Abstract
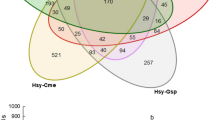
Aphid populations were collected on cowpea, dolichos, redgram and black gram from Belagavi and Udupi locations. The samples were shotgun sequenced using the Illumina NovaSeq 6000 system to understand the spatial distribution and community structure of microbiota (especially bacteria) associated with aphids. In the present study, we identified obligatory nutritional symbiont Buchnera aphidicola and facultative symbionts Rickettsia sp. and Bacteroidetes endosymbiont of Geopemphigus sp. in all the aphid samples studied, although in varied abundance. On the other hand, Serratia symbiotica, Arsenophonus sp. and Acinetobacter sp. were only found in aphids on specific host plants, suggesting that host plants might influence the bacterial community structure. Furthermore, our study revealed that microbiota other than bacteria were highly insignificant in the aphid populations. Additionally, functional annotation of aphid metagenomes identified several pathways and enzymes involved in various physiological and ecological functions. Amino acid and vitamin biosynthesis-related pathways were predominant than carbohydrate metabolism, owing to their feeding habit and nutritional requirement. Chaperones related to stress tolerance such as GroEL and DnaK were identified. Enzymes involved in toxic chemical metabolisms such as glutathione transferase, phosphodiesterases and ABC transferases were observed. These enzymes may confer resistance to pesticides in the aphid populations. Overall, our results support the importance of host plants in structuring bacterial communities in aphids and show the functional roles of symbionts in aphid survival and development. Thus, these findings can be the basis for further detailed investigations and devising better strategies to manage the pests in field conditions.




Similar content being viewed by others
Data availability
The raw shotgun metagenomic sequence data of the present study was deposited in the NCBI SRA database with the SRA Accession Numbers: SRR14744453—SRR14744457 under the BioProject PRJNA735325.
References
Bansal R, Hulbert S, Schemerhorn B, Reese JC, Whitworth RJ, Stuart JJ, Chen MS (2011) Hessian fly-associated bacteria: transmission, essentiality, and composition. PLoS ONE 6:e23170
Berendsen RL, Pieterse CM, Bakker PA (2012) The rhizosphere microbiome and plant health. Trends Plant Sci 17:478–486
Besemer J, Borodovsky M (1999) Heuristic approach to deriving models for gene finding. Nucleic Acids Res 27:3911–3920
Blackman RL, Eastop VF (2007) Taxonomic issues. In: van Emden HF, Harrington R (eds) Aphids as crop pests. CABI, Wallingford, pp 1–29
Blankenchip CL, Michels DE, Braker HE, Goffredi SK (2018) Diet breadth and exploitation of exotic plants shift the core microbiome of Cephaloleia, a group of tropical herbivorous beetles. PeerJ 6:e4793
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30:2114–2120
Brady CM, White JA (2013) Cowpea aphid (Aphis craccivora) associated with different host plants has different facultative endosymbionts. Ecol Entomol 38:433–437
Brady CM, Asplen MK, Desneux N, Heimpel GE, Hopper KR, Linnen CR et al (2014) Worldwide populations of the aphid Aphis craccivora are infected with diverse facultative bacterial symbionts. Microb Ecol 67:195–204
Buchfink B, Xie C, Huson DH (2015) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12:59–60
Burke GR, Moran NA (2011) Massive genomic decay in Serratia symbiotica, a recently evolved symbiont of aphids. Genome Biol Evol 3:195–208
Burke G, Fiehn O, Moran N (2010) Effects of facultative symbionts and heat stress on the metabolome of pea aphids. ISME J 4:242–252
Caspi-Fluger A, Inbar M, Mozes-Daube N, Katzir N, Portnoy V, Belausov E et al (2012) Horizontal transmission of the insect symbiont Rickettsia is plant-mediated. Proc R Soc B 279:1791–1796
Chen DQ, Montllor CB, Purcell AH (2000) Fitness effects of two facultative endosymbiotic bacteria on the pea aphid, Acyrthosiphon pisum, and the blue alfalfa aphid A Kondoi. Entomol Exp Appl 95:315–323
