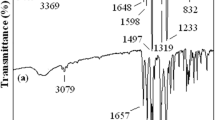
Abstract
A novel unsymmetrical diamine, 1,3-bis(4-aminophenoxy)naphthalene, was prepared through the nucleophilic displacement of 1,3-dihydroxynaphthalene with p-fluoronitrobenzene in the presence of potassium carbonate, followed by catalytic reduction with hydrazine. A series of novel aromatic polyimides containing bis(phenoxy)naphthalene units were synthesized from the unsymmetrical diamine and a range of aromatic tetracarboxylic dianhydrides via the usual two-step procedures that included ring-opening polyaddition and thermal/chemical cyclodehydration. The poly(amic acid)s had inherent viscosities ranging from 0.65 to 2.01 dL/g. All the poly(amic acid)s could be converted thermally into transparent, flexible, and tough polyimide films. Most of the polyimides were soluble in organic solvents, such as dimethylacetamide, N-methyl-2-pyrrolidone, m-cresol, or o-chlorophenol due to the unsymmetrical and bulky pendant naphthalene groups. The tensile strength and elongation at break of the polyimide films ranged from 74 to 97 MPa and 4 to 7%, respectively. These polyimides had glass transition temperatures ranging from 221 to 312 °C. Thermogravimetric analysis revealed these polymers to be fairly stable up to 500 °C, and the 10% weight loss temperature were recorded in the range of 531–580 °C in nitrogen and 522–570 °C in air. The substituted position effect of bis(phenoxy)naphthalene on the properties of the polyimides were investigated. The polyimides derived from unsymmetrical 1,3-substituted bis(4-aminophenoxy) naphthalene showed better solubility without sacrificing their thermal and mechanical properties due to the unsymmetrical and bulky pendant effects.
Similar content being viewed by others
References
C. H. Jung and Y. M. Lee, Macromol. Res., 16, 555 (2008).
S.-H. Park, K.-J. Kim, W.-W. So, S.-J. Moon, and S.-B. Lee, Macromol. Res., 11, 157 (2003).
H.-S. Lee, A. Roy, A. S. Badami, and J. E. McGrath, Macromol. Res., 15, 160 (2007).
H. Liu, M.-H. Lee, and J. Lee, Macromol. Res., 17, 725 (2009).
H.-J. Kim, M. H. Litt, S. Y. Nam, and E.-M. Shin, Macromol. Res., 11, 458 (2003).
H.-J. Kim, M. H. Litt, E.-M. Shin, and Y. N. Sang, Macromol. Res., 12, 545 (2004).
J.-Y. Lee, J.-H. Kim, and B. K. Rhee, Macromol. Res., 15, 234 (2007).
G.-Y. Lee, H.-N. Jang, W.-T. Jung, and J.-Y. Lee, Macromol. Res., 16, 741 (2008).
S. G. Hahm, K. S. Jin, S. Park, M. Ree, H.-S. Kim, S.-K. Kwon, and Y.-H. Kim, Macromol. Res., 17, 976 (2009).
M. Ree, Macromol. Res., 14, 1 (2006).
W. Dong and S.-H. Paek, Macromol. Res., 12, 251 (2004).
Y. M. Jang, J. Y. Seo, K. H. Chae, and M. H. Yi, Macromol. Res., 14, 300 (2006).
X.-D. Li, Z.-X. Zhong, G. Jin, S. H. Lee, and M.-H. Lee, Macromol. Res., 14, 257 (2006).
A. S. Mathews, I. Kim, and C.-S. Ha, Macromol. Res., 15, 114 (2007).
M. M. Ghosh and K. L. Mittal, Eds., Polyimides: Fundamentals and Applications, Marcel Dekker, New York, 1996.
N. Ueyama and A. Harada, Eds., Macromolecular Nanostructured Materials, Kodansha & Springer, Berlin, 2004, Chapter 2.2, pp 80–100.
A. Naoki, M. Kenji, and W. Masahiro, Chem. Mater., 16, 2841 (2005).
X. Wang, Y. Li, C. Gong, S. Zhang, and T. Ma, J. Appl. Polym. Sci., 104, 212 (2007).
C. E. Sroog, Prog. Polym. Sci., 16, 561 (1991).
S. Ando, T. Sawada, and Y. Inoue, Electron. Lett., 29, 2143 (1993).
S. Y. Tsay, B. K. Chen, and C. P. Chen, J. Appl. Polym. Sci., 99, 2966 (2006).
K. L. Wang, T. Y. Tseng, H. L. Tsai, and S. C. Wu, J. Polym. Sci. Part A: Polym. Chem., 46, 6861 (2008).
D. J. Liaw, K. L. Wang, E. T. Kang, S. P. Pujari, M. H. Chen, Y. C. Huang, B. C. Tao, K. R. Lee, and J. Y. Lai, J. Polym. Sci. Part A: Polym. Chem., 47, 991 (2009).
D. J. Liaw, K. L. Wang, S. P. Pujari, Y. C. Huang, B. C. Tao, M. H. Chen, K. R. Lee, and J. Y. Lai, Dyes Pigments, 82, 109 (2009).
K. L. Wang, W. T. Liou, D. J. Liaw, and S. T. Huang, Polymer, 49, 1538 (2008).
K. L. Wang, W. T. Liou, D. J. Liaw, and W. T. Chen, Dyes Pigments, 78, 93 (2008).
D. J. Liaw, K. L. Wang, and F. C. Chang, Macromolecules, 40, 3568 (2007).
D. J. Liaw, K. L. Wang, F. C. Chang, K. R. Lee, and J. Y. Lai, J. Polym. Sci. Part A: Polym. Chem., 45, 2367 (2007).
C. P. Yang and J. A. Chen, Polym. J., 30, 571 (1998).
C. P. Yang and W. T. Chen, J. Polym. Sci. Part A: Polym. Chem., 31, 2799 (1993).
C. P. Yang and W. T. Chen, Macromolecules, 26, 4865 (1993).
C. P. Yang and W. T. Chen, Makromol. Chem., 194, 3061 (1993).
C. P. Yang, S. H. Hsiao, and C. C. Jiang, J. Polym. Sci. Part A: Polym. Chem., 33, 1487 (1995).
C. P. Yang and J. H. Lin, J. Polym. Sci. Part A: Polym. Chem., 33, 2183 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, WT., Wang, KL. Novel organosoluable aromatic polyimides derived from unsymmetrical 1,3-bis(4-aminophenoxy)naphthalene and aromatic dianhydrides. Macromol. Res. 18, 821–827 (2010). https://doi.org/10.1007/s13233-010-0902-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13233-010-0902-5