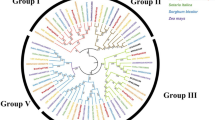
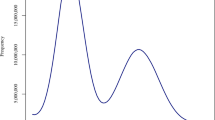
Abstract
Cercospora sesami is a plant pathogen that causes leaf spot disease in sesame plants worldwide. In this study, genome sequence assembly of C. sesami isolate Cers 52–10 (MCC 9069) was generated using native paired-end and mate-pair DNA sequencing based on the Illumina HiSeq 2500 platform. The genome assembly of C. sesami is 34.3 Mb in size with an N50 of 26,222 bp and an average GC content of 53.02%. A total number of 10,872 genes were predicted in this study, out of which 9,712 genes were functionally annotated. Genes assigned to carbohydrate-active enzyme classes were also identified during the study. A total of 80 putative effector candidates were predicted and functionally annotated. The C. sesami genome sequence is available at DDBJ/ENA/GenBank, and other associated information is submitted to Mendeley's data.




Similar content being viewed by others
Availability of Data and materials
The authors state that all the necessary data to confirm the conclusions presented in the article are entirely included within the article, and the supplementary data to this article have been deposited to Mendeley’s data with DOI number https://doi.org/10.17632/26m5grnb88.1.
References
Akinsanmi OA, Carvalhais LC (2020) Draft genome of the macadamia husk spot pathogen, Pseudocercospora macadamiae. Phytopathology 110:1503–1506. https://doi.org/10.1094/PHYTO-12-19-0460-A
Alice D, Nadarajan N (2007) Pulses: screening techniques and assessment for disease resistance. All India coordinated research project on MULLaRP.
Amarillas L, Estrada-Acosta M, León-Chan RG, López-Orona C, Lightbourn L (2020) First draft genome sequence resource of a strain of Pseudocercospora fijiensis isolated in North America. Phytopathology 110:1620–1622. https://doi.org/10.1094/PHYTO-04-20-0121-A
Andrews S (2010) FastQC: a quality control tool for high throughput sequence data.
Anilakumar KR, Pal A, Khanum F, Bawa AS (2010) Nutritional, medicinal and industrial uses of sesame (Sesamum indicum L.) seeds - an overview. Agric Conspec Sci 75:159–168
Armenteros JJA, Sønderby CK, Sønderby SK, Nielsen H, Winther O (2017) DeepLoc: prediction of protein subcellular localization using deep learning. Bioinformatics 33:3387–3395. https://doi.org/10.1093/bioinformatics/btx431
Armenteros JJA, Salvatore M, Emanuelsson O, Winther O, Von Heijne G, Elofsson A, Nielsen H (2019a) Detecting sequence signals in targeting peptides using deep learning. Life Sci Alliance 2:e201900429. https://doi.org/10.26508/lsa.201900429
Armenteros JJA, Tsirigos KD, Sønderby CK, Petersen TN, Winther O, Brunak S, von Heijne G, Nielsen H (2019b) SignalP 5.0 improves signal peptide predictions using deep neural networks. Nat Biotechnol 37:420–423. https://doi.org/10.1038/s41587-019-0036-z
Baker DN, Langmead B (2019) Dashing: fast and accurate genomic distances with hyperloglog. Genome Biol 20:1–12. https://doi.org/10.1186/s13059-019-1875-0
Cantarel BL, Coutinho PM, Rancurel C, Bernard T, Lombard V, Henrissat B (2009) The carbohydrate-active enzymes database (CAZy): an expert resource for glycogenomics. Nucleic Acids Res 37(suppl_1):D233–D238. https://doi.org/10.1093/nar/gkn663
Chand R, Kumar P, Singh V, Pal C (2013) Technique for spore production in Cercospora canescens. Indian Phytopathol 66:159–163
Chang TC, Salvucci A, Crous PW, Stergiopoulos I (2016) Comparative genomics of the sigatoka disease complex on banana suggests a link between parallel evolutionary changes in Pseudocercospora fijiensis and Pseudocercospora eumusae and increased virulence on the banana host. PLoS Genetics 12:e1005904. https://doi.org/10.1371/journal.pgen.1005904
