Abstract
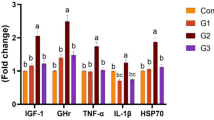
The experiment was designed to validate the effect of Artemisia annua and its novel commercial product (Navy Cox) on the control of necrotic enteritis (NE). A total of one hundred forty broiler chicks were randomly distributed into seven equal groups: G1, control negative; G2, infected with Eimeria (day 15) and C. perfringens (day 19); G3, treated with Navy Cox before challenge; G4, treated with Artemisia before challenge; G5, infected and then treated with Navy Cox; G6, infected and then treated with Artemisia; and G7, infected and treated with amoxicillin. Chicken response and immune organ indicants were recorded during the observation period (4 weeks). Whole blood and serum samples were collected for immunological evaluation, and tissue samples were collected for bacterial counts and estimation of mRNA expression of genes encoding apoptosis, tight junctions, and immunity. Chickens in the infected group revealed a significant decrease in RBCS, HB, PCV% total protein, Lysozyme, and nitric oxide activity in addition to leukocytosis, heterophilia, monocytosis, increase in cortisol, interleukins, and malondialdehyde. Treated groups displayed lower lesions, colony-forming units, and no mortality. Concurrently, a complete blood profile, antioxidants, and immune markers showed significant improvements. The mRNA expression levels of CASP, CLDN-1, OCLN, TJPI, MUC2, and cell-mediated immune response genes (p < 0.0001) were significantly alleviated in the treated groups compared with the challenged counterpart. This is the first-ever report on the efficacy valuation of Navy Cox compared to standard antibiotic treatment of clostridial NE. Navy Cox proved remarkable capability to minimize C. perfringens colonization in broiler intestines, modulation of mucus production, gut health integrity, immune organs, and immune response when used as a prophylactic agent in this form or naturally as Artemisia.







Similar content being viewed by others
Availability of data and material
All data used have been included in the manuscript.
Code availability
Not applicable.
References
Abadeen ZU, Javed MT, Rizvi F, Rahman SU (2021) Salutary effects of anti-Clostridium perfringens type A egg yolk antibodies (IgY) on growth performance and hemato-biochemical parameters in experimentally infected broiler chicken. Pak Vet J 41:562–566
Abd El Tawab AA, Amar AM, Elshorbagy MA, Mostafa EW (2015) Impact of probiotic and prebiotic on the lysozyme response of broilers against clostridial infection. Benha Vet Med J 28:208–213
Abd El-Hack ME, El-Saadony MT, Elbestawy AR et al (2021) Necrotic enteritis in broiler chickens: disease characteristics and prevention using organic antibiotic alternatives–a comprehensive review. Poult Sci 101:101590
Abd El-Hamid MI, El-Sayed ME, Ali AR et al (2019) Marjoram extract down-regulates the expression of Pasteurella multocida adhesion, colonization and toxin genes: A potential mechanism for its antimicrobial activity. Comp Immunol Microbiol Infect Dis 62:101–108
Abdul-Careem MF, Hunter DB, Thanthrige-Don N et al (2008) Cellular and cytokine responses associated with dinitrofluorobenzene-induced contact hypersensitivity in the chicken. Vet Immunol Immunopathol 122:275–284
Aebi H (1984) Catalase in vitro. Methods in enzymology. Elsevier, pp 121–126
Avalos-Téllez R, Suárez-Güemes F, Carrillo-Casas EM et al (2010) Bacteria and yeast normal microbiota from respiratory tract and genital area of bottlenose dolphins (Tursiops truncatus). Curr Res Technol Educ Top Appl Microbiol Microb Biotechnol 2:666–673
Awad WA, Hess C, Hess M (2017) Enteric pathogens and their toxin-induced disruption of the intestinal barrier through alteration of tight junctions in chickens. Toxins (basel) 9:60
