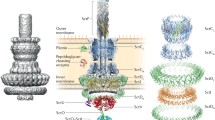
Abstract
Background
Type Three Secretion Systems (T3SS) are nanomachine complexes, which display the ability to inject effector proteins directly into host cells. This skill allows for gram-negative bacteria to modulate several host cell responses, such as cytoskeleton rearrangement, signal transduction, and cytokine production, which in turn increase the pathogenicity of these bacteria. The Salmonella enterica subsp. enterica serovar Typhimurium (ST) T3SS has been the most characterized so far. Among gram-negative bacterium, ST is one of enterica groups predicted to have two T3SSs activated during different phases of infection.
Objective
To comprise current information about ST T3SS structure and function as well as an overview of its assembly and hierarchical regulation.
Methods
With a brief and straightforward reading, this review summarized aspects of both ST T3SS, such as its structure and function. That was possible due to the development of novel techniques, such as X-ray crystallography, cryoelectron microscopy, and nano-gold labelling, which also elucidated the mechanisms behind T3SS assembly and regulation, which was addressed in this review.
Conclusion
This paper provided fundamental overview of ST T3SS assembly and regulation, besides summarized the structure and function of this complex. Due to T3SS relevance in ST pathogenicity, this complex could become a potential target in therapeutic studies as this nanomachine modulates the infection process.



Similar content being viewed by others
References
Abby SS, Rocha EPC (2012) The non-flagellar type III secretion system evolved from the bacterial flagellum and diversified into host-cell adapted systems. PLOS Genet 8:e1002983. https://doi.org/10.1371/journal.pgen.1002983
Abrusci P, Vergara-Irigaray M, Johnson S et al (2013) Architecture of the major component of the type III secretion system export apparatus. Nat Struct Mol Biol 20:99–104. https://doi.org/10.1038/nsmb.2452
Bakowski MA, Braun V, Brumell JH (2008) Salmonella-containing vacuoles: directing traffic and nesting to grow. Traffic 9:2022–2031. https://doi.org/10.1111/j.1600-0854.2008.00827.x
Bergeron JRC, Worrall LJ, Sgourakis NG et al (2013) a refined model of the prototypical Salmonella SPI-1 T3SS basal body reveals the molecular basis for its assembly. PLOS Pathog 9:e1003307. https://doi.org/10.1371/journal.ppat.1003307
Botteaux A, Sory MP, Biskri L et al (2009) MxiC is secreted by and controls the substrate specificity of the Shigella flexneri type III secretion apparatus. Mol Microbiol 71:449–460. https://doi.org/10.1111/j.1365-2958.2008.06537.x
Brawn LC, Hayward RD, Koronakis V (2007) Salmonella SPI1 effector SipA persists after entry and cooperates with a SPI2 effector to regulate phagosome maturation and intracellular replication. Cell Host Microbe 1:63–75. https://doi.org/10.1016/j.chom.2007.02.001
Burkinshaw BJ, Strynadka NCJ (2014) Assembly and structure of the T3SS. Biochim Biophys Acta BBA 1843:1649–1663. https://doi.org/10.1016/j.bbamcr.2014.01.035
Büttner D (2012) Protein export according to schedule: architecture, assembly, and regulation of type III secretion systems from plant- and animal-pathogenic bacteria. Microbiol Mol Biol Rev 76:262–310. https://doi.org/10.1128/MMBR.05017-11
Chatterjee S, Zhong D, Nordhues BA et al (2010) The crystal structures of the Salmonella type III secretion system tip protein SipD in complex with deoxycholate and chenodeoxycholate. Protein Sci 20:75–86. https://doi.org/10.1002/pro.537
