Abstract
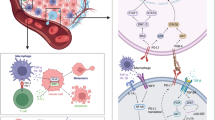
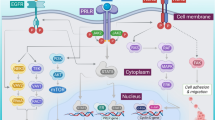
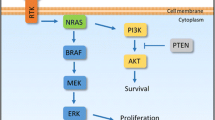
Based on the well-documented studies, numerous tumors episodically regress permanently without treatment. Knowing the host tissue-initiated causative factors would offer considerable translational applicability, as a permanent regression process may be therapeutically replicated on patients. For this, we developed a systems biological formulation of the regression process with experimental verification and identified the relevant candidate biomolecules for therapeutic utility. We devised a cellular kinetics-based quantitative model of tumor extinction in terms of the temporal behavior of three main tumor-lysis entities: DNA blockade factor, cytotoxic T-lymphocyte and interleukin-2. As a case study, we analyzed the time-wise biopsy and microarrays of spontaneously regressing melanoma and fibrosarcoma tumors in mammalian/human hosts. We analyzed the differentially expressed genes (DEGs), signaling pathways, and bioinformatics framework of regression. Additionally, prospective biomolecules that could cause complete tumor regression were investigated. The tumor regression process follows a first-order cellular dynamics with a small negative bias, as verified by experimental fibrosarcoma regression; the bias is necessary to eliminate the residual tumor. We identified 176 upregulated and 116 downregulated DEGs, and enrichment analysis showed that the most significant were downregulated cell-division genes: TOP2A–KIF20A–KIF23–CDK1–CCNB1. Moreover, Topoisomerase-IIA inhibition might actuate spontaneous regression, with collateral confirmation provided from survival and genomic analysis of melanoma patients. Candidate molecules such as Dexrazoxane/Mitoxantrone, with interleukin-2 and antitumor lymphocytes, may potentially replicate permanent tumor regression process of melanoma. To conclude, episodic permanent tumor regression is a unique biological reversal process of malignant progression, and signaling pathway understanding, with candidate biomolecules, may plausibly therapeutically replicate the regression process on tumors clinically.




















Similar content being viewed by others
Data availability
All the data are included in this manuscript. For further clarifications, please communicate with the corresponding author PKR at Dept. of Life Sciences, Shiv Nadar University, Dadri 201314, India.
References
Bayat Mokhtari R, Homayouni TS, Baluch N, Morgatskaya E, Kumar S, Das B, Yeger H (2017) Combination therapy in combating cancer. Oncotarget 8:38022–38043. https://doi.org/10.18632/oncotarget.16723
Biktimirov TZ, Butov AA, Savinov YuG (2005) Optimal control of the moment of spontaneous tumor regression. Autom Remote Control 66:658–663. https://doi.org/10.1007/s10513-005-0108-z
Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, Fridman W-H, Pagès F, Trajanoski Z, Galon J (2009) ClueGO: a Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 25:1091–1093. https://doi.org/10.1093/bioinformatics/btp101
Blessing K, McLaren KM (1992) Histological regression in primary cutaneous melanoma: recognition, prevalence and significance. Histopathology 20:315–322. https://doi.org/10.1111/j.1365-2559.1992.tb00988.x
Borghaei H, Smith MR, Campbell KS (2009) Immunotherapy of cancer. Eur J Pharmacol 625:41–54. https://doi.org/10.1016/j.ejphar.2009.09.067