Chiel E, Gottlieb Y, Zchori-Fein E, Mozes-Daube N, Katzir N, Inbar M, Ghanim M (2007) Biotype-dependent secondary symbiont communities in sympatric populations of Bemisia tabaci. Bull Entomol Res 97:407
Chong RA, Moran NA (2018) Evolutionary loss and replacement of Buchnera, the obligate endosymbiont of aphids. ISME J 12:898–908
Chong J, Liu P, Zhou G, Xia J (2020) Using MicrobiomeAnalyst for comprehensive statistical, functional, and meta-analysis of microbiome data. Nat Protoc 15:799–821
Clark AG, Shamaan NA, Sinclair MD, Dauterman WC (1986) Insecticide metabolism by multiple glutathione S-transferases in two strains of the house fly, Musca domestica (L). Pestic Biochem Physiol 25:169–175
Costopoulos K, Kovacs JL, Kamins A, Gerardo NM (2014) Aphid facultative symbionts reduce survival of the predatory lady beetle Hippodamia convergens. BMC Ecol 14:1–7
Daumann LJ, Larrabee JA, Ollis D, Schenk G, Gahan LR (2014) Immobilization of the enzyme GpdQ on magnetite nanoparticles for organophosphate pesticide bioremediation. J Inorg Biochem 131:1–7
Dhariwal A, Chong J, Habib S, King IL, Agellon LB, Xia J (2017) MicrobiomeAnalyst: a web-based tool for comprehensive statistical, visual and meta-analysis of microbiome data. Nucleic Acids Res 45:W180–W188
Douglas AE (1996) Reproductive failure and the free amino acid pools in pea aphids (Acyrthosiphon pisum) lacking symbiotic bacteria. J Insect Physiol 42:247–255
Douglas AE (2006) Phloem-sap feeding by animals: problems and solutions. J Exp Bot 57:747–754
Douglas AE (2011) Lessons from studying insect symbioses. Cell Host Microbe 10:359–367
Feder ME, Hofmann GE (1999) Heat-shock proteins, molecular chaperones, and the stress response: evolutionary and ecological physiology. Annu Rev Physiol 61:243–282
Ferrari J, West JA, Via S, Godfray HCJ (2012) Population genetic structure and secondary symbionts in host-associated populations of the pea aphid complex. Evolution 66:375–390
Gómez-Valero L, Soriano-Navarro M, Pérez-Brocal V, Heddi A, Moya A, García-Verdugo JM, Latorre A (2004) Coexistence of Wolbachia with Buchnera aphidicola and a secondary symbiont in the aphid Cinara cedri. J Bacteriol 186:6626
Gott RC, Kunkel GR, Zobel ES, Lovett BR, Hawthorne DJ (2017) Implicating ABC transporters in insecticide resistance: research strategies and a decision framework. J Econ Entomol 110:667–677
Guay JF, Boudreault S, Michaud D, Cloutier C (2009) Impact of environmental stress on aphid clonal resistance to parasitoids: role of Hamiltonella defensa bacterial symbiosis in association with a new facultative symbiont of the pea aphid. J Insect Physiol 55:919–926
Guidolin AS, Cônsoli FL (2017) Symbiont diversity of Aphis (Toxoptera) citricidus (Hemiptera: Aphididae) as influenced by host plants. Microb Ecol 73:201–210
Handelsman J (2004) Metagenomics: application of genomics to uncultured microorganisms. Microbiol Mol Biol Rev 68:669–685
Hansen T (2018) Identifying mechanisms of host plant specialization in Aphis craccivora and its bacterial symbionts. Dissertation, University of Kentucky. https://doi.org/10.13023/ETD.2018.018
Hulle M, d’Acier AC, Bankhead-Dronnet S, Harrington R (2010) Aphids in the face of global changes. C R Biol 333:497–503
Hunter DJ, Torkelson JL, Bodnar J, Mortazavi B, Laurent T, Deason J et al (2015) The Rickettsia endosymbiont of Ixodes pacificus contains all the genes of de novo folate biosynthesis. PLoS ONE 10:e0144552
Huson DH, Auch AF, Qi J, Schuster SC (2007) MEGAN analysis of metagenomic data. Genome Res 17:377–386
Jones RT, Bressan A, Greenwell AM, Fierer N (2011) Bacterial communities of two parthenogenetic aphid species cocolonizing two host plants across the Hawaiian Islands. Appl Environ Microbiol 77:8345
Kaakeh W, Dutcher JD (1993) Effect of rainfall on population abundance of aphids (Homoptera Aphididae) on pecan. J Entomol Sci 28:283–283
Kamphuis LG, Gao L, Singh KB (2012) Identification and characterization of resistance to cowpea aphid (Aphis craccivora Koch) in Medicago truncatula. BMC Plant Biol 12:1–12
Koga R, Tsuchida T, Fukatsu T (2003) Changing partners in an obligate symbiosis: a facultative endosymbiont can compensate for loss of the essential endosymbiont Buchnera in an aphid. Proc R Soc B 270:2543–2550