Cheng LW, Anderson DM, Schneewind O (1997) Two independent type III secretion mechanisms for YopE in Yersinia enterocolitica. Mol Microbiol 24:757–765. https://doi.org/10.1046/j.1365-2958.1997.3831750.x
Cherradi Y, Schiavolin L, Moussa S et al (2013) Interplay between predicted inner-rod and gatekeeper in controlling substrate specificity of the type III secretion system. Mol Microbiol 87:1183–1199. https://doi.org/10.1111/mmi.12158
Cirillo DM, Valdivia RH, Monack DM, Falkow S (1998) Macrophage-dependent induction of the Salmonella pathogenicity island 2 type III secretion system and its role in intracellular survival. Mol Microbiol 30:175–188. https://doi.org/10.1046/j.1365-2958.1998.01048.x
Coburn B, Li Y, Owen D et al (2005) Salmonella enterica serovar Typhimurium pathogenicity island 2 is necessary for complete virulence in a mouse model of infectious enterocolitis. Infect Immun 73:3219–3227. https://doi.org/10.1128/IAI.73.6.3219-3227.2005
Collazo CM, Galán JE (1997) The invasion-associated type III system of Salmonella Typhimurium directs the translocation of Sip proteins into the host cell. Mol Microbiol 24:747–756. https://doi.org/10.1046/j.1365-2958.1997.3781740.x
Collazo CM, Zierler MK, Gatan JE (1995) Functional analysis of the Salmonella Typhimurium invasion genes invl and invJ and identification of a target of the protein secretion apparatus encoded in the inv locus. Mol Microbiol 15:25–38. https://doi.org/10.1111/j.1365-2958.1995.tb02218.x
Cornelis GR (2006) The type III secretion injectisome. Nat Rev Microbiol 4:811–825. https://doi.org/10.1038/nrmicro1526
Costa TRD, Felisberto-Rodrigues C, Meir A et al (2015) Secretion systems in Gram-negative bacteria: structural and mechanistic insights. Nat Rev Microbiol 13:343–359. https://doi.org/10.1038/nrmicro3456
Crago AM, Vassilis K (2002) Salmonella InvG forms a ring-like multimer that requires the InvH lipoprotein for outer membrane localization. Mol Microbiol 30:47–56. https://doi.org/10.1046/j.1365-2958.1998.01036.x
Deiwick J, Nikolaus T, Shea JE et al (1998) Mutations in Salmonella pathogenicity island 2 (SPI2) genes affecting transcription of SPI1 genes and resistance to antimicrobial agents. J Bacteriol 180:4775–4780
Deng W, Marshall NC, Rowland JL et al (2017) Assembly, structure, function and regulation of type III secretion systems. Nat Rev Microbiol 15:323–337. https://doi.org/10.1038/nrmicro.2017.20
Desvaux M, Hébraud M, Talon R, Henderson IR (2009) Secretion and subcellular localizations of bacterial proteins: a semantic awareness issue. Trends Microbiol 17:139–145. https://doi.org/10.1016/j.tim.2009.01.004
Dickenson NE, Choudhari SP, Adam PR et al (2013) Oligomeric states of the Shigella translocator protein IpaB provide structural insights into formation of the type III secretion translocon. Protein Sci 22:614–627. https://doi.org/10.1002/pro.2245
Diepold A, Wagner S (2014) Assembly of the bacterial type III secretion machinery. FEMS Microbiol Rev 38:802–822. https://doi.org/10.1111/1574-6976.12061
Diepold A, Wiesand U, Cornelis GR (2011) The assembly of the export apparatus (YscR, S, T, U, V) of the Yersinia type III secretion apparatus occurs independently of other structural components and involves the formation of an YscV oligomer. Mol Microbiol 82:502–514. https://doi.org/10.1111/j.1365-2958.2011.07830.x
dos Santos AMP, Ferrari RG, Conte-Junior CA (2018) Virulence factors in SalmonellaTyphimurium: the sagacity of a bacterium. Curr Microbiol. https://doi.org/10.1007/s00284-018-1510-4
Economou A (1999) Following the leader: bacterial protein export through the Sec pathway. Trends Microbiol 7:315–320. https://doi.org/10.1016/S0966-842X(99)01555-3
Edqvist PJ, Olsson J, Lavander M et al (2003) YscP and YscU regulate substrate specificity of the yersinia type iii secretion system. J Bacteriol 185:2259–2266. https://doi.org/10.1128/JB.185.7.2259-2266.2003
Eichelberg K, Ginocchio CC, Galán JE (1994) Molecular and functional characterization of the Salmonella Typhimurium invasion genes invB and invC: homology of InvC to the F0F1 ATPase family of proteins. J Bacteriol 176:4501–4510. https://doi.org/10.1128/jb.176.15.4501-4510.1994
Ellermeier JR, Slauch JM (2007) Adaptation to the host environment: regulation of the SPI1 type III secretion system in Salmonella enterica serovar Typhimurium. Curr Opin Microbiol 10:24–29. https://doi.org/10.1016/j.mib.2006.12.002
Erhardt M, Singer HM, Wee DH et al (2011) An infrequent molecular ruler controls flagellar hook length in Salmonella enterica. EMBO J 30:2948–2961. https://doi.org/10.1038/emboj.2011.185
Espina M, Olive AJ, Kenjale R et al (2006) IpaD localizes to the tip of the type III secretion system needle of Shigella flexneri. Infect Immun 74:4391–4400. https://doi.org/10.1128/IAI.00440-06
Fan F, Ohnishi K, Francis NR, Macnab RM (1997) The FliP and FliR proteins of Salmonella Typhimurium, putative components of the type III flagellar export apparatus, are located in the flagellar basal body. Mol Microbiol 26:1035–1046. https://doi.org/10.1046/j.1365-2958.1997.6412010.x
Feria JVM, Lefebre MD, Stierhof Y-D et al (2015) Role of autocleavage in the function of a type III secretion specificity switch protein in Salmonella enterica serovar. mBio. https://doi.org/10.1128/mBio.01459-15
Ferris HU, Minamino T (2006) Flipping the switch: bringing order to flagellar assembly. Trends Microbiol 14:519–526. https://doi.org/10.1016/j.tim.2006.10.006
Francis CL, Ryan TA, Jones BD et al (1993) Ruffles induced by Salmonella and other stimuli direct macropinocytosis of bacteria. Nature 364:639–642. https://doi.org/10.1038/364639a0
Fujii T, Cheung M, Blanco A et al (2012) Structure of a type III secretion needle at 7-Å resolution provides insights into its assembly and signaling mechanisms. Proc Natl Acad Sci 109:4461–4466. https://doi.org/10.1073/pnas.1116126109
Galán JE, Wolf-Watz H (2006) Protein delivery into eukaryotic cells by type III secretion machines. Nature 444:567–573. https://doi.org/10.1038/nature05272
Galán JE, Lara-Tejero M, Marlovits TC, Wagner S (2014) Bacterial type III secretion systems: specialized nanomachines for protein delivery into target cells. Annu Rev Microbiol 68:415–438. https://doi.org/10.1146/annurev-micro-092412-155725
Galkin VE, Schmied WH, Schraidt O et al (2010) The structure of the Salmonella Typhimurium type III secretion system needle shows divergence from the flagellar system. J Mol Biol 396:1392–1397. https://doi.org/10.1016/j.jmb.2010.01.001
Ginocchio CC, Olmsted SB, Wells CL, Galán JE (1994) Contact with epithelial cells induces the formation of surface appendages on SalmonellaTyphimurium. Cell 76:717–724. https://doi.org/10.1016/0092-8674(94)90510-X
Groisman E, Ochman H (1993) Cognate gene clusters govern invasion of host epithelial cells by Salmonella Typhimurium and Shigella flexneri. EMBO J 12:3779–3787. https://doi.org/10.1002/j.1460-2075.1993.tb06056.x
Guilvout I, Hardie KR, Sauvonnet N, Pugsley AP (1999) Genetic dissection of the outer membrane secretin PulD: are there distinct domains for multimerization and secretion specificity? J Bacteriol 181:7212–7220
Gunn JS (2000) Mechanisms of bacterial resistance and response to bile. Microbes Infect 2:907–913. https://doi.org/10.1016/S1286-4579(00)00392-0
Hansen-Wester I, Hensel M (2001) Salmonella pathogenicity islands encoding type III secretion systems. Microbes Infect 3:549–559. https://doi.org/10.1016/S1286-4579(01)01411-3
Haraga A, Ohlson MB, Miller SI (2008) Salmonellae interplay with host cells. Nat Rev Microbiol 6:53–66. https://doi.org/10.1038/nrmicro1788
Hensel M (2000) Salmonella pathogenicity island 2. Mol Microbiol 36:1015–1023. https://doi.org/10.1046/j.1365-2958.2000.01935.x
Hensel M (2004) Evolution of pathogenicity islands of Salmonella enterica. Int J Med Microbiol 294:95–102. https://doi.org/10.1016/j.ijmm.2004.06.025
Hensel M, Shea JE, Waterman SR et al (1998) Genes encoding putative effector proteins of the type III secretion system of Salmonella pathogenicity island 2 are required for bacterial virulence and proliferation in macrophages. Mol Microbiol 30:163–174. https://doi.org/10.1046/j.1365-2958.1998.01047.x
Herrero-Fresno A, Olsen JE (2017) Salmonella Typhimurium metabolism affects virulence in the host : a mini-review. Food Microbiol. https://doi.org/10.1016/j.fm.2017.04.016
Hueck CJ (1998) Type III protein secretion systems in bacterial pathogens of animals and plants. Microbiol Mol Biol Rev 62:379–433
Izoré T, Job V, Dessen A (2011) Biogenesis, regulation, and targeting of the type iii secretion system. Structure 19:603–612. https://doi.org/10.1016/j.str.2011.03.015
Jennings E, Thurston TLM, Holden DW (2017) Salmonella SPI-2 type III secretion system effectors: molecular mechanisms and physiological consequences. Cell Host Microbe 22:217–231. https://doi.org/10.1016/j.chom.2017.07.009
Journet L (2003) The needle length of bacterial injectisomes is determined by a molecular ruler. Science 302:1757–1760. https://doi.org/10.1126/science.1091422
Kimbrough TG, Miller SI (2000) Contribution of SalmonellaTyphimurium type III secretion components to needle complex formation. Proc Natl Acad Sci 97:11008–11013. https://doi.org/10.1073/pnas.200209497
Kimbrough TG, Miller SI (2002) Assembly of the type III secretion needle complex of Salmonella Typhimurium. Microbes Infect 4:75–82. https://doi.org/10.1016/S1286-4579(01)01512-X
Klein JR, Fahlen TF, Jones BD (2000) Transcriptional organization and function of invasion genes within Salmonella enterica serovar typhimurium pathogenicity island 1, including the prgH, prgI, prgJ, prgK, orgA, orgB, and orgC Genes. Infect Immun 68:3368–3376. https://doi.org/10.1128/IAI.68.6.3368-3376.2000
Kubori T, Galán JE (2002) Salmonella type III secretion-associated protein InvE controls translocation of effector proteins into host cells. J Bacteriol 184:4699–4708. https://doi.org/10.1128/JB.184.17.4699-4708.2002
Kubori T, Matsushima Y, Nakamura D et al (1998) Supramolecular structure of the SalmonellaTyphimurium type III protein secretion system. Science 280:602–605. https://doi.org/10.1126/science.280.5363.602
Kubori T, Sukhan A, Aizawa S-I, Galán JE (2000) Molecular characterization and assembly of the needle complex of the SalmonellaTyphimurium type III protein secretion system. Proc Natl Acad Sci 97:10225–10230. https://doi.org/10.1073/pnas.170128997
Kuhlen L, Abrusci P, Johnson S et al (2018) Structure of the core of the type three secretion system export apparatus. bioRxiv. https://doi.org/10.1101/249128
Lara-Tejero M, Kato J, Wagner S et al (2011) A sorting platform determines the order of protein secretion in bacterial type III systems. Science 331:1188–1191. https://doi.org/10.1126/science.1201476
Lee CA (1997) Type III secretion systems: machines to deliver bacterial proteins into eukaryotic cells? Trends Microbiol 5:148–156. https://doi.org/10.1016/S0966-842X(97)01029-9
Lefebre MD, Galán JE (2014) The inner rod protein controls substrate switching and needle length in a Salmonella type III secretion system. Proc Natl Acad Sci 111:817–822. https://doi.org/10.1073/pnas.1319698111
Loquet A, Sgourakis NG, Gupta R et al (2012) Atomic model of the type III secretion system needle. Nature 486:276–279. https://doi.org/10.1038/nature11079
Lunelli M, Hurwitz R, Lambers J, Kolbe M (2011) Crystal structure of PrgI-SipD: insight into a secretion competent state of the type three secretion system needle tip and its interaction with host ligands. PLOS Pathog 7:e1002163. https://doi.org/10.1371/journal.ppat.1002163
Makino F, Shen D, Kajimura N et al (2016) The Architecture of the cytoplasmic region of type III secretion systems. Sci Rep 6:33341. https://doi.org/10.1038/srep33341
Marcus SL, Brumell JH, Pfeifer CG, Finlay BB (2000) Salmonella pathogenicity islands: big virulence in small packages. Microbes Infect 2:145–156. https://doi.org/10.1016/S1286-4579(00)00273-2
Marlovits TC, Kubori T, Sukhan A et al (2004) Structural insights into the assembly of the type iii secretion needle complex. Science 306:1040–1042. https://doi.org/10.1126/science.1102610
Marlovits TC, Kubori T, Lara-Tejero M et al (2006) Assembly of the inner rod determines needle length in the type III secretion injectisome. Nature 441:637–640. https://doi.org/10.1038/nature04822
Maserati A, Fink RC, Lourenco A et al (2017) General response of Salmonella enterica serovar typhimurium to desiccation: a new role for the virulence factors sopD and sseD in survival. PLOS One 12:e0187692. https://doi.org/10.1371/journal.pone.0187692
Matteï P-J, Faudry E, Job V et al (2011) Membrane targeting and pore formation by the type III secretion system translocon. FEBS J 278:414–426. https://doi.org/10.1111/j.1742-4658.2010.07974.x
Miao EA, Rajan JV (2011) Salmonella and Caspase-1: a complex interplay of detection and evasion. Front Microbiol. https://doi.org/10.3389/fmicb.2011.00085
Miao EA, Mao DP, Yudkovsky N et al (2010) Innate immune detection of the type III secretion apparatus through the NLRC4 inflammasome. Proc Natl Acad Sci 107:3076–3080. https://doi.org/10.1073/pnas.0913087107
Minamino T, Macnab RM (1999) Components of the Salmonella flagellar export apparatus and classification of export substrates. J Bacteriol 181:1388–1394
Minamino T, Imada K, Namba K (2008) Mechanisms of type III protein export for bacterial flagellar assembly. Mol Biosyst 4:1105–1115. https://doi.org/10.1039/B808065H
Morita-Ishihara T, Ogawa M, Sagara H et al (2006) Shigella Spa33 is an essential c-ring component of type III secretion machinery. J Biol Chem 281:599–607. https://doi.org/10.1074/jbc.M509644200
Mueller CA, Broz P, Müller SA et al (2005) The V-antigen of yersinia forms a distinct structure at the tip of injectisome needles. Science 310:674–676. https://doi.org/10.1126/science.1118476
Nikolaus T, Deiwick J, Rappl C et al (2001) SseBCD proteins are secreted by the type III secretion system of salmonella pathogenicity island 2 and function as a translocon. J Bacteriol 183:6036–6045. https://doi.org/10.1128/JB.183.20.6036-6045.2001
Parsot C, Hamiaux C, Page A-L (2003) The various and varying roles of specific chaperones in type III secretion systems. Curr Opin Microbiol 6:7–14. https://doi.org/10.1016/S1369-5274(02)00002-4
Portaliou AG, Tsolis KC, Loos MS et al (2016) Type III secretion: building and operating a remarkable nanomachine. Trends Biochem Sci 41:175–189. https://doi.org/10.1016/j.tibs.2015.09.005
Prouty AM, Gunn JS (2000) Salmonella enterica serovar typhimurium invasion is repressed in the presence of bile. Infect Immun 68:6763–6769. https://doi.org/10.1128/IAI.68.12.6763-6769.2000
Rathinavelan T, Tang C, Guzman RND (2011) Characterization of the interaction between the Salmonella type III secretion system tip protein SipD and the needle protein PrgI by paramagnetic relaxation enhancement. J Biol Chem 286:4922–4930. https://doi.org/10.1074/jbc.M110.159434
Rathinavelan T, Lara-Tejero M, Lefebre M et al (2014) NMR model of PrgI–sipd interaction and its implications in the needle-tip assembly of the Salmonella type III secretion system. J Mol Biol 426:2958–2969. https://doi.org/10.1016/j.jmb.2014.06.009
Russo BC, Stamm LM, Raaben M et al (2016) Intermediate filaments enable pathogen docking to trigger type 3 effector translocation. Nat Microbiol 1:16025. https://doi.org/10.1038/nmicrobiol.2016.25
Schraidt O, Marlovits TC (2011) Three-dimensional model of Salmonella’s needle complex at subnanometer resolution. Science 331:1192–1195. https://doi.org/10.1126/science.1199358
Schraidt O, Lefebre MD, Brunner MJ et al (2010) Topology and organization of the SalmonellaTyphimurium type III secretion needle complex components. PLOS Pathog 6:e1000824. https://doi.org/10.1371/journal.ppat.1000824
Snider NT, Ku NO, Omary MB (2018) Intermediate filaments: the sweet side of vimentin. eLife 7:e35336. https://doi.org/10.7554/eLife.35336
Sorg I, Wagner S, Amstutz M et al (2007) YscU recognizes translocators as export substrates of the Yersinia injectisome. EMBO J 26:3015–3024. https://doi.org/10.1038/sj.emboj.7601731
Spreter T, Yip CK, Sanowar S et al (2009) A conserved structural motif mediates formation of the periplasmic rings in the type III secretion system. Nat Struct Mol Biol 16:468–476. https://doi.org/10.1038/nsmb.1603
Stebbins CE, Galán JE (2001) Maintenance of an unfolded polypeptide by a cognate chaperone in bacterial type III secretion. Nature 414:77–81. https://doi.org/10.1038/35102073
Stecher B, Robbiani R, Walker AW et al (2007) Salmonella enterica serovar typhimurium exploits inflammation to compete with the intestinal microbiota. PLOS Biol 5:e244. https://doi.org/10.1371/journal.pbio.0050244
Sukhan A, Kubori T, Wilson J, Galán JE (2001) Genetic analysis of assembly of the Salmonella enterica serovar typhimurium type III secretion-associated needle complex. J Bacteriol 183:1159–1167. https://doi.org/10.1128/JB.183.4.1159-1167.2001
Sukhan A, Kubori T, Galán JE (2003) Synthesis and Localization of the Salmonella SPI-1 type III secretion needle complex proteins PrgI and PrgJ. J Bacteriol 185:3480–3483. https://doi.org/10.1128/JB.185.11.3480-3483.2003