Chen T, Sun Y, Ji P, Kopetz S, Zhang W (2015) Topoisomerase IIα in Chromosome Instability and Personalized Cancer Therapy. Oncogene 34:4019–4031. https://doi.org/10.1038/onc.2014.332
Cole WH (1981) Efforts to explain spontaneous regression of cancer. J Surg Oncol 17:201–209. https://doi.org/10.1002/jso.2930170302
Combination Treatments | SEER Training [WWW Document] (2022) https://training.seer.cancer.gov/treatment/combination/?msclkid=b91f81b3ced211ec93f5c37d7391014b. Accessed 8 May 2022
de Pillis LG, Gu W, Radunskaya AE (2006) Mixed immunotherapy and chemotherapy of tumors: modeling, applications and biological interpretations. J Theor Biol 238:841–862. https://doi.org/10.1016/j.jtbi.2005.06.037
Del Monte U (2009) Does the cell number 10(9) still really fit one gram of tumor tissue? Cell Cycle 8:505–506. https://doi.org/10.4161/cc.8.3.7608
Dennis G, Sherman BT, Hosack DA, Yang J, Gao W, Lane HC, Lempicki RA (2003) DAVID: database for annotation, visualization, and integrated discovery. Genome Biol 4:R60. https://doi.org/10.1186/gb-2003-4-9-r60
Dutta MD, Roy PK (2000) Cancer self-remission and tumour instability—a cybernetic analysis Towards a fresh paradigm for cancer treatment. Kybernetes 29:896–927. https://doi.org/10.1108/03684920010342035
Freeman LC, Borgatti SP, White DR (1991) Centrality in valued graphs: a measure of betweenness based on network flow. Social Networks 13:141–154. https://doi.org/10.1016/0378-8733(91)90017-N
Fryback DG, Stout NK, Rosenberg MA, Trentham-Dietz A, Kuruchittham V, Remington PL (2006) Chapter 7: the Wisconsin breast cancer epidemiology simulation model. JNCI Monographs 2006:37–47. https://doi.org/10.1093/jncimonographs/lgj007
GEPIA (Gene Expression Profiling Interactive Analysis) [WWW Document] (2022) http://gepia.cancer-pku.cn/index.html. Accessed 18 July 22
Hall JE, Guyton AC (2011) Guyton and Hall textbook of medical physiology, 12th edn. Saunders/Elsevier, Philadelphia
Hicks AM, Riedlinger G, Willingham MC, Alexander-Miller MA, Von Kap-Herr C, Pettenati MJ, Sanders AM, Weir HM, Du W, Kim J, Simpson AJG, Old LJ, Cui Z (2006) Transferable anticancer innate immunity in spontaneous regression/complete resistance mice. Proc Natl Acad Sci USA 103:7753–7758. https://doi.org/10.1073/pnas.0602382103
Hubenak JR, Zhang Q, Branch CD, Kronowitz SJ (2014) Mechanisms of injury to normal tissue after radiotherapy: a review. Plast Reconstr Surg 133:49e–56e. https://doi.org/10.1097/01.prs.0000440818.23647.0b
Jacob J (2017) Analyze your own microarray data in R/Bioconductor—BITS wiki [WWW Document], 2022 https://wiki.bits.vib.be/index.php/Analyze_your_own_microarray_data_in_R/Bioconductor. Accessed 9 May 2022
Jeong H, Mason SP, Barabási A-L, Oltvai ZN (2001) Lethality and centrality in protein networks. Nature 411:41–42. https://doi.org/10.1038/35075138
Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K (2016) KEGG: New perspectives on genomes, pathways, diseases and drugs. Nucleic Acids Res. https://doi.org/10.1093/nar/gkw1092
Kirkwood JM, Butterfield LH, Tarhini AA, Zarour H, Kalinski P, Ferrone S (2012) Immunotherapy of cancer in 2012. CA Cancer J Clin 62:309–335. https://doi.org/10.3322/caac.20132
Kutmon M, Ehrhart F, Willighagen EL, Evelo CT, Coort SL (2019) CyTargetLinker app update: a flexible solution for network extension in Cytoscape. F1000Res 7:ELIXIR-743. https://doi.org/10.12688/f1000research.14613.2
