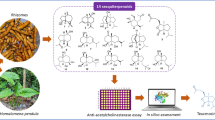
Abstract
Alzheimer’s disease (AD) is a neurodegenerative disease and the fourth leading cause of death after cardiovascular disease, tumors, and stroke. Acetylcholinesterase (AChE) inhibitors, which are based on cholinergic damage, remain the mainstream drugs to alleviate AD-related symptoms. This study aimed to explore novel AChE inhibitors produced by the endophytic fungus FL15 from Huperzia serrata. The fungus was identified as Talaromyces aurantiacus FL15 according to its morphological characteristics and ITS, 18S rDNA, and 28S rDNA sequence analysis. Subsequently, seven natural metabolites were isolated from strain FL15, and identified as asterric acid (1), methyl asterrate (2), ethyl asterrate (3), emodin (4), physcion (5), chrysophanol (6), and sulochrin (7). Compounds 1–3, which possess a diphenyl ether structure, exhibited highly selective and moderate AChE inhibitory activities with IC50 values of 66.7, 23.3, and 20.1 μM, respectively. The molecular docking analysis showed that compounds 1–3 interacted with the active catalytic site and peripheral anionic site of AChE, and the esterification substitution groups at position 8 of asterric acid may contribute to its bioactivity. The asterric acid derivatives showed highly selective and moderate AChE inhibitory activities, probably via interaction with the peripheral anionic site and catalytic site of AChE. To the best of our knowledge, this study was the first report of the AChE inhibitory activity of asterric acid derivatives, which opens new perspectives for the design of more effective derivatives that could serve as a drug carrier for new chemotherapeutic agents to treat AD.






Similar content being viewed by others
References
Bartus RT, Dean RL, Beer B et al (1982) The cholinergic hypothesis of geriatric memory dysfunction. Science 217:408–417. https://doi.org/10.1126/science.7046051
Biasetto CR, Somensi A, Sordi R et al (2020) The new koninginins T-U from Phomopsis stipata, an endophytic fungus isolated from Styrax camporum pohl. Phytochem Lett 36:106–110
Cao D, Sun P, Bhowmick S et al (2021) Secondary metabolites of endophytic fungi isolated from Huperzia serrata. Fitoterapia 155:104970. https://doi.org/10.1016/j.fitote.2021.104970
Chapla VM, Zeraik ML, Ximenes VF et al (2014) Bioactive secondary metabolites from Phomopsis sp., an endophytic fungus from Senna spectabilis. Molecules 19:6597–6608. https://doi.org/10.3390/molecules19056597
Chapla VM, Honório AE, Gubiani JR et al (2020) Acetylcholinesterase inhibition and antifungal activity of cyclohexanoids from the endophytic fungus Saccharicola sp. Phytochem Lett 39:116–123
Chen XW, Yang ZD, Li XF et al (2019) Colletotrichine B, a new sesquiterpenoid from Colletotrichum gloeosporioides GT-7, a fungal endophyte of Uncaria rhynchophylla. Nat Prod Res 33:108–112. https://doi.org/10.1080/14786419.2018.1437437
Chen CM, Chen WH, Pang XY et al (2021) Pyrrolyl 4-quinolone alkaloids from the mangrove endophytic fungus Penicillium steckii SCSIO 41025: Chiral resolution, configurational assignment, and enzyme inhibitory activities. Phytochem 186:112730
Curtis RF, Hassall CH, Jones DW et al (1960) The biosynthesis of phenols. Part II. Asterric acid, a metatoblic product of Aspergillus terreus Thom. J Chem Soc. https://doi.org/10.1039/JR9600004838
Dai Y, Li K, She J et al (2020) Lipopeptide epimers and a phthalide glycerol ether with AChE inhibitory activities from the marine-derived fungus Cochliobolus Lunatus SCSIO41401. Mar Drugs 18:547. https://doi.org/10.3390/md18110547
Devidas SB, Rahmatkar SN, Singh R et al (2021) Amelioration of cognitive deficit in zebrafish by an undescribed anthraquinone from Juglans regia L: an in-silico, in-vitro and in-vivo approach. Eur J Pharmacol 906:174234. https://doi.org/10.1016/j.ejphar.2021.174234
Ellman GL, Courtney KD, Andres V et al (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Fang MJ, Fang H, Li WJ et al (2012) A new diphenyl ether from Phoma sp. strain, SHZK-2. Nat Prod Res 26:1224–1228. https://doi.org/10.1080/14786419.2011.559947
Figueroa L, Jimenez C, Rodríguez ́J, et al (2015) 3-Nitroasterric acid derivatives from an antarctic sponge-derived Pseudogymnoascus sp. fungus. J Nat Prod 78:919–923. https://doi.org/10.1021/np500906k
Friker LL, Scheiblich H, Hochheiser IV et al (2020) β-Amyloid clustering around ASC fibrils boosts its toxicity in microglia. Cell Rep 30:3743-3754.e6. https://doi.org/10.1016/j.celrep.2020.02.025
Goedert M, Spillantini MG (2006) A century of Alzheimer’s disease. Science 314: 777–781. https://science.sciencemag.org/content/314/5800/777
Guo S, Feng B, Zhu R et al (2011) Preparative isolation of three anthraquinones from Rumex japonicus by high-speed counter-current chromatography. Molecules 16:1201–1210. https://doi.org/10.3390/molecules16021201
Gupta S, Chaturvedi P, Kulkarni MG et al (2019) A critical review on exploiting the pharmaceutical potential of plant endophytic fungi. Biotechnol Adv 39:107462. https://doi.org/10.1016/j.biotechadv.2019.107462
Hargreaves J, Park JO, Ghisalberti EL et al (2002) New chlorinated diphenyl ethers from an Aspergillus species. J Nat Prod 65:7–10. https://doi.org/10.1021/np0102758
Huang X, Sun X, Ding B et al (2013) A new anti-acetylcholinesterase α -pyrone meroterpene, arigsugacin I, from mangrove endophytic fungus Penicillium sp. sk5GW1L of Kandelia candel. Planta Med 79:1572–1575
Jalili-Baleh L, Babaei E, Abdpour S et al (2018) A review on flavonoid-based scaffolds as multi-target-directed ligands (MTDLs) for Alzheimer’s disease. Eur J Med Chem 152:570–589. https://doi.org/10.1016/j.ejmech.2018.05.004
Jayasuriya H, Ball RG, Zink DL et al (1995) Barceloneic acid A, a new farnesyl-protein transferase inhibitor from a Phoma species. J Nat Prod 58:986–991. https://doi.org/10.1021/np50121a002
Kim W, Song N, Yoo CD (2001) Quinolactacins Al and A2, new acetylcholinesterase inhibitors from Penicillium citrinum in MeOH was further purified by reverse phase HPLC. J Antibiot (tokyo) 54:831–835
Kingston DG (2011) Modern natural products drug discovery and its relevance to biodiversity conservation. J Nat Prod 74:496–511. https://doi.org/10.1021/np100550t
Kou X, Liu J, Chen Y et al (2021) Emodin derivatives with multi-factor anti-AD activities: AChE inhibitor, anti-oxidant and metal chelator. J Mol Struct 1239:130459. https://doi.org/10.1016/j.molstruc.2021.130459
Kryger G, Harel M, Giles K et al (2000) Structures of recombinant native and E202Q mutant human acetylcholinesterase complexed with the snake-venom toxin fasciculin-II. Acta Crystallogr D Biol Crystallogr 56:1385–1394. https://doi.org/10.1107/S0907444900010659
Kumar S, Bhardwaj VK, Singh R et al (2021) Explicit-solvent molecular dynamics simulations revealed conformational regain and aggregation inhibition of I113T SOD1 by Himalayan bioactive molecules. J Mol Liq 339:116798. https://doi.org/10.1016/j.molliq.2021.116798
Lai Z, Wang D, Wang Y et al (2014) Molecular identification of endophytic fungi with inhibitory activityagainst acetylcholinesterase from Huperzia serrata. Mycosystema 33: 858–866. http://journals.im.ac.cn/jwxten/ch/reader/view_abstract.aspx?file_no=jw14040858&flag=1
Li Y, Sun B, Liu S et al (2008) Bioactive asterric acid derivatives from the antarctic ascomycete fungus Geomyces sp. J Nat Prod 71:1643–1646. https://doi.org/10.1021/np8003003
Li JL, Huang L, Liu J et al (2015) Acetylcholinesterase inhibitory dimeric indole derivatives from the marine actinomycetes Rubrobacter radiotolerans. Fitoterapia 102:203–207. https://doi.org/10.1016/j.fitote.2015.01.014
Li HT, Tang L, Liu T et al (2019) Polyoxygenated meroterpenoids and a bioactive illudalane derivative from a co-culture of Armillaria sp. and Epicoccum sp. Org Chem Front 6:3847–3853. https://doi.org/10.1039/C9QO01087D
Lima GS, Rocha AM, Santos GF et al (2018) Metabolic response of Aspergillus sydowii to OSMAC modulation produces acetylcholinesterase inhibitors. Phytochem Lett 24:39–45. https://doi.org/10.1016/j.phytol.2018.01.007
Lin T, Lu CH, Shen YM (2009) Secondary metabolites of Aspergillus sp. F1, a commensal fungal strain of Trewia nudiflora. Nat Prod Res 23:77–85. https://doi.org/10.1080/14786410701852826
Liu R, Zhu W, Zhang Y et al (2006) A new diphenyl ether from marine-derived fungus Aspergillus sp. B-F-2. J Antibiot 59:362–365. https://doi.org/10.1038/ja.2006.52
Liu L, Li Y, Liu S et al (2009) Chloropestolide A, an antitumor metabolite with an unprecedented spiroketal skeleton from Pestalotiopsis fici. Org Lett 11:2836–2839. https://doi.org/10.1021/ol901039m
Liu D, Yan L, Ma L et al (2015) Diphenyl derivatives from coastal saline soil fungus Aspergillus iizukae. Arch Pharm Res 38:1038–1043. https://doi.org/10.1007/s12272-014-0371-z
Liu WC, Gong T, Zhu P (2016) Advances in exploring alternative taxol sources. RSC Adv 6:48800–48809. https://doi.org/10.1039/C6RA06640B
Liu CM, Wang YL, Wang X et al (2021) Active metabolites from the fungus Pestalotiopsis sp. ymf1.0474. Chem Nat Comp 57:1–3
Long Y, Cui H, Liu X et al (2017) Acetylcholinesterase inhibitory meroterpenoid from a mangrove endophytic fungus Aspergillus sp. 16–5c. Molecules 22:727. https://doi.org/10.3390/molecules22050727
Ma X, Tan C, Zhu D et al (2007) Hup A from Huperzia species—an ethnopharmacological review. J Ethnopharmacol 113:15–34. https://doi.org/10.1016/j.jep.2007.05.030
Mahmoodian A, Stickings CE (1964) Studies in the biochemistry of micro-organisms. 115. Metabolites of Penicillium frequentans westling: isolated of sulochrin, asterric acid, (+)- bisdechlorogeodin and two new substituted anthraquinones, questin and questinol. Biochem J 92:369–378. https://doi.org/10.1042/bj0920369
Meng FC, Mao F, Shan WJ et al (2012a) Design, synthesis, and evaluation of indanone derivatives as acetylcholinesterase inhibitors and metal-chelating agents. Bioorg Med Chem Lett 22:4462–4466. https://doi.org/10.1016/j.bmcl.2012.04.029
Meng X, Mao Z, Lou J et al (2012b) Benzopyranones from the endophytic fungus Hyalodendriella sp. Ponipodef12 and their bioactivities. Molecules 17:11303–11314. https://doi.org/10.3390/molecules171011303
Miles JA, Ross BP (2021) Recent advances in virtual screening for cholinesterase inhibitors. ACS Chem Neurosci 12:30–41. https://doi.org/10.1021/acschemneuro.0c00627
Mohammadi-Khanaposhtani M, Saeedi M, Zafarghandi NS et al (2015) Potential acetylcholinesterase inhibitors: design, synthesis, biological evaluation, and docking study of acridone linked to 1, 2, 3-triazol derivatives. Eur J Med Chem 92:799–806. https://doi.org/10.1016/j.ejmech.2015.01.044
Natori S, Nishikawa H (1962) Structures of osoic acids and related compounds, metabolites of Oospora sulphurea-ochracea v. BEYMA Chem Pharm Bull 10:117–124. https://doi.org/10.1248/cpb.10.117
Ogawa T, Ando K, Aotani Y et al (1995) RES-1214-1 and 2, novel non-peptidic endothelin type a receptor antagonists produced by Pestalotiopsis sp. J Antibiot (tokyo) 48:1401–1406. https://doi.org/10.1002/chin.199623286
Oh JM, Kang MG, Hong A et al (2019) Potent and selective inhibition of human monoamine oxidase-B by 4-dimethylaminochalcone and selected chalcone derivatives. Int J Biol Macromol 137:426–432. https://doi.org/10.1016/j.ijbiomac.2019.06.167
Ohashi H, Akiyama H, Nishikori K et al (1992) Asterric acid, a new endothelin binding inhibitor. J Antibiot 45:1684–1685. https://doi.org/10.7164/antibiotics.45.1684
Omura S, Kuno F, Otoguro K et al (1995) Arisugacin, a novel and selective inhibitor of acetylcholinesterase from Penicillium sp. FO-4259. J Antibiot 4:745–746
Pan H, Zhang J, Wang Y et al (2019) Linarin improves the dyskinesia recovery in Alzheimer’s disease zebrafish by inhibiting the acetylcholinesterase activity. Life Sci 222:112–116. https://doi.org/10.1016/j.lfs.2019.02.046
Prince PM, Wimo A, Guerchet M et al (2015) The global impact of dementia: word Alzheimer report 2015. Washington: ADI. https://www.alzint.org/resource/world-alzheimer-report-2015/
Santos GF, Lima GS, Oliveira GP et al (2018) New AChE inhibitors from microbial transformation of trachyloban-19-oic acid by Syncephalastrum racemosum. Bioorg Chem 79:60–63. https://doi.org/10.1016/j.bioorg.2018.04.011
Sekhar Rao KC, Divakar S, Karanth NG et al (2001) (2’, 3’, 5’ -Trihydroxyphenyl) tetradecan-2-ol, a novel acetylcholinesterase inhibitor from Chrysosporium sp. J Antibiot (tokyo) 54:848–849
Shaikh S, Zainab T, Shakil S et al (2015) A neuroinformatics study to compare inhibition efficiency of three natural ligands (Fawcettimine, Cernuine and Lycodine) against human brain acetylcholinesterase. Netw-Comp Neur 26:25–34. https://doi.org/10.3109/0954898X.2014.994145
Singh R, Bhardwaj VK, Sharma J et al (2020) Discovery and in silico evaluation of aminoarylbenzosuberene molecules as novel checkpoint kinase 1 inhibitor determinants. Genomics 113:707–715. https://doi.org/10.1016/j.ygeno.2020.10.001
Singh R, Bhardwaj VK, Sharma J et al (2021) Identification of selective cyclin-dependent kinase 2 inhibitor from the library of pyrrolone-fused benzosuberene compounds: an in silico exploration. J Biomol Struct Dyn 5:1–9. https://doi.org/10.1080/07391102.2021.1900918
Sonmez F, Zengin Kurt B, Gazioglu I et al (2017) Design, synthesis and docking study of novel coumarin ligands as potential selective acetylcholinesterase inhibitors. J Enzyme Inhib Med Chem 32:285–297. https://doi.org/10.1080/14756366.2016.1250753
Stermits FR, Schroeder HA, Geigert J (1973) Asterric acid from Scytalidium. Phytochemistry 12:1173. https://doi.org/10.1016/0031-9422(73)85038-1
Su J, Liu H, Guo K et al (2017) Research advances and detection methodologies for microbe-derived acetylcholinesterase inhibitors: a systemic review. Molecules 22:176. https://doi.org/10.3390/molecules22010176
Tang H, Zhen Y, Lao X et al (2007) Research progress in receptor antagonists from microorganisms. World Notes Antibiot 28: 19–24. http://en.cnki.com.cn/Article_en/CJFDTOTAL-GYKS200701004.htm
Teles APC, Takahashi JA (2013) Paecilomide, a new acetylcholinesterase inhibitor from Paecilomyces lilacinus. Microbiol Res 168:204–210. https://doi.org/10.1016/j.micres.2012.11.007
Vig R, Bhadra F, Gupta SK et al (2021) Neuroprotective effects of quercetin produced by an endophytic fungus Nigrospora oryzae isolated from Tinospora cordifolia. J Appl Microbiol. https://doi.org/10.1111/jam.15174
Wang FW, Ye YH, Chen JR et al (2006) Neoplaether, a new cytotoxic and antifungal endophyte metabolite from Neoplaconema napellum IFB-E016. FEMS Microbiol Lett 261:218–223. https://doi.org/10.1111/j.1574-6968.2006.00358.x
Wang M, Sun M, Hao H et al (2015) Avertoxins A-D, prenyl asteltoxin derivatives from Aspergillus versicolor Y10, an endophytic fungus of Huperzia serrata. J Nat Prod. https://doi.org/10.1021/acs.jnatprod.5b00600
Wang C, Guo L, Hao J et al (2016a) α-glucosidase inhibitors from the marine-derived fungus Aspergillus flavipes HN4-13. J Nat Prod 79:2977–2981. https://doi.org/10.1021/acs.jnatprod.6b00766
Wang Y, Lai Z, Li XX et al (2016b) Isolation, diversity and acetylcholinesterase inhibitory activity of the culturable endophytic fungi harboured in Huperzia serrata from Jinggang Mountain, China. World J Microbiol Biotechnol 32:20. https://doi.org/10.1007/s11274-015-1966-3
Wu B, Ohlendorf B, Oesker V et al (2014) Acetylcholinesterase inhibitors from a marine fungus Talaromyces sp. strain LF458. Mar Biotechnol 17:110–119. https://doi.org/10.1007/s10126-014-9599-3
Xu K, Zhou Q, Li XQ et al (2020) Cadinane- and drimane-type sesquiterpenoids produced by Paecilomyces sp. TE-540, an endophyte from Nicotiana tabacum L., are acetylcholinesterase inhibitors. Bioorg Chem 104:104252. https://doi.org/10.1016/j.bioorg.2020.104252
Yang ZD, Zhang XD, Yang X et al (2021) A norbisabolane and an arabitol benzoate from Talaromyces marneffei, an endophytic fungus of Epilobium angustifolium. Fitoterapia 153:104948
Zaki AG, El-Sayed ER, Abd Elkodous M et al (2020) Microbial acetylcholinesterase inhibitors for Alzheimer’s therapy: recent trends on extraction, detection, irradiation-assisted production improvement and nano-structured drug delivery. Appl Microbiol Biotechnol 104:4717–4735. https://doi.org/10.1007/s00253-020-10560-9
Zhang D, Yang Y, Castlebury LA et al (1996) A method for the large scale isolation of high transformation effificiency fungal genomic DNA. FEMS Microbiol Lett 145:261–265. https://doi.org/10.1016/S0378-1097(96)00421-1
Zhang ZB, Zeng QG, Yan RM et al (2011) Endophytic fungus Cladosporium cladosporioides LF70 from Huperzia serrata produces Huperzine A. World J Microbiol Biotechnol 27:479–486. https://doi.org/10.1007/s11274-010-0476-6
Zhang LH, Feng BM, Zhao YQ et al (2016) Polyketide butenolide, diphenyl ether, and benzophenone derivatives from the fungus Aspergillus flavipes PJ03-11. Bioorg Med Chem Lett 26:346–350. https://doi.org/10.1016/j.bmcl.2015.12.009
Acknowledgements
This study was supported by Natural Science Foundation of China (81760649), the Natural Science Foundation of Jiangxi Province of China (20181BAB215044), and Funds of Jiangxi Science and Technology Normal University (2017XJZD004).
Author information
Authors and Affiliations
Contributions
YWX designed and performed all experiments under the supervision of DZ, and developed the manuscript draft. WZL and DL supervised studies on the experiments on identification of endophytic fungi, isolation and purification of compound. ZBZ and JC analyzed the experimental data and discussed the results with the coauthors and revised this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xiao, Y., Liang, W., Liu, D. et al. Isolation and acetylcholinesterase inhibitory activity of asterric acid derivatives produced by Talaromyces aurantiacus FL15, an endophytic fungus from Huperzia serrata. 3 Biotech 12, 60 (2022). https://doi.org/10.1007/s13205-022-03125-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03125-2