Choi YW, Hyde KD, Ho WH (1999) Single spore isolation of fungi. Fungal Divers 3:29–38
Chupp C (1953) A monograph of fungus genus Cercospora. Cornel Univ, Ithaca, New York, p 667
El-Gebali S, Mistry J, Bateman A, Eddy SR, Luciani A, Potter SC et al (2019) The Pfam protein families database in 2019. Nucleic Acids Res 47:D427–D432. https://doi.org/10.1093/nar/gky995
Enikuomehin OA, Aduwo AM, Olowe VIO, Popoola AR, Oduwaye A (2010) Incidence and severity of foliar diseases of sesame (Sesamum indicum L.) intercropped with maize (Zea mays L.). Arch Phytopathol Plant Prot 43:972–986. https://doi.org/10.1080/03235400802214810
Goode MJ, Brown GR (1970) Detection and characterization of Cercospora citrullina isolates that sporulate readily in culture. Phytopathology 60:1502–1503. https://doi.org/10.1094/Phyto-60-1502
Goodwin SB, Ben M’Barek S, Dhillon B, Wittenberg AH, Crane CF, Hane JK et al (2011) Finished genome of the fungal wheat pathogen Mycosphaerella graminicola reveals dispensome structure, chromosome plasticity, and stealth pathogenesis. PLoS Genetics 7:e1002070. https://doi.org/10.1371/journal.pgen.1002070
Gostinčar C (2020) Towards genomic criteria for delineating fungal species. J Fungi 6:246. https://doi.org/10.3390/jof6040246
Götz S, Garcia-Gomez JM, Terol J, Williams TD, Nagaraj SH, Nueda MJ, Robles M, Talon M, Dopazo J, Conesa A (2008) High-throughput functional annotation and data mining with the Blast2GO suite. Nucleic Acids Res 36:3420–3435. https://doi.org/10.1093/nar/gkn176
Groenewald JZ, Nakashima C, Nishikawa J, Shin HD, Park JH, Jama AN, Groenewald M, Braun U, Crous PW (2013) Species concepts in Cercospora: spotting the weeds among the roses. Stud Mycol 75:115–170. https://doi.org/10.3114/sim0012
Gurevich A, Saveliev V, Vyahhi N, Tesler G (2013) QUAST: a quality assessment tool for genome assemblies. Bioinformatics 29:1072–1075. https://doi.org/10.1093/bioinformatics/btt086
Horton P, Park KJ, Obayashi T, Fujita N, Harada H, Adams-Collier CJ, Nakai K (2007) WoLF PSORT: protein localization predictor. Nucleic Acids Res 35(suppl_2):W585–W587. https://doi.org/10.1093/nar/gkm259
Huerta-Cepas J, Forslund K, Coelho LP, Szklarczyk D, Jensen LJ, Von Mering C et al (2017) Fast genome-wide functional annotation through orthology assignment by eggNOG-mapper. Mol Biol Evol 34:2115–2122. https://doi.org/10.1093/molbev/msx148
Jones P, Binns D, Chang HY, Fraser M, Li W, McAnulla C et al (2014) InterProScan 5: genome-scale protein function classification. Bioinformatics 30:1236–1240. https://doi.org/10.1093/bioinformatics/btu031
Kamal (2010) Cercosporoid fungi of India. Bishen Singh Mahendra Pal Singh, Dehradun (Uttarakhand), India
Kanehisa M, Goto S (2000) KEGG: kyoto encyclopedia of genes and genomes. Nucleic Acids Res 28:27–30. https://doi.org/10.1093/nar/28.1.27
Kanja C, Hammond-Kosack KE (2020) Proteinaceous effector discovery and characterization in filamentous plant pathogens. Mol Plant Pathol 21:1353–1376. https://doi.org/10.1111/mpp.12980
Krogh A, Larsson B, von Heijne G, Sonnhammer EL (2001) Predicting transmembrane protein topology with a hidden Markov model: application to complete genomes. J Mol Biol 305:567–580. https://doi.org/10.1006/jmbi.2000.4315
Lo Presti L, Lanver D, Schweizer G, Tanaka S, Liang L, Tollot M et al (2015) Fungal effectors and plant susceptibility. Annu Rev Plant Biol 66:513–545. https://doi.org/10.1146/annurev-arplant-043014-114623
Lyu X, Shen C, Fu Y, Xie J, Jiang D, Li G, Cheng J (2015) Comparative genomic and transcriptional analyses of the carbohydrate-active enzymes and secretomes of phytopathogenic fungi reveal their significant roles during infection and development. Sci Rep 5:1–16. https://doi.org/10.1038/srep15565