MacDonald V (2009) Chemotherapy: managing side effects and safe handling. Can Vet J 50:665–668
Matias-Barrios VM, Radaeva M, Song Y, Alperstein Z, Lee AR, Schmitt V, Lee J, Ban F, Xie N, Qi J, Lallous N, Gleave ME, Cherkasov A, Dong X (2021) Discovery of new catalytic topoisomerase II inhibitors for anticancer therapeutics. Front Oncol. https://doi.org/10.3389/fonc.2020.633142
Pathan M, Keerthikumar S, Ang C-S, Gangoda L, Quek CYJ, Williamson NA, Mouradov D, Sieber OM, Simpson RJ, Salim A, Bacic A, Hill AF, Stroud DA, Ryan MT, Agbinya JI, Mariadason JM, Burgess AW, Mathivanan S (2015) FunRich: an open access standalone functional enrichment and interaction network analysis tool. Proteomics 15:2597–2601. https://doi.org/10.1002/pmic.201400515
Perry MC, Doll DC, Freter CE (eds) (2012) Chemotherapy source book, 5th edn. Wolters Kluwer/Lippincott Williams & Wilkins, Philadelphia
Rambow F, Piton G, Bouet S, Leplat J-J, Baulande S, Marrau A, Stam M, Horak V, Vincent-Naulleau S (2008) Gene expression signature for spontaneous cancer regression in melanoma pigs. Neoplasia 10:714-IN5. https://doi.org/10.1593/neo.08344
Ratner S (ed) (1992) In: Karger S (ed) Mechanisms of lymphocyte extravasation, 1st edn
Ravindran Menon D, Luo Y, Arcaroli JJ, Liu S, KrishnanKutty LN, Osborne DG, Li Y, Samson JM, Bagby S, Tan A-C, Robinson WA, Messersmith WA, Fujita M (2018) CDK1 interacts with Sox2 and promotes tumor initiation in human melanoma. Cancer Res 78:6561–6574. https://doi.org/10.1158/0008-5472.CAN-18-0330
Ribero S, Gualano MR, Osella-Abate S, Scaioli G, Bert F, Sanlorenzo M, Balagna E, Fierro MT, Macripò G, Sapino A, Siliquini R, Quaglino P (2015) Association of histologic regression in primary melanoma with sentinel lymph node status: a systematic review and meta-analysis. JAMA Dermatol 151:1301–1307. https://doi.org/10.1001/jamadermatol.2015.2235
Roy PK, Kozma R, Majumder DD (2002) From neurocomputation to immunocomputation—a model and algorithm for fluctuation-induced instability and phase transition in biological systems. IEEE Trans Evol Comput 6:292–305. https://doi.org/10.1109/TEVC.2002.1011542
Sabidussi G (1966) The centrality index of a graph. Psychometrika 31:581–603. https://doi.org/10.1007/BF02289527
Salman T (2016) Spontaneous tumor regression. J Oncol Sci 2:1–4. https://doi.org/10.1016/j.jons.2016.04.008
Shannon P, Markiel A, Ozier O, Baliga NS, Wang JT, Ramage D, Amin N, Schwikowski B, Ideker T (2003) Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res 13:2498–2504. https://doi.org/10.1101/gr.1239303
Song L, Robson T, Doig T, Brenn T, Mathers M, Brown ER, Doherty V, Bartlett JMS, Anderson N, Melton DW (2013) DNA repair and replication proteins as prognostic markers in melanoma. Histopathology 62:343–350. https://doi.org/10.1111/j.1365-2559.2012.04362.x
Spontaneous cancer regression of melanoma - Search Results - PubMed [WWW Document] (2022) https://pubmed.ncbi.nlm.nih.gov/?term=spontaneous+cancer+regression+of+melanoma. Accessed 14 June 2022
Spontaneous regression of cancer or spontaneous remission of cancer - Search Results - PubMed [WWW Document] (2022) https://pubmed.ncbi.nlm.nih.gov/?term=spontaneous+regression+of+cancer+or+sponteneous++remission+of+cancer. Accessed 14 June 22
Szklarczyk D, Gable AL, Lyon D, Junge A, Wyder S, Huerta-Cepas J, Simonovic M, Doncheva NT, Morris JH, Bork P, Jensen LJ, von Mering C (2019) STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res 47:D607–D613. https://doi.org/10.1093/nar/gky1131
