Abstract
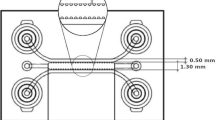
Vessel co-option (VC) differs from angiogenesis in that tumor cells grow toward blood vessels. Through VC, tumor cells can receive relatively more nutrients and oxygen from blood vessels. Despite its clinical significance, VC is relatively less studied compared to angiogenesis because of difficulties in longitudinal observation of VC in vivo and lack of proper VC models in vitro. A needle template method in which microchannels are formed in hydrogel by needles was used to form blood vessels and mimic angiogenesis. However, it has not yet been used to mimic VC. In this study, we report the development of VC on chip based on the needle template method. On the VC on chip, the effect of distance between spheroids and blood vessels on VC induction was investigated by seeding glioblastoma (GBM) spheroids 50 and 250 μm from the preformed blood vessels. Irrespective of distance, cancer cells from the spheroids grew toward the blood vessels but did not penetrate the vessels, indicating that GBM cells showed VC-like behavior. These results suggest that our chip could recapitulate VC in GBM.




Similar content being viewed by others
References
Seano, G., Jain, R.K.: Vessel co-option in glioblastoma: emerging insights and opportunities. Angiogenesis 23, 9–16 (2020)
Ribatti, D., Pezzella, F.: Vascular co-option and other alternative modalities of growth of tumor vasculature in glioblastoma. Front. Oncol. 12, 874554 (2022)
Kuczynski, E.A., Vermeulen, P.B., Pezzella, F., Kerbel, R.S., Reynolds, A.R.: Vessel co-option in cancer. Nat. Rev. Clin. Oncol. 16, 469–493 (2019)
Kuczynski, E.A., Reynolds, A.R.: Vessel co-option and resistance to anti-angiogenic therapy. Angiogenesis 23, 55–74 (2020)
Donnem, T., et al.: Vessel co-option in primary human tumors and metastases: an obstacle to effective anti-angiogenic treatment? Cancer Med. 2, 427–436 (2013)
Rada, M., Lazaris, A., Kapelanski-Lamoureux, A., Mayer, T.Z., Metrakos, P.: Tumor microenvironment conditions that favor vessel co-option in colorectal cancer liver metastases: a theoretical model. Semin. Cancer Biol. 71, 52–64 (2021)
Kim, M.-H., van Noort, D., Sung, J.H., Park, S.: Organ-on-a-chip for studying gut-brain interaction mediated by extracellular vesicles in the gut microenvironment. Int. J. Mol. Sci. 22, 13513 (2021)
Alves, A.H., et al.: The advances in glioblastoma on-a-chip for therapy approaches. Cancers 14, 869 (2022)
Kim, S., Kim, W., Lim, S., Jeon, J.S.: Vasculature-on-a-chip for in vitro disease models. Bioeng. (Basel, Switzerland) 4, 8 (2017)
Moses, S.R., Adorno, J.J., Palmer, A.F., Song, J.W.: Vessel-on-a-chip models for studying microvascular physiology, transport, and function in vitro. Am. J. Physiol. Physiol. 320, C92–C105 (2020)
Aazmi, A., et al.: Engineered vasculature for organ-on-a-chip systems. Engineering 9, 131–147 (2022)
Pollet, A.M.A.O., den Toonder, J.M.J.: Recapitulating the vasculature using organ-on-chip technology. Bioengineering 7, 17 (2020)
Lim, J., Ching, H., Yoon, J.-K., Jeon, N.L., Kim, Y.: Microvascularized tumor organoids-on-chips: advancing preclinical drug screening with pathophysiological relevance. Nano Converg. 8, 12 (2021)
Seo, S., et al.: An engineered neurovascular unit for modeling neuroinflammation. Biofabrication 13, 35039 (2021)
Kwak, T.J., Lee, E.: In vitro modeling of solid tumor interactions with perfused blood vessels. Sci. Rep. 10, 20142 (2020)
Seo, S., Nah, S.-Y., Lee, K., Choi, N., Kim, H.N.: Triculture model of in vitro bbb and its application to study BBB-associated chemosensitivity and drug delivery in glioblastoma. Adv. Funct. Mater. 32, 2106860 (2022)
Bernstein, J.J., Woodard, C.A.: Glioblastoma cells do not intravasate into blood vessels. Neurosurgery 36, 124–32 (1995). ((discussion 132))
Seo, Y.J., Cho, W.H., Kang, D.W., Cha, S.H.: Extraneural metastasis of glioblastoma multiforme presenting as an unusual neck mass. J. Korean Neurosurg. Soc. 51, 147–150 (2012)
Elena, A., et al.: Extraneural metastases in glioblastoma patients: two cases with YKL-40-positive glioblastomas and a meta-analysis of the literature. Neurosurg. Rev. 39, 37–46 (2016)
Kim, J., Kim, S., Uddin, S., Lee, S.S., Park, S.: Microfabricated stretching devices for studying the effects of tensile stress on cells and tissues. BioChip J. (2022). https://doi.org/10.1007/s13206-022-00073-0
Son, J., Kim, H.-H., Lee, J.-H., Jeong, W.-I., Park, J.-K.: Assembly and disassembly of the micropatterned collagen sheets containing cells for location-based cellular function analysis. BioChip J. 15, 77–89 (2021)
Han, S., et al.: 3D bioprinted vascularized tumour for drug testing. Int. J. Mol. Sci. 21, 2993 (2020)
Delannoy, E., et al.: Multi-layered human blood vessels-on-chip design using double viscous finger patterning. Biomedicines 10, 797 (2022)
Wang, Y.I., Shuler, M.L.: UniChip enables long-term recirculating unidirectional perfusion with gravity-driven flow for microphysiological systems. Lab Chip 18, 2563–2574 (2018)
Sung, J.H., Kam, C., Shuler, M.L.: A microfluidic device for a pharmacokinetic–pharmacodynamic (PK–PD) model on a chip. Lab Chip 10, 446–455 (2010)
Kwak, B.S., et al.: Microfluidic skin chip with vasculature for recapitulating the immune response of the skin tissue. Biotechnol. Bioeng. 117, 1853–1863 (2020)
Lee, D.W., Choi, N., Sung, J.H.: A microfluidic chip with gravity-induced unidirectional flow for perfusion cell culture. Biotechnol. Prog. 35, e2701 (2019)
Lee, Y., et al.: Gut-kidney axis on chip for studying effects of antibiotics on risk of hemolytic uremic syndrome by shiga toxin-producing Escherichia coli. Toxins 13, 775 (2021)
Norton, K.-A., Popel, A.S.: Effects of endothelial cell proliferation and migration rates in a computational model of sprouting angiogenesis. Sci. Rep. 6, 36992 (2016)
Ko, J., et al.: Tumor spheroid-on-a-chip: a standardized microfluidic culture platform for investigating tumor angiogenesis. Lab Chip 19, 2822–2833 (2019)
Bae, J., Han, S., Park, S.: Recent advances in 3D bioprinted tumor microenvironment. BioChip J. 14, 137–147 (2020)
Beck, B., et al.: A vascular niche and a VEGF–Nrp1 loop regulate the initiation and stemness of skin tumours. Nature 478, 399–403 (2011)
Kumar, S., et al.: Intra-tumoral metabolic zonation and resultant phenotypic diversification are dictated by blood vessel proximity. Cell Metab. 30, 201-211.e6 (2019)
Sarveswaran, K., et al.: Synthetic capillaries to control microscopic blood flow. Sci. Rep. 6, 21885 (2016)
Rouwkema, J., Koopman, B.F.J.M., Van Blitterswijk, C.A., Dhert, W.J.A., Malda, J.: Supply of nutrients to cells in engineered tissues. Biotechnol. Genet. Eng. Rev. 26, 163–178 (2009)
Leung, E., et al.: Blood vessel endothelium-directed tumor cell streaming in breast tumors requires the HGF/C-Met signaling pathway. Oncogene 36, 2680–2692 (2017)
Xiao, Y., et al.: Ex vivo dynamics of human glioblastoma cells in a microvasculature-on-a-chip system correlates with tumor heterogeneity and subtypes. Adv. Sci. 6, 1801531 (2019)
Cuddapah, V.A., Robel, S., Watkins, S., Sontheimer, H.: A neurocentric perspective on glioma invasion. Nat. Rev. Neurosci. 15, 455–465 (2014)
Frentzas, S., et al.: Vessel co-option mediates resistance to anti-angiogenic therapy in liver metastases. Nat. Med. 22, 1294–1302 (2016)
Leenders, W.P.J., et al.: Antiangiogenic therapy of cerebral melanoma metastases results in sustained tumor progression via vessel co-option. Clin. Cancer Res. 10, 6222–6230 (2004)
Kienast, Y., et al.: Real-time imaging reveals the single steps of brain metastasis formation. Nat. Med. 16, 116–122 (2010)
Valiente, M., et al.: Serpins promote cancer cell survival and vascular co-option in brain metastasis. Cell 156, 1002–1016 (2014)
Jeong, H.-S., et al.: Investigation of the lack of angiogenesis in the formation of lymph node metastases. J. Natl. Cancer Inst. 107, djv155 (2015)
Pereira, E.R., et al.: Lymph node metastases can invade local blood vessels, exit the node, and colonize distant organs in mice. Science 359, 1403–1407 (2018)
Acknowledgements
This work was supported by the Technology Innovation Program (Industrial Strategic Technology Development Program-Development of disease models based on 3D microenvironmental platforms mimicking multiple organs and evaluation of drug efficacy) (20008413) funded by the MOTIE (Ministry of Trade, Industry, and Energy) in Korea.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing financial interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bae, J., Kim, MH., Han, S. et al. Development of Tumor-Vasculature Interaction on Chip Mimicking Vessel Co-Option of Glioblastoma. BioChip J 17, 77–84 (2023). https://doi.org/10.1007/s13206-022-00090-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13206-022-00090-z