Balda MS, Matter K (2008) Tight junctions at a glance. J Cell Sci 121:3677–3682
Brandtzaeg P, Johansen F-E (2005) Mucosal B cells: phenotypic characteristics, transcriptional regulation, and homing properties. Immunol Rev 206:32–63
Bulfon C, Volpatti D, Galeotti M (2015) Current research on the use of plant-derived products in farmed fish. Aquac Res 46:513–551
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326:1–16
Couper KN, Blount DG, Riley EM (2008) IL-10: the master regulator of immunity to infection. J Immunol 180:5771–5777
Coursodon CF, Glock RD, Moore KL et al (2012) TpeL-producing strains of Clostridium perfringens type A are highly virulent for broiler chicks. Anaerobe 18:117–121
Crippen TL, Sheffield CL, He H et al (2003) Differential nitric oxide production by chicken immune cells. Dev Comp Immunol 27:603–610
Dahiya JP, Hoehler D, Wilkie DC et al (2005) Dietary glycine concentration affects intestinal Clostridium perfringens and lactobacilli populations in broiler chickens1. Poult Sci 84:1875–1885
Das S, Vörös-Horváth B, Bencsik T et al (2020) Antimicrobial activity of different Artemisia essential oil formulations. Molecules 25:2390
Dawwam GE, Al-Shemy MT, El-Demerdash AS (2022) Green synthesis of cellulose nanocrystal/ZnO bio-nanocomposites exerting antibacterial activity and downregulating virulence toxigenic genes of food-poisoning bacteria. Sci Rep 12:1–18
do Vale A, Cabanes D, Sousa S (2016) Bacterial toxins as pathogen weapons against phagocytes. Front Microbiol 7:42
Doumas BT, Bayse DD, Carter RJ et al (1981) A candidate reference method for determination of total protein in serum. I Dev Valid Clin Chem 27:1642–1650
Du E, Wang W, Gan L et al (2016) Effects of thymol and carvacrol supplementation on intestinal integrity and immune responses of broiler chickens challenged with Clostridium perfringens. J Anim Sci Biotechnol 7:1–10
El-Azzouny MM, El-Demerdash AS, Seadawy HG, Abou-Khadra SH (2018) Antimicrobial effect of garlic (Allium sativum) and thyme (Zataria multiflora Boiss) extracts on some food borne pathogens and their effect on virulence gene expression. Cell Mol Biol 64:79–86
Elbadawy M, Aboubakr M (2017) Efficacy of colimox® (a new combination of amoxicillin and colistin) in the control of experimentally induced necrotic enteritis in broiler chickens. Int J Pharmacol Toxicol 5:51–56
El-Demerdash AS, Aggour MG, El-Azzouny MM, Abou-Khadra SH (2018) Molecular analysis of integron gene cassette arrays associated multi-drug resistant Enterobacteriaceae isolates from poultry. Cell Mol Biol 64:149–156
Elkouby-Naor L, Ben-Yosef T (2010) Functions of claudin tight junction proteins and their complex interactions in various physiological systems. Int Rev Cell Mol Biol 279:1–32
Elphick DA, Mahida YR (2005) Paneth cells: their role in innate immunity and inflammatory disease. Gut 54:1802–1809
Engberg RM, Grevsen K, Ivarsen E et al (2012) The effect of Artemisia annua on broiler performance, on intestinal microbiota and on the course of a Clostridium perfringens infection applying a necrotic enteritis disease model. Avian Pathol 41:369–376
Fan X, Liu S, Liu G et al (2015) Vitamin A deficiency impairs mucin expression and suppresses the mucosal immune function of the respiratory tract in chicks. PLoS ONE 10:e0139131
Ferreira JFS, Luthria DL, Sasaki T, Heyerick A (2010) Flavonoids from Artemisia annua L. as antioxidants and their potential synergism with artemisinin against malaria and cancer. Molecules 15:3135–3170
Forder REA, Nattrass GS, Geier MS et al (2012) Quantitative analyses of genes associated with mucin synthesis of broiler chickens with induced necrotic enteritis. Poult Sci 91:1335–1341
Fotiadis D, Kanai Y, Palacin M (2013) The SLC3 and SLC7 families of amino acid transporters. Mol Aspects Med 34:139–158