Tang Y, Li M, Wang J, Pan Y, Wu F-X (2015) CytoNCA: a cytoscape plugin for centrality analysis and evaluation of protein interaction networks. Biosystems 127:67–72. https://doi.org/10.1016/j.biosystems.2014.11.005
Tannock IF (1989) Combined modality treatment with radiotherapy and chemotherapy. Radiother Oncol 16:83–101. https://doi.org/10.1016/0167-8140(89)90025-X
The Human Protein Atlas [WWW Document] (2022) https://www.proteinatlas.org/. Accessed 6 Feb 2022
Treating Cancer with Immunotherapy | Types of Immunotherapy [WWW Document] (2022) https://www.cancer.org/treatment/treatments-and-side-effects/treatment-types/immunotherapy/what-is-immunotherapy.html. Accessed 8 May 2022
Vladimirova IG (2019) The energetics of regeneration processes. In: The Energetics of Regeneration Processes. De Gruyter, pp 243–256. https://doi.org/10.1515/9783110860511-027
Wendorff TJ, Schmidt BH, Heslop P, Austin CA, Berger JM (2012) The structure of DNA-bound human topoisomerase II alpha: conformational mechanisms for coordinating inter-subunit interactions with DNA cleavage. J Mol Biol 424:109–124. https://doi.org/10.1016/j.jmb.2012.07.014
Wishart DS, Feunang YD, Guo AC, Lo EJ, Marcu A, Grant JR, Sajed T, Johnson D, Li C, Sayeeda Z, Assempour N, Iynkkaran I, Liu Y, Maciejewski A, Gale N, Wilson A, Chin L, Cummings R, Le D, Pon A, Knox C, Wilson M (2018) DrugBank 5.0: a major update to the DrugBank database for 2018. Nucleic Acids Res 46:D1074–D1082. https://doi.org/10.1093/nar/gkx1037
Yang Y, Liu Q, Guo X, Yuan Q, Nian S, Kang P, Xu Z, Li L, Ye Y (2022) Systematic Pan-Cancer analysis identifies CDK1 as an immunological and Prognostic Biomarker. J Oncol. https://doi.org/10.1155/2022/8115474
Zahl P-H, Gøtzsche PC, Mæhlen J (2011) Natural history of breast cancers detected in the Swedish mammography screening programme: a cohort study. Lancet Oncol 12:1118–1124. https://doi.org/10.1016/S1470-2045(11)70250-9
Zhang Y, Hunter T (2014) Roles of Chk1 in cell biology and cancer therapy. Int J Cancer. https://doi.org/10.1002/ijc.28226.10.1002/ijc.28226
Acknowledgements
Bindu Kumari is thankful for the student opportunity furnished by Indian Institute of Technology – Banaras Hindu University, Varanasi. Deep appreciation is acknowledged for the support extended by the iHub NTIHAC Foundation, sponsored by Department of Science & Technology, Ministry of Science & Technology, Govt. of India.
Author information
Authors and Affiliations
Contributions
BK: conceptualisation, methodology, validation, formal analysis, writing–original draft, writing—review and editing. CS, RL, PP, AB: methodology, formal analysis, validation. PKR: conceptualisation, methodology, validation, formal analysis, writing—original draft, writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Research involving human participants and/or animals.
The work performed in the manuscript does not involve any human participants or animal preparations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kumari, B., Sakode, C., Lakshminarayanan, R. et al. A mechanistic analysis of spontaneous cancer remission phenomenon: identification of genomic basis and effector biomolecules for therapeutic applicability. 3 Biotech 13, 113 (2023). https://doi.org/10.1007/s13205-023-03515-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03515-0