Abstract
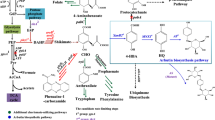
2-chloronicotinic acid (2-CA) is a key precursor for the synthesis of a series of pesticides and pharmaceuticals. Nitrilase-catalyzed bioprocess is a promising method for 2-CA production from 2-chloronicotinonitrile (2-CN). In this study, a mutant of nitrilase from Rhodococcus zopfii (RzNIT/W167G) was constitutively overexpressed with Escherichia coli as host, which exhibited a onefold increase in enzymatic activity compared with inducible expression. Biosynthesis of 2-CA using whole cells harboring nitrilase as biocatalysts were investigated and 318.5 mM 2-CA was produced, which was the highest level for 2-CA production catalyzed by nitrilase to date. 2-CA was recovered from the reaction mixture through a simple acidification step with a recