Abstract
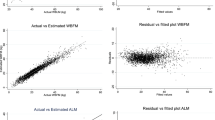
This initial study aimed at testing whether fat-containing agents can be used for the fat mass estimation methods using magnetic resonance imaging (MRI). As an example for clinical application, fat-containing agents (based on soybean oil, 10% and 20%), 100% soybean oil, and saline as reference substances were placed outside the proximal femurs obtained from 14 participants and analyzed by 0.3 T MRI. Fat content was the estimated fat fraction (FF) based on signal intensity (SIeFF, %). The SIeFF values of the femoral bone marrow, including the femoral head, neck, shaft, and trochanter area, were measured. MRI data were compared in terms of bone mineral content (BMC) and bone mineral density (BMD) by dual-energy X-ray absorptiometry (DXA) in the proximal femur. Twelve pig femurs were also used to confirm the correlation between FF by the DIXON method and SIeFF. According to Pearson’s correlation coefficient, the SIeFF and total BMC and BMD data revealed strong and moderate negative correlations in the femoral head (r < − 0.74) and other sites (r = − 0.66 to − 0.45). FF and SIeFF showed a strong correlation (r = 0.96). This study was an initial investigation of a method for estimating fat mass with fat-containing agents and showed the potential for use in MRI. SIeFF and FF showed a strong correlation, and SIeFF and BMD and BMC showed correlation; however, further studies are needed to use SIeFF as a substitute for DXA.







Similar content being viewed by others
Data availability
Not applicable.
References
Reeder SB, Cruite I, Hamilton G, Sirlin CB (2011) Quantitative assessment of liver fat with magnetic resonance imaging and spectroscopy. J Magn Reson Imaging 34:729–749. https://doi.org/10.1002/jmri.22775
Gaeta M, Messina S, Mileto A, Vita GL, Ascenti G, Vinci S, Bottari A, Vita G, Settineri N, Bruschetta D, Racchiusa S, Minutoli F (2012) Muscle fat-fraction and mapping in Duchenne muscular dystrophy: evaluation of disease distribution and correlation with clinical assessments. Preliminary experience. Skelet Radiol 41:955–961. https://doi.org/10.1007/s00256-011-1301-5
Gondim Teixeira PA, Cherubin T, Badr S, Bedri A, Gillet R, Albuisson E, Blum A (2019) Proximal femur fat fraction variation in healthy subjects using chemical shift-encoding based MRI. Sci Rep 9:20212. https://doi.org/10.1038/s41598-019-56611-8
Cordes C, Baum T, Dieckmeyer M, Ruschke S, Diefenbach MN, Hauner H, Kirschke JS, Karampinos DC (2016) MR-based assessment of bone marrow fat in osteoporosis, diabetes, and obesity. Front Endocrinol (Lausanne) 7:74. https://doi.org/10.3389/fendo.2016.00074
Paccou J, Penel G, Chauveau C, Cortet B, Hardouin P (2019) Marrow adiposity and bone: review of clinical implications. Bone 118:8–15. https://doi.org/10.1016/j.bone.2018.02.008
Zhang L, Li S, Hao S, Yuan Z (2016) Quantification of fat deposition in bone marrow in the lumbar vertebra by proton MRS and in-phase and out-of-phase MRI for the diagnosis of osteoporosis. J Xray Sci Technol 24:257–266. https://doi.org/10.3233/XST-160549
Cordes C, Baum T, Dieckmeyer M, Ruschke S, Diefenback MN, Hauner H, Kirschke JS, Karampinos DC (2016) MR-based assessment of bone marrow fat in osteoporosis, diabetes, and obesity. Front Endocrinol (Lausanne) 7:74. https://doi.org/10.3389/fendo.2016.00074
Dixon WT (1984) Simple proton spectroscopic imaging. Radiology 153:189–194. https://doi.org/10.1148/radiology.153.1.6089263
Zhang Y, Zhou Z, Wang C, Cheng X, Wang L, Duanmu Y, Zhang C, Veronese N, Guglielmi G (2018) Reliability of measuring the fat content of the lumbar vertebral marrow and paraspinal muscles using MRI mDIXON-Quant sequence. Diagn Interv Radiol 24:302–307. https://doi.org/10.5152/dir.2018.17323
Loau J, Shiehmorteza M, Girard OM, Sirlin CB, Bydder M (2013) Evaluation of MRI fat fraction in the liver and spine pre and post SPIO infusion. Magn Reson Imaging 31:1012–1016. https://doi.org/10.1016/j.mri.2013.01.016
Fishbein MH, Gardner KG, Potter CJ, Schmalbrock P, Smith MA (1997) Introduction of fast MR imaging in the assessment of hepatic steatosis. Magn Reson Imaging 15:287–293. https://doi.org/10.1016/s0730-725x(96)00224-x
Sane S, Baba M, Kusano C, Shirao K, Kamada T, Aikou T (1999) Fat emulsion administration in the early postoperative period in patients undergoing esophagectomy for carcinoma depresses arachidonic acid metabolism in neutrophils. Nutrition 15:341–346. https://doi.org/10.1016/s0899-9007(99)00032-5
Hoshino R, Kamiya Y, Fujii Y, Ttsubokawa T (2017) Lipid emulsion injection-induced reversal of cardiac toxicity and acceleration of emergence from general anesthesia after scalp infiltration of a local anesthetic: a case report. JA Clin Rep 3:9. https://doi.org/10.1186/s40981-017-0077-6
Peterson P, Svensson J, Månsson S (2014) Relaxation effects in MRI-based quantification of fat content and fatty acid composition. Magn Reason Med 72:1320–1329. https://doi.org/10.1002/mrm.25048
Mashhood A, Railkar R, Yokoo T, Levin Y, Clark L, Fox-Bosetti S, Middleton MS, Riek J, Kauh E, Dardzinski BJ, Williams D, Sirlin C, Shire NJ (2013) Reproducibility of hepatic fat fraction measurement by magnetic resonance imaging. J Magn Reason Imaging 37:1359–1370. https://doi.org/10.1002/jmri.23928
Paccou J, Hardouin P, Cotten A, Penel G, Cortet B (2015) The role of bone marrow fat in skeletal health: usefulness and perspectives for clinicians. J Clin Endocrinol Metab 100:3613–3621. https://doi.org/10.1210/jc.2015-2338
Baum T, Yap SP, Karampinos DC, Nardo L, Kuo D, Burghardt AJ, Masharani UB, Schwartz AV, Li X, Link TM (2012) Does vertebral bone marrow fat content correlate with abdominal adipose tissue, lumbar spine BMD and blood biomarkers in women with type 2 diabetes mellitus? J Magn Reson Imaging 35:117–124. https://doi.org/10.1002/jmri.22757
Cummings SR, Black D (1995) Bone mass measurements and risk of fracture in Caucasian women: a review of findings from prospective studies. Am J Med 98:24S-28S. https://doi.org/10.1016/s0002-9343(05)80041-5
Pietschmann P, Rauner M, Sipos W, Kerschan-Schindl K (2009) Osteoporosis: an age-related and gender-specific disease–a mini-review. Gerontology 55:3–12. https://doi.org/10.1159/000166209
Hofbauer LC, Brueck CC, Singh SK, Dobnig H (2007) Review osteoporosis in patients with Diabetes Mellitus. J Bone Miner Res 22:1317–1328. https://doi.org/10.1359/jbmr.070510
Zhao LJ, Jiang H, Papasian CJ, Maulik D, Dress B, Hamilton J, Deng HW (2008) Review correlation of obesity and osteoporosis: effect of fat mass on the determination of osteoporosis. J Bone Miner Res 23:17–29. https://doi.org/10.1359/jbmr.070813
WHO study group (1994) Assessment of fracture risk and its application to screening for postmenopausal osteoporosis. WHO technical report series 843, Geneva, Switzland.
Bredella MA, Daley SM, Kalra MK, Keenan BJ, Miller KK, Torriani M (2015) Marrow adipose tissue quantification of the lumbar spine by using dual-energy CT and single-voxel (1)H MR spectroscopy: a feasibility study. Radiology 277:230–235. https://doi.org/10.1148/radiol.2015142876
Japan Osteoporosis Society, Bone Strength Evaluation Committee (2007) Proximal femur BMD measurement manual (in Japanese). Osteoporos Jpn 15:1–41
Belaroussi B, Milles J, Carme S, Zhu YM, Benoit-Cattin H (2006) Intensity non-uniformity correction in MRI: existing methods and their validation. Med Image Anal 10:234–246. https://doi.org/10.1016/j.media.2005.09.004
Sieron D, Drakopoulos D, Loebelenz LI, Schroeder C, Ebner L, Obmann VC, Huber AT, Christe A (2020) Correlation between fat signal ratio on T1-weighted MRI in the lower vertebral bodies and age, comparing 1.5-T and 3-T scanners. Acta Radiol Open 9:2058460120901517. https://doi.org/10.1177/2058460120901517
Meurens F, Summerfield A, Nauwynck H, Saif L, Gerdts V (2012) The pig: a model for human infectious diseases. Trends Microbiol 20:50–57. https://doi.org/10.1016/j.tim.2011.11.002
Reichert JC, Saifzadeh S, Wullschleger ME, Epari DR, Schütz MA, Duda GN, Schell H, van Griensven M, Redl H, Hutmacher DW (2009) The challenge of establishing preclinical models for segmental bone defect research. Biomaterials 30:2149–2163. https://doi.org/10.1016/j.biomaterials.2008.12.050
Aerssens J, Boonen S, Lowet G, Dequeker J (1998) Interspecies differences in bone composition, density, and quality: potential implications for in vivo bone research. Endocrinology 139:663–670. https://doi.org/10.1210/endo.139.2.5751
Thorwarth M, Schultze-Mosgau S, Kessler P, Wiltfang J, Schlegel KA (2005) Bone regeneration in osseous defects using a resorbable nanoparticular hydroxyapatite. J Oral Maxillofac Surg 63:1626–1633. https://doi.org/10.1016/j.joms.2005.06.010
Kanda Y (2013) Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/10.1038/bmt.2012.244
Binkovitz LA, Henwood MJ (2007) Pediatric DXA: technique and interpretation. Pediatr Radiol 37:21–31. https://doi.org/10.1007/s00247-006-0153-y
Carter DR, Bouxsein ML, Marcus R (1992) New approaches for interpreting projected bone densitometry data. J Bone Miner Res 7:137–145. https://doi.org/10.1002/jbmr.5650070204
Prentice A, Parsons TJ, Cole TJ (1994) Uncritical use of bone mineral density in absorptiometry may lead to size- related artifacts in the identification of bone mineral determinants. Am J Clin Nutr 60:837–842. https://doi.org/10.1093/ajcn/60.6.837
Kröger H, Vainio P, Nieminen J, Kotaniemi A (1995) Comparison of different models for interpreting bone mineral density measurements using DXA and MRI technology. Bone 17:157–159. https://doi.org/10.1016/s8756-3282(95)00162-x
Arokoski MH, Arokoski JPA, Vainio P, Niemitukia LH, Kröger H, Jurvelin JS (2002) Comparison of DXA and MRI methods for interpreting femoral neck bone mineral density. J Clin Densitom 5:289–296. https://doi.org/10.1385/jcd:5:3:289
Acknowledgements
The authors would like to thank Mr. Koji Uchida in Center for Information and Neural Networks National Institute of Information and Communications Technology, Dr. Shunichi Motegi in Gunma Paz University, Dr. Rei Yoshida in Kurihara Central Hospital, Mis. Yuriko Nohara in Gazou no mori Diagnostic Clinic, Kenichiro Yamamura in Tokushima Bunri University, and Kunihiro Yabe in Yamagata Prefectural Shinjo Hospital for his valuable advice and technical support on measurements.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that he/she has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takatsu, Y., Ohnishi, H., Tateyama, T. et al. Usefulness of fat-containing agents: an initial study on estimating fat content for magnetic resonance imaging. Phys Eng Sci Med 47, 339–350 (2024). https://doi.org/10.1007/s13246-023-01372-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13246-023-01372-y