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357
Leather SR (1985) Atmospheric humidity and aphid reproduction. Z Für Angew Entomol 100:510–513
Lenhart PA, White JA (2020) Endosymbionts facilitate rapid evolution in a polyphagous herbivore. J Evol Biol 33:1507–1511
Leonardo TE (2004) Removal of a specialization-associated symbiont does not affect aphid fitness. Ecol Lett 7:461–468
Li D, Liu CM, Luo R, Sadakane K, Lam TW (2015) MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics 31:1674–1676
Llorca O, Galán A, Carrascosa JL, Muga A, Valpuesta JM (1998) GroEL under heat-shock: switching from a folding to a storing function. J Biol Chem 273:32587–32594
Ma CS, Hau B, Poehling HM (2004) Effects of pattern and timing of high temperature exposure on reproduction of the rose grain aphid, Metopolophium dirhodum. Entomol Exp Appl 110:65–71
Ma YJ, He HP, Zhao HM, Xian YD, Guo H, Liu B, Xue K (2021) Microbiome diversity of cotton aphids (Aphis gossypii) is associated with host alternation. Sci Rep 11:1–10
McLean AHC, Van Asch M, Ferrari J, Godfray HCJ (2011) Effects of bacterial secondary symbionts on host plant use in pea aphids. Proc R Soc B 278:760–766
Medina RF, Nachappa P, Tamborindeguy C (2011) Differences in bacterial diversity of host-associated populations of Phylloxera notabilis Pergande (Hemiptera: Phylloxeridae) in pecan and water hickory. J Evol Biol 24:761–771
Montllor CB, Maxmen A, Purcell AH (2002) Facultative bacterial endosymbionts benefit pea aphids Acyrthosiphon pisum under heat stress. Ecol Entomol 27:189–195
Moran NA, Wernegreen JJ (2000) Lifestyle evolution in symbiotic bacteria: insights from genomics. Trends Ecol Evol 15:321–326
Moran NA, Munson MA, Baumann P, Ishikawa H (1993) A molecular clock in endosymbiotic bacteria is calibrated using the insect hosts. Proc R Soc B Biol Sci 253:167–171
Moran NA, McCutcheon JP, Nakabachi A (2008) Genomics and evolution of heritable bacterial symbionts. Annu Rev Genet 42:165–190
Najar-Rodríguez AJ, McGraw EA, Mensah RK, Pittman GW, Walter GH (2009) The microbial flora of Aphis gossypii: patterns across host plants and geographical space. J Invertebr Pathol 100:123–126
Oliver KM, Moran NA, Hunter MS (2005) Variation in resistance to parasitism in aphids is due to symbionts not host genotype. Proc Natl Acad Sci USA 102:12795–12800
Oliver KM, Moran NA, Hunter MS (2006) Costs and benefits of a superinfection of facultative symbionts in aphids. Proc R Soc B 273:1273–1280
Oliver KM, Campos J, Moran NA, Hunter MS (2008) Population dynamics of defensive symbionts in aphids. Proc R Soc B 275:293–299
Oliver KM, Degnan PH, Hunter MS, Moran NA (2009) Bacteriophages encode factors required for protection in a symbiotic mutualism. Science 325:992–994
Oliver KM, Degnan PH, Burke GR, Moran NA (2010) Facultative symbionts in aphids and the horizontal transfer of ecologically important traits. Annu Rev Entomol 55:247–266
Overbeek R, Olson R, Pusch GD, Olsen GJ, Davis JJ, Disz T et al (2014) The SEED and the Rapid Annotation of microbial genomes using Subsystems Technology (RAST). Nucleic Acids Res 42:D206–D214
Pons I, Renoz F, Noël C, Hance T (2019) Circulation of the cultivable symbiont Serratia symbiotica in aphids is mediated by plants. Front Microbiol 10:764
Rao Q, Rollat-Farnier PA, Zhu DT, Santos-Garcia D, Silva FJ, Moya A et al (2015) Genome reduction and potential metabolic complementation of the dual endosymbionts in the whitefly Bemisia tabaci. BMC Genom 16:1–13
Ravin NV, Mardanov AV, Skryabin KG (2015) Metagenomics as a tool for the investigation of uncultured microorganisms. Russ J Genet 51:431–439
Russell JA, Moran NA (2006) Costs and benefits of symbiont infection in aphids: variation among symbionts and across temperatures. Proc R Soc B 273:603–610
Sakurai M, Koga R, Tsuchida T, Meng XY, Fukatsu T (2005) Rickettsia symbiont in the pea aphid Acyrthosiphon pisum: novel cellular tropism, effect on host fitness, and interaction with the essential symbiont Buchnera. Appl Environ Microbiol 71:4069–4075