McWilliam H, Li W, Uludag M, Squizzato S, Park YM, Buso N et al (2013) Analysis tool web services from the EMBL-EBI. Nucleic Acids Res 41:W597–W600. https://doi.org/10.1093/nar/gkt376
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4326. https://doi.org/10.1093/nar/8.19.4321
Nguanhom J, Cheewangkoon R, Groenewald JZ, Braun U, To-Anun C, Crous PW (2015) Taxonomy and phylogeny of Cercospora spp. from Northern Thailand. Phytotaxa 233:27–48. https://doi.org/10.11646/phytotaxa.233.1.2
Ondov BD, Treangen TJ, Melsted P, Mallonee AB, Bergman NH, Koren S, Phillippy AM (2016) Mash: fast genome and metagenome distance estimation using MinHash. Genome Biol 17:1–14. https://doi.org/10.1186/s13059-016-0997-x
Pierleoni A, Martelli PL, Casadio R (2008) PredGPI: a GPI-anchor predictor. BMC Bioinform 9:1–11. https://doi.org/10.1186/1471-2105-9-392
R Core Team (2021) R: a language and environment for statistical computing. R Foundation for Statistical computing, Vienna, Austria. https://www.r-project.org/
Rehner SA, Samuels GJ (1995) Molecular systematics of the hypocreales: a teleomorph gene phylogeny and the status of their anamorphs. Can J Bot 73:816–823. https://doi.org/10.1139/b95-327
Saitou N, Nei M (1987) The neighbour-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425
Schubert M, Lindgreen S, Orlando L (2016) AdapterRemoval v2: rapid adapter trimming, identification, and read merging. BMC Res Notes 9:1–7. https://doi.org/10.1186/s13104-016-1900-2
Selin C, De Kievit TR, Belmonte MF, Fernando W (2016) Elucidating the role of effectors in plant-fungal interactions: progress and challenges. Front Microbiol 7:600. https://doi.org/10.3389/fmicb.2016.00600
Simao FA, Waterhouse RM, Ioannidis P, Kriventseva EV, Zdobnov EM (2015) BUSCO: assessing genome assembly and annotation completeness with single-copy orthologs. Bioinformatics 31:3210–3212. https://doi.org/10.1093/bioinformatics/btv351
Smit AFA, Hubley R, Green P (2015) RepeatMasker Open-4.0. 2013–2015.
Sonah H, Deshmukh RK, Bélanger RR (2016) Computational prediction of effector proteins in fungi: opportunities and challenges. Front Plant Sci 7:126. https://doi.org/10.3389/fpls.2016.00126
Sperschneider J, Dodds PN, Gardiner DM, Manners JM, Singh KB, Taylor JM (2015a) Advances and challenges in computational prediction of effectors from plant pathogenic fungi. PLoS Pathog 11:e1004806. https://doi.org/10.1371/journal.ppat.1004806
Sperschneider J, Gardiner DM, Dodds PN, Tini F, Covarelli L, Singh KB et al (2015b) EffectorP: predicting fungal effector proteins from secretomes using machine learning. New Phytol 210:743–761. https://doi.org/10.1111/nph.13794
Sperschneider J, Dodds PN, Gardiner DM, Singh KB, Taylor JM (2018) Improved prediction of fungal effector proteins from secretomes with effectorP 2.0. Mol Plant Pathol 19:2094–2110. https://doi.org/10.1111/mpp.12682
Stanke M, Morgenstern B (2005) AUGUSTUS: a web server for gene prediction in eukaryotes that allows user-defined constraints. Nucleic Acids Res 33(suppl_2):465–467. https://doi.org/10.1093/nar/gki458
Stergiopoulos I, de Wit PJ (2009) Fungal effector proteins. Annu Rev Phytopathol 47:233–263. https://doi.org/10.1146/annurev.phyto.112408.132637
Tajima F, Nei M (1984) Estimation of evolutionary distance between nucleotide sequences. Mol Biol Evol 1:269–285. https://doi.org/10.1093/oxfordjournals.molbev.a040317
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
UniProt Consortium (2021) UniProt: the universal protein knowledge base in 2021. Nucleic Acids Res 49:D480–D489. https://doi.org/10.1093/nar/gkaa1100
Vilgalys R, Hester M (1990) Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J Bacteriol 172:4238–4246. https://doi.org/10.1128/jb.172.8.4238-4246.1990