Furuse M, Hata M, Furuse K et al (2002) Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1–deficient mice. J Cell Biol 156:1099–1111
Gharaibeh MH, Khalifeh MS, Nawasreh AN et al (2021) Assessment of immune response and efficacy of essential oils application on controlling necrotic enteritis induced by Clostridium perfringens in broiler chickens. Molecules 26:4527
Gharib-Naseri K, Dorigam JCP, Doranalli K et al (2021) Bacillus amyloliquefaciens CECT 5940 improves performance and gut function in broilers fed different levels of protein and/or under necrotic enteritis challenge. Anim Nutr 7:185–197
Guo S, Ma J, Xing Y et al (2020) Artemisia annua L. aqueous extract as an alternative to antibiotics improving growth performance and antioxidant function in broilers. Ital J Anim Sci 19:399–409
Hediger MA, Romero MF, Peng J-B et al (2004) The ABCs of solute carriers: physiological, pathological and therapeutic implications of human membrane transport proteins. Pflug Arch 447:465–468
Helal SS, Khalaf NM, el Menisy AA, Lebdah MA (2019) Clostridium perfringens type A causing necrotic enteritis outbreaks among chickens in Egypt. Zagazig Vet J 47:398–407
Horn NL, Donkin SS, Applegate TJ, Adeola O (2009) Intestinal mucin dynamics: response of broiler chicks and White Pekin ducklings to dietary threonine. Poult Sci 88:1906–1914
Hoseinifar SH, Sun Y-Z, Zhou Z et al (2020) Boosting immune function and disease bio-control through environment-friendly and sustainable approaches in finfish aquaculture: herbal therapy scenarios. Rev Fish Sci Aquacult 28:303–321
Hosseinzadeh L, Malekshahi A, Ahmadi F et al (2018) The protective effect of different extracts of three Artemisia species against H2O2-induced oxidative stress and apoptosis in PC12 neuronal cells. Pharmacogn Res 10:64
Ibrahim D, Abdelfattah-Hassan A, Badawi M et al (2021) Thymol nanoemulsion promoted broiler chicken’s growth, gastrointestinal barrier and bacterial community and conferred protection against Salmonella Typhimurium. Sci Rep 11:1–20
Innis CJ, Conley K, Gibbons P et al (2022) Veterinary observations and biological specimen use after a massive confiscation of palawan forest turtles (Siebenrockiella leytensis). Chelonian Conserv Biol 21:46–62
Kaldhusdal M, Hofshagen M, Løvland A et al (1999) Necrotic enteritis challenge models with broiler chickens raised on litter: evaluation of preconditions, Clostridium perfringens strains and outcome variables. FEMS Immunol Med Microbiol 24:337–343
Kaldhusdal M, Benestad SL, Løvland A (2016) Epidemiologic aspects of necrotic enteritis in broiler chickens–disease occurrence and production performance. Avian Pathol 45:271–274
Konashi S, Takahashi K, Akiba Y (2000) Effects of dietary essential amino acid deficiencies on immunological variables in broiler chickens. Br J Nutr 83:449–456
Krys K, Vignoles VL, de Almeida I, Uchida Y (2022) Outside the “cultural binary”: understanding why Latin American collectivist societies foster independent selves. Perspect Psychol Sci 17:1166–1187
Lammers A, Wieland WH, Kruijt L et al (2010) Successive immunoglobulin and cytokine expression in the small intestine of juvenile chicken. Dev Comp Immunol 34:1254–1262
Lan Y, Verstegen MWA, Tamminga S, Williams BA (2005) The role of the commensal gut microbial community in broiler chickens. Worlds Poult Sci J 61:95–104
Latorre JD, Adhikari B, Park SH et al (2018) Evaluation of the epithelial barrier function and ileal microbiome in an established necrotic enteritis challenge model in broiler chickens. Front Vet Sci 5:199
Lee K-W, Lillehoj HS (2022) Role of Clostridium perfringens necrotic enteritis B-like toxin in disease pathogenesis. Vaccines (basel) 10:61