Thanassi DG, Hultgren SJ (2000) Multiple pathways allow protein secretion across the bacterial outer membrane. Curr Opin Cell Biol 12:420–430. https://doi.org/10.1016/S0955-0674(00)00111-3
Tucker SC, Galán JE (2000) Complex function for SicA, a Salmonella enterica serovar typhimurium type III secretion-associated chaperone. J Bacteriol 182:2262–2268. https://doi.org/10.1128/JB.182.8.2262-2268.2000
van der Heijden J, Finlay BB (2012) Type III effector-mediated processes in Salmonella infection. Future Microbiol 7:685–703. https://doi.org/10.2217/fmb.12.49
Vincent MS, Canestrari MJ, Leone P et al (2017) Characterization of the Porphyromonas gingivalis type IX secretion trans-envelope PorKLMNP core complex. J Biol Chem 292:3252–3261. https://doi.org/10.1074/jbc.M116.765081
Wagner S, Königsmaier L, Lara-Tejero M et al (2010) Organization and coordinated assembly of the type III secretion export apparatus. Proc Natl Acad Sci 107:17745–17750. https://doi.org/10.1073/pnas.1008053107
Wang Y, Ouellette AN, Egan CW et al (2007) Differences in the electrostatic surfaces of the type III secretion needle proteins PrgI, BsaL, and MxiH. J Mol Biol 371:1304–1314. https://doi.org/10.1016/j.jmb.2007.06.034
Wang Y, Nordhues BA, Zhong D, De Guzman RN (2010) NMR Characterization of the interaction of the Salmonella type III secretion system protein SipD and bile salts. Biochemistry 49:4220–4226. https://doi.org/10.1021/bi100335u
Waterman SR, Holden DW (2003) Functions and effectors of the Salmonella pathogenicity island 2 type III secretion system. Cell Microbiol 5:501–511. https://doi.org/10.1046/j.1462-5822.2003.00294.x
Wattiau P, Bernier B, Deslée P et al (1994) Individual chaperones required for Yop secretion by Yersinia. Proc Natl Acad Sci 91:10493–10497. https://doi.org/10.1073/pnas.91.22.10493
Wee DH, Hughes KT (2015) Molecular ruler determines needle length for the Salmonella Spi-1 injectisome. Proc Natl Acad Sci 112:4098–4103. https://doi.org/10.1073/pnas.1423492112
Worrall LJ, Hong C, Vuckovic M et al (2016) Near-atomic-resolution cryo-EM analysis of the Salmonella T3S injectisome basal body. Nature 540:597–601. https://doi.org/10.1038/nature20576
Zarivach R, Deng W, Vuckovic M et al (2008) Structural analysis of the essential self-cleaving type III secretion proteins EscU and SpaS. Nature 453:124–127. https://doi.org/10.1038/nature06832
Zhong D, Lefebre M, Kaur K et al (2012) The Salmonella type III secretion system inner rod protein PrgJ is partially folded. J Biol Chem 287:25303–25311. https://doi.org/10.1074/jbc.M112.381574
Zilkenat S, Franz-Wachtel M, Stierhof Y-D et al (2016) Determination of the stoichiometry of the complete bacterial type III secretion needle complex using a combined quantitative proteomic approach. Mol Cell Proteom. https://doi.org/10.1074/mcp.M115.056598
Acknowledgements
We thank to Virgínia P. Silveira for the design of the figures.
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (process no. 202.225/2017 and E-26/201.577/2018, FAPERJ, Brazil); Conselho Nacional de Desenvolvimento Científico e Tecnológico (process no. 311422/2016-0, CNPq, Brazil), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (process no. 125, CAPES/Embrapa 2014, CAPES, Brazil).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
dos Santos, A.M.P., Ferrari, R.G. & Conte-Junior, C.A. Type three secretion system in Salmonella Typhimurium: the key to infection. Genes Genom 42, 495–506 (2020). https://doi.org/10.1007/s13258-020-00918-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-020-00918-8