Sato S, Ishikawa H (1997) Structure and expression of the dnaKJ operon of Buchnera, an intracellular symbiotic bacteria of aphid. J Biochem 122:41–48
Scarborough CL, Ferrari J, Godfray HCJ (2005) Aphid protected from pathogen by endosymbiont. Science 310:1781–1781
Schloss PD, Handelsman J (2005) Metagenomics for studying unculturable microorganisms: cutting the Gordian knot. Genome Biol 6:1–4
Sepúlveda DA, Zepeda-Paulo F, Ramírez CC, Lavandero B, Figueroa CC (2017) Diversity, frequency, and geographic distribution of facultative bacterial endosymbionts in introduced aphid pests. Insect Sci 24:511–521
Sivakumar G, Rangeshwaran R, Yandigeri MS, Mohan M, Venkatesan T, Verghese A (2016) Diversity of culturable gut bacteria associated with the field populations of cotton leafhopper (Amrasca biguttula biguttula) in India. Indian J Agric Sci 86:208–215
Skaljac M, Vogel H, Wielsch N, Mihajlovic S, Vilcinskas A (2019) Transmission of a protease-secreting bacterial symbiont among pea aphids via host plants. Front Physiol 10:438
Tian PP, Chang CY, Miao NH, Li MY, Liu XD (2019) The facultative endosymbiont Arsenophonus infections alter aphid (Aphis gossypii) performance on the amino acid-deficient diet. Appl Environ Microbiol 85:e01407-e1419
Trivedi M, Singh R (2014) Systematics and nymphal characteristics of black bean aphid, Aphis craccivora Koch (Homoptera: Aphididae). Int J Life Sci 3:205–224
Tsuchida T, Koga R, Fukatsu T (2004) Host plant specialization governed by facultative symbiont. Science 303:1989–1989
Vorburger C, Gehrer L, Rodriguez P (2010) A strain of the bacterial symbiont Regiella insecticola protects aphids against parasitoids. Biol Lett 6:109–111
Wagner SM, Martinez AJ, Ruan YM, Kim KL, Lenhart PA, Dehnel AC et al (2015) Facultative endosymbionts mediate dietary breadth in a polyphagous herbivore. Funct Ecol 29:1402–1410
Weldon SR, Oliver KM (2016) Diverse bacteriophage roles in an aphid-bacterial defensive mutualism. In: Hurst C (ed) The mechanistic benefits of microbial symbionts. Advances in environmental microbiology, vol 2. Springer, Cham, pp 173–206
Xu S, Jiang L, Qiao G, Chen J (2020) The bacterial flora associated with the polyphagous aphid Aphis gossypii Glover (Hemiptera: Aphididae) is strongly affected by host plants. Microb Ecol 79:971–984
Zhang YC, Cao WJ, Zhong LR, Godfray HCJ, Liu XD (2016) Host plant determines the population size of an obligate symbiont (Buchnera aphidicola) in aphids. Appl Environ Microbiol 82:2336
Zhao Y, Zhang S, Luo JY, Wang CY, Lv LM, Cui JJ (2016) Bacterial communities of the cotton aphid Aphis gossypii associated with Bt cotton in northern China. Sci Rep 6:1–8
Zhu W, Lomsadze A, Borodovsky M (2010) Ab initio gene identification in metagenomic sequences. Nucleic Acids Res 38:e132–e132
Zytynska SE, Meyer ST, Sturm S, Ullmann W, Mehrparvar M, Weisser WW (2016) Secondary bacterial symbiont community in aphids responds to plant diversity. Oecologia 180:735–747
Zytynska SE, Tighiouart K, Frago E (2021) Benefits and costs of hosting facultative symbionts in plant-sucking insects: a meta-analysis. Mol Ecol 30:2483–2494
Acknowledgements
This study was funded by the Empowerment and Equity Opportunities for Excellence in Science (EMEQ) Grant (Project Number: EEQ/2017/000726) of Science and Engineering Research Board (SERB), Department of Science & Technology, Government of India to Shivanna B. We thank Dr. Sunil Joshi, Principal Scientist, ICAR-NBAIR, Bengaluru, for the taxonomic identification of aphid samples. We are greatly thankful to Molsys Pvt. Ltd., Bangalore, for their guidance during metagenomic analysis.
Author information
Authors and Affiliations
Contributions
SB conceived the idea and designed the study, MMP and MNH performed the experiment, MMP analysed the results and drafted the manuscript, and MKP, SB, PBP, and KS revised the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pawar, M.M., Shivanna, B., Prasannakumar, M.K. et al. Spatial distribution and community structure of microbiota associated with cowpea aphid (Aphis craccivora Koch). 3 Biotech 12, 75 (2022). https://doi.org/10.1007/s13205-022-03142-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03142-1