Lee KW, Lillehoj HS, Jang SI et al (2011) Effects of coccidiosis control programs on antibody levels against selected pathogens and serum nitric oxide levels in broiler chickens. J Appl Poult Res 20:143–152
Liu J, Liu L, Li L et al (2018) Protective immunity induced by Eimeria common antigen 14–3–3 against Eimeria tenella, Eimeria acervulina and Eimeria maxima medical and health sciences. BMC Vet Res Immunology 14:1107
McIlwain DR, Berger T, Mak TW (2013) Caspase functions in cell death and disease. Cold Spring Harb Perspect Biol 5:a008656
Mitchell LA, Koval M (2010) Specificity of interaction between Clostridium perfringens enterotoxin and claudin-family tight junction proteins. Toxins (basel) 2:1595–1611
Nafa QF, Hussin SM, Hamadi WF (2021) Characterization of some active organic compound from cold and hot aqueous solvent and study their antibiotic of Artemisia herba-alba Asso plant oil. Egypt J Chem 64:6691–6709
Nasr El-Deen N, Gamal El-Deen I, Khodary M (2019) Effect of experimental Clostridium perfringens infection on some immunological, hematological and biochemical values in broiler chickens. Zagazig Vet J 47:222–233
Norbury CJ, Hickson ID (2001) Cellular responses to DNA damage. Annu Rev Pharmacol Toxicol 41:367–401
Nunes VA, Gozzo AJ, Cruz-Silva I et al (2005) Vitamin E prevents cell death induced by mild oxidative stress in chicken skeletal muscle cells. Compar Biochem Physiol Part C 141:225–240
Paulsen SM, Engstad RE, Robertsen B (2001) Enhanced lysozyme production in Atlantic salmon (Salmo salar L.) macrophages treated with yeast β-glucan and bacterial lipopolysaccharide. Fish Shellfish Immunol 11:23–37
Pope JL, Ahmad R, Bhat AA et al (2014) Claudin-1 overexpression in intestinal epithelial cells enhances susceptibility to adenamatous polyposis coli-mediated colon tumorigenesis. Mol Cancer 13:1–13
Riedl SJ, Shi Y (2004) Molecular mechanisms of caspase regulation during apoptosis. Nat Rev Mol Cell Biol 5:897–907
Rothwell L, Young JR, Zoorob R et al (2004) Cloning and characterization of chicken IL-10 and its role in the immune response to Eimeria maxima. J Immunol 173:2675–2682
Saitoh Y, Suzuki H, Tani K et al (2015) Structural insight into tight junction disassembly by Clostridium perfringens enterotoxin. Science (1979) 347:775–778
Sakaguchi T, Gu X, Golden HM et al (2002) Cloning of the human claudin-2 5′-flanking region revealed a TATA-less promoter with conserved binding sites in mouse and human for caudal-related homeodomain proteins and hepatocyte nuclear factor-1α. J Biol Chem 277:21361–21370
Saleem G (2013) Necrotic enteritis, disease induction, predisposing factors and novel biochemical markers in broilers chickens. University of Glasgow
Savva CG, da Costa SPF, Bokori-Brown M et al (2013) Molecular architecture and functional analysis of NetB, a pore-forming toxin from Clostridium perfringens. J Biol Chem 288:3512–3522
Seo HW, Rengaraj D, Choi JW et al (2011) The expression profile of apoptosis-related genes in the chicken as a human epithelial ovarian cancer model. Oncol Rep 25:49–56
Sharopov FS, Salimov A, Numonov S et al (2020) Chemical composition, antioxidant, and antimicrobial activities of the essential oils from Artemisia annua L. growing wild in Tajikistan. Nat Prod Commun 15:1934578X20927814
Shojadoost B, Vince AR, Prescott JF (2012) The successful experimental induction of necrotic enteritis in chickens by Clostridium perfringens: a critical review. Vet Res 43:1–12
Soares MP, Cardoso IL, Ishikawa MM et al (2020) Effects of Artemisia annua alcohol extract on physiological and innate immunity of Nile tilapia (Oreochromis niloticus) to improve health status. Fish Shellfish Immunol 105:369–377
Spitz DR, Oberley LW (1989) An assay for superoxide dismutase activity in mammalian tissue homogenates. Anal Biochem 179:8–18
Strong RA, Hester PY, Eicher SD et al (2015) The effect of cooled perches on immunological parameters of caged white leghorn hens during the hot summer months. PLoS ONE 10:e0141215
Sun J, Madan R, Karp CL, Braciale TJ (2009) Effector T cells control lung inflammation during acute influenza virus infection by producing IL-10. Nat Med 15:277–284
Thrall MA, Weiser G, Allison R, Campbell T (2012) Veterinary hematology and clinical chemistry. John Wiley & Sons
To H, Suzuki T, Kawahara F et al (2017) Experimental induction of necrotic enteritis in chickens by a netB-positive Japanese isolate of Clostridium perfringens. J Vet Med Sci 79:350–358
Uldry M, Ibberson M, Hosokawa M, Thorens B (2002) GLUT2 is a high affinity glucosamine transporter. FEBS Lett 524:199–203
Wade B, Keyburn A (2015) The true cost of necrotic enteritis. World Poult 31:16–17
Wang X, Zhao D, Milby AC et al (2021) Evaluation of euthanasia methods on behavioral and physiological responses of newly hatched male layer chicks. Animals 11:1802
Wei X, Yang Z, Rey FE et al (2012) Fatty acid synthase modulates intestinal barrier function through palmitoylation of mucin 2. Cell Host Microbe 11:140–152
Wherry EJ, Teichgräber V, Becker TC et al (2003) Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat Immunol 4:225–234
Wiedosari E, Wardhana AH et al (2018) Anticoccidial activity of artemisinin and extract of Artemesia annua leaves in chicken infected by Eimeria tenella. JITV. https://doi.org/10.14334/jitv.v22i4.1622
Williams JM, Duckworth CA, Burkitt MD et al (2015) Epithelial cell shedding and barrier function: a matter of life and death at the small intestinal villus tip. Vet Pathol 52:445–455
Wu S-B, Rodgers N, Choct M (2011) Real-time PCR assay for Clostridium perfringens in broiler chickens in a challenge model of necrotic enteritis. Appl Environ Microbiol 77:1135–1139
Yuan JS, Reed A, Chen F, Stewart CN (2006) Statistical analysis of real-time PCR data. BMC Bioinform 7:85
Zanu HK, Keerqin C, Kheravii SK et al (2020) Influence of meat and bone meal, phytase, and antibiotics on broiler chickens challenged with subclinical necrotic enteritis: 2. intestinal permeability, organ weights, hematology, intestinal morphology, and jejunal gene expression. Poult Sci 99:2581–2594
Zhang PF, Shi BL, Su JL et al (2017a) Relieving effect of Artemisia argyi aqueous extract on immune stress in broilers. J Anim Physiol Anim Nutr (berl) 101:251–258
Zhang Q, Chen X, Eicher SD et al (2017b) Effect of threonine on secretory immune system using a chicken intestinal ex vivo model with lipopolysaccharide challenge. Poult Sci 96:3043–3051
Zhang P, Shi B, Li T et al (2018) Immunomodulatory effect of Artemisia argyi polysaccharide on peripheral blood leucocyte of broiler chickens. J Anim Physiol Anim Nutr (berl) 102:939–946
Zhao F, Zhang Z, Yao H et al (2013) Effects of cold stress on mRNA expression of immunoglobulin and cytokine in the small intestine of broilers. Res Vet Sci 95:146–155
Zhou H, Lepp D, Pei Y et al (2017) Influence of pCP1NetB ancillary genes on the virulence of Clostridium perfringens poultry necrotic enteritis strain CP1. Gut Pathog 9:1–7
Zhu Q, Sun P, Zhang B et al (2021) Progress on gut health maintenance and antibiotic alternatives in broiler chicken production. Front Nutr 8:903
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors manifested that they have no conflicts of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
El-Demerdash, A.S., Mohamady, S.N., Megahed, H.M. et al. Evaluation of gene expression related to immunity, apoptosis, and gut integrity that underlies Artemisia's therapeutic effects in necrotic enteritis-challenged broilers. 3 Biotech 13, 181 (2023). https://doi.org/10.1007/s13205-023-03560-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03560-9