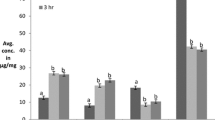
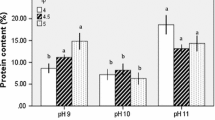
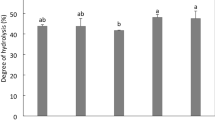
Abstract
Tamarind seeds are one of the major sources of different nutrients and minerals and are used as antioxidant components in various food products. Bioactive peptides are released by enzymatic hydrolysis of major food proteins followed by different separation techniques to isolate them from the mixture of other components. Further incorporation of bioactive peptides in various food products may lead to more diversification, fulfilling different nutritional requirements, and promoting health benefits. The objective of this study was to optimize the parameters for the formation of tamarind protein hydrolysates with the use of pepsin and trypsin. It was observed that pepsin and trypsin derived hydrolysate showed the maximum degree of hydrolysis and other bioactive characteristics (DPPH activity, reducing power, ABTS activity, and antidiabetic activity) under optimized conditions. High-performance liquid chromatography was performed for amino acid profiling on trypsin derived protein hydrolysate. From this study, pepsin and trypsin derived hydrolysate can be considered as a rich source of natural antioxidants for developing food formulation.





Similar content being viewed by others
Code availability
Not applicable.
References
Alam MN, Bristi NJ, Rafiquzzaman M (2013) Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm J 21(2):143–152. https://doi.org/10.1016/j.jsps.2012.05.002
Baharuddin SH, Mustahil NA, Abdullah AA, Sivapragasam M, Moniruzzaman M (2016) Ecotoxicity study of amino acid ionic liquids towards Danio rerio fish: effect of cations. Proc Eng 148:401–408. https://doi.org/10.1016/j.proeng.2016.06.431
Bhandari D, Rafiq S, Gat Y, Gat P, Waghmare R, Kumar V (2019) A review on bioactive peptides: physiological functions, bioavailability and safety. Int J Pept Res Ther. https://doi.org/10.1007/s10989-019-09823-5
Bougatef A, Hajji M, Balti R, Lassoued I, Triki-Ellouz Y, Nasri M (2009) Antioxidant and free radical-scavenging activities of smooth hound (Mustelus mustelus) muscle protein hydrolysates obtained by gastrointestinal proteases. Food Chem 114(4):1198–1205. https://doi.org/10.1016/j.foodchem.2008.10.075
Chaudhari MG, Joshi BB, Mistry KN (2013) In vitro antidiabetic and anti-inflammatory activity of stem bark of Bauhinia purpurea. Bull Pharm Med Sci (BOPAMS) 1(2)
Chi CF, Hu FY, Wang B, Li T, Ding GF (2015) Antioxidant and anticancer peptides from the protein hydrolysate of blood clam (Tegillarca granosa) muscle. J Func Foods 15:301–313. https://doi.org/10.1016/j.jff.2015.03.045
Dey SS, Dora KC (2014) Antioxidative activity of protein hydrolysate produced by alcalase hydrolysis from shrimp waste (Penaeus monodon and Penaeus indicus). J Food Sci Technol 51(3):449–457. https://doi.org/10.1007/s13197-011-0512-z
Guerard F, Guimas L, Binet A (2002) Production of tuna waste hydrolysates by a commercial neutral protease preparation. J Mol Catal B Enzym 19:489–498. https://doi.org/10.1016/S1381-1177(02)00203-5
Jian J, Tian QY, Hettinghouse A, Zhao S, Liu H, Wei J, Overkleeft HS (2016) Progranulin recruits HSP70 to β-glucocerebrosidase and is therapeutic against Gaucher disease. EBioMedicine 13:212–224. https://doi.org/10.1016/j.ebiom.2016.10.010
Kanbargi KD, Sonawane SK, Arya SS (2016) Functional and antioxidant activity of Ziziphus jujube seed protein hydrolysates. J Food Meas Character 10(2):226–235. https://doi.org/10.1007/s11694-015-9297-5
Kanbargi KD, Sonawane SK, Arya SS (2017) Encapsulation characteristics of protein hydrolysate extracted from Ziziphus jujube seed. Int J Food Prop 20(12):3215–3224. https://doi.org/10.1080/10942912.2017.1282516
Kedare SB, Singh RP (2011) Genesis and development of DPPH method of antioxidant assay. J Food Sci Technol 48(4):412–422. https://doi.org/10.1007/s13197-011-0251-1
Kumanan R, Manimaran S, Saleemulla K, Dhanabal SP, Nanjan MJ (2010) Screening of bark of Cinnamomum tamala (Lauraceae) by using α -amylase inhibition assay for anti-diabetic activity. Int J Pharm Biomed Res 1(2):69–72
Lv C, Jia X, Li M, Yang J, Zhao G (2011) Optimization of extraction process of crude protein from grape seeds by RSM. Food Sci Technol Res 17(5):437–445. https://doi.org/10.3136/fstr.17.437
Mahmoud A-H et al (2017) Angiotensin I-converting enzyme inhibitory activity and antioxidant capacity of bioactive peptides derived from enzymatic hydrolysis of buffalo milk proteins. Int Dairy J 66:91–98
Moon JK, Shibamoto T (2009) Antioxidant assays for plant and food components. J Agric Food Chem 57(5):1655–1666. https://doi.org/10.1021/jf803537k
Naqash SY, Nazeer RA (2010) Antioxidant activity of hydrolysates and peptide fractions of Nemipterus japonicus and Exocoetus volitans muscle. J Aquat Food Prod Technol 19(3–4):180–192. https://doi.org/10.1080/10498850.2010.506256
Natukunda S, Muyonga JH, Mukisa IM (2016) Effect of tamarind (Tamarindus indica L.) seed on antioxidant activity, phytocompounds, physicochemical characteristics, and sensory acceptability of enriched cookies and mango juice. Food Sci Nutr 4(4):494–507. https://doi.org/10.1002/fsn3.311
Ngoh YY, Gan CY (2016) Enzyme-assisted extraction and identification of antioxidative and α-amylase inhibitory peptides from Pinto beans (Phaseolus vulgaris cv. Pinto). Food Chem 190:331–337. https://doi.org/10.1016/j.foodchem.2015.05.120
Nielsen PM, Petersen D, Dambmann C (2001) Improved method for determining food protein degree of hydrolysis. J Food Sci 66(5):642–646. https://doi.org/10.1111/j.1365-2621.2001.tb04614.x
Razali N, Mat Junit S, Ariffin A, Ramli NS, Abdul Aziz A (2015) Polyphenols from the extract and fraction of T. indica seeds protected HepG2 cells against oxidative stress. BMC Complementary Altern Med 15:438. https://doi.org/10.1186/s12906-015-0963-2
Sathiavelu A, Sangeetha S, Archit R, Mythili S (2013) In vitro antidiabetic activity of aqueous extract of the medicinal plants Nigella sativa, Eugenia jambolana, Andrographis paniculata and Gymnema sylvestre. Int J Drug Dev Res 5(2):323–328
Singh D, Wangchu L, Moond SK (2007) Processed products of Tamarind. Nat Prod Rad 6(4):315–321
Sonawane SK, Arya SS (2017a) Bioactive L acidissima protein hydrolysates using Box-Behnken design. 3 Biotech 7(3):218. https://doi.org/10.1007/s13205-017-0862-y
Sonawane SK, Arya SS (2017b) Citrullus lanatus protein hydrolysate optimization for antioxidant potential. J Food Meas Charact 11(4):1834–1843. https://doi.org/10.1007/s11694-017-9565-7
Spiegel M, Kapusta K, Kołodziejczyk W, Saloni J, Żbikowska B, Hill GA, Sroka Z (2020) Antioxidant activity of selected phenolic acids–ferric reducing antioxidant power assay and QSAR analysis of the structural features. Molecules 25(13):3088
Zhu D, Lai Y, Shelat PB, Hu C, Sun GY, Lee JC (2006) Phospholipases A2 mediate amyloid-β peptide-induced mitochondrial dysfunction. J Neurosci 26(43):11111–11119. https://doi.org/10.1523/JNEUROSCI.3505-06.2006
Funding
Authors declare that no financial support is received for proposed work.
Author information
Authors and Affiliations
Contributions
DB and SS have drafted the manuscript and YG have guided and have conceded proof reading of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Authors have read and approved the manuscript and all are aware of its submission to 3 Biotech.
Consent for publication
Not applicable.
Human and animal rights
Author declares that proposed work does not contain any animal and human studies.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhandari, D., Shirsat, S. & Gat, Y. Bioactive characterization and optimization of Tamarindus indica seed protein hydrolysate: technology and application of natural enzymes. 3 Biotech 12, 338 (2022). https://doi.org/10.1007/s13205-022-03386-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03386-x