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR protocols: a guide to methods and application. Academic Press, San Diego, pp 315–322
Wijayawardene NN, Hyde KD, Rajeshkumar KC et al (2017) Notes for genera: ascomycota. Fungal Divers 86:1–594. https://doi.org/10.1007/s13225-017-0386-0
Wilken PM, Aylward J, Chand R et al (2020) IMA genome - F13. IMA Fungus 11:1–17. https://doi.org/10.1186/s43008-020-00039-7
Wingfield BD, Berger DK, Steenkamp ET et al (2017) Draft genome of Cercospora zeina, Fusarium pininemorale, Hawksworthiomyces lignivorus, Huntiella decipiens and Ophiostoma ips. IMA Fungus 8:385–396. https://doi.org/10.5598/imafungus.2017.08.02.10
Wingfield BD, De Vos L, Wilson AM et al (2022) IMA Genome - F16. IMA Fungus 13:1–22. https://doi.org/10.1186/s43008-022-00089-z
Xu H, Luo X, Qian J, Pang X, Song J, Qian G, Chen J, Chen S (2012) FastUniq: a fast de novoduplicate removal tool for paired short reads. PLoS One. 7:e52249. https://doi.org/10.1371/journal.pone.0052249
Zhang H, Miao H, Wang L, Qu L, Liu H, Wang Q, Yue M (2013) Genome sequencing of the important oilseed crop Sesamum indicum L. Genome Biol 14:1–9. https://doi.org/10.1186/gb-2013-14-1-401
Zhang H, Yohe T, Huang L, Entwistle S, Wu P, Yang Z, Busk PK, Xu Y, Yin Y (2018) dbCAN2: a meta server for automated carbohydrate-active enzyme annotation. Nucleic Acids Res 46:W95–W101. https://doi.org/10.1093/nar/gky418
Zimin AV, Marçais G, Puiu D, Roberts M, Salzberg SL, Yorke JA (2013) The MaSuRCA genome assembler. Bioinformatics 29:2669–2677. https://doi.org/10.1093/bioinformatics/btt476
Acknowledgements
Shagun Sinha is thankful to the Council of Scientific and Industrial Research (CSIR), New Delhi, India, for the Senior Research Fellowship (SRF) and the Department of Biotechnology (DBT) and the British Council for Newton-Bhabha PhD placement fellowship 2019-2020. All authors are thankful to the Head and Coordinator, Center of Advanced Studies (CAS) in Botany, Banaras Hindu University (BHU), Varanasi, India and the Head, Department of Mycology and Plant Pathology, Institute of Agricultural Sciences, BHU for financial and infrastructural support.
Funding
Council of Scientific and Industrial Research, India, 09/013(0824)/2018-EMR-I, Shagun Sinha.
Author information
Authors and Affiliations
Contributions
RC, RNK and SN planned the whole-genome sequencing study. RC and SS contributed to the identification, isolation and characterization of the sequenced pathogen. SS and SN performed DNA extraction, gene annotation and phylogenetic analysis. SS and DKG helped in bioinformatics analysis. SS and SN drafted the manuscript. RNK was involved in adding critical inputs to the manuscript. All authors have read and contributed to the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Research involving human participants and/or animals
The authors declare that the research did not involve human participants and/or animals.
Informed consent
All authors have given consent to the publication of this manuscript in 3 Biotech.
Additional information
Accession Numbers: The Cercospora sesami genomic data have been deposited as accession JAASLG000000000 in DDBJ/ENA/GenBank. The version described in this paper is version JAASLG010000000. The genome raw sequencing data and the reported assembly are associated with NCBI BioProject: PRJNA613165 and BioSample: SAMN14395398 within GenBank. The SRA accession of Cercospora sesami is SRX7967432.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sinha, S., Navathe, S., Singh, S. et al. Genome sequencing and annotation of Cercospora sesami, a fungal pathogen causing leaf spot to Sesamum indicum. 3 Biotech 13, 55 (2023). https://doi.org/10.1007/s13205-023-03468-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03468-4