shc in oxidative stress induced by deltamethrin. Chem Biol Interact 278:162–169
Dinis-Oliveira RJ et al (2006) Acute paraquat poisoning: report of a survival case following intake of a potential lethal dose. Pediatr Emerg Care 22:537–540
Doi H et al (2006) Motor neuron disorder simulating ALS induced by chronic inhalation of pyrethroid insecticides. Neurology 67:1894–1895
El Golli-Bennour E et al (2019) Protective effects of kegir against deltamethrin-induced hepatotoxicity in rats. Environ Sci Pollut Res Int 26:18856–18865
Ellman GL, Courtney KD, Andres V Jr, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Ensibi C et al (2013) Effects of deltamethrin on biometric parameters and liver biomarkers in common carp (Cyprinus carpio L.). Environ Toxicol Pharmacol 36:384–391
Flohé L, Günzler WA (1984) Assays of glutathione peroxidase. Methods Enzymol 105:114–120
Fulton MH, Key PB (2001) Acetylcholinesterase inhibition in estuarine fish and invertebrates as an indicator of organophosphorus insecticide exposure and effects. Environ Toxicol Chem 20:37–45
Gautrey H et al (2015) SRSF3 and hnRNP H1 regulate a splicing hotspot of HER2 in breast cancer cells. RNA Biol 12:1139–1151
Glorieux C, Calderon PB (2017) Catalase, a remarkable enzyme: targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Biol Chem 398:1095–1108
Hadwan MH (2018) Simple spectrophotometric assay for measuring catalase activity in biological tissues. BMC Biochem 19:1–8
Hileman EA, Achanta G, Huang P (2001) Superoxide dismutase: an emerging target for cancer therapeutics. Expert Opin Ther Targets 5:697–710
Ingkaninan K, Temkitthawon P, Chuenchom K, Yuyaem T, Thongnoi W (2003) Screening for acetylcholinesterase inhibitory activity in plants used in thai traditional rejuvenating and neurotonic remedies. J Ethnopharmacol 89:261–264
Jia ZZ, Zhang JW, Zhou D, Xu DQ, Feng XZ (2019) Deltamethrin exposure induces oxidative stress and affects meiotic maturation in mouse oocyte. Chemosphere 223:704–713
Kumar A, Sharma R, Rana D, Sharma N (2019a) Protective effect of alpha-tocopherol in deltamethrin induced immunotoxicity. Endocr Metab Immune Disord Drug Targets 19:171–184
Kumar A, Gupta M, Sharma R, Sharma N (2019b) Deltamethrin induced immunotoxicity and its protection by quercetin: an experimental study. Endocr Metab Immune Disord Drug Targets 19:1
Lu W et al (2014) Peroxiredoxin 2 is upregulated in colorectal cancer and contributes to colorectal cancer cells’ survival by protecting cells from oxidative stress. Mol Cell Biochem 387:261–270
Lu Q et al (2018) Deltamethrin toxicity: a review of oxidative stress and metabolism. Environ Res 170:260–281
Mannervik B (1999) Measurement of glutathione reductase activity. Curr Protoc Toxicol 7:1–4
Min CW et al (2017) Gel-based and gel-free proteome data associated with controlled deterioration treatment of glycine max seeds. Data Br 15:449–453
Nakamura M et al (2006) Phosphoproteomic profiling of human SH-SY5Y neuroblastoma cells during response to 6-hydroxydopamine-induced oxidative stress. Biochim Biophys Acta 1763:977–989
Oda SS, El-Maddawy ZK (2012) Protective effect of vitamin E and selenium combination on deltamethrin-induced reproductive toxicity in male rats. Exp Toxicol Pathol 64:813–819
Pitzer EM et al (2019) Deltamethrin exposure daily from postnatal day 3–20 in sprague-dawley rats causes long-term cognitive and behavioral deficits. Toxicol Sci 169:511–523
Ríos JC et al (2003) Tribromophenol induces the differentiation of SH-SY5Y human neuroblastoma cells in vitro. Toxicol In Vitro 17:635–641
Rubin CI, Atweh GF (2004) The role of stathmin in the regulation of the cell cycle. J Cell Biochem 93:242–250
Ryu AR, Kim YW, Lee MY (2019) Chlorin e6 and halogen light as a sebostatic photomedicine modulates linoleic acid-induced lipogenesis. Mol Cell Toxicol 15:49–56
Shim DH, Lim JW, Kim H (2015) Differentially expressed proteins in nitric oxide-stimulated NIH/3T3 fibroblasts: implications for inhibiting cancer development. Yonsei Med J 56:563–571
Shishkin SS, Kovalev LI, Pashintseva NV, Kovaleva MA, Lisitskaya K (2019) Heterogeneous nuclear ribonucleoproteins involved in the functioning of telomeres in malignant cells. Int J Mol Sci 20:745
Shrivastava R, Köster D, Kalme S, Mayor S, Neerathilingam M (2015) Tailor-made ezrin actin binding domain to probe its interaction with actin in-vitro. PLoS One 10:1–12
Skandrani D et al (2003) Effect of selected insecticides on growth rate and stress protein expression in cultured human A549 and SH-SY5Y cells. Toxicol In Vitro 20:1378–1386
Souza MF et al (2018) Deltamethrin intranasal administration induces memory, emotional and tyrosine hydroxylase immunoreactivity alterations in rats. Brain Res Bull 142:297–303
Suenaga S et al (2013) Human pancreatic cancer cells with acquired gemcitabine resistance exhibit significant up-regulation of peroxiredoxin-2 compared to sensitive parental cells. Anticancer Res 33:4821–4826
Traverso N et al (2013) Role of glutathione in cancer progression and chemoresistance. Oxidative Med Cell Longev 2013:1–10
Acknowledgements
This study was supported by Soonchunhyang University.
Author information
Authors and Affiliations
Contributions
MJK performed the experiments and wrote the initial draft of manuscript. MYL designed the research and wrote the final version of manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Min-Joo Kang and Mi-Young Lee declare that they have no conflict of interest.
Human and animal rights
The article does not contain any studies with humans or animals and this study was performed following institutional and national guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kang, MJ., Lee, MY. Toxicoproteomic analysis of deltamethrin exposure in neuroblastoma cell lines. Mol. Cell. Toxicol. 16, 93–101 (2020). https://doi.org/10.1007/s13273-019-00064-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13273-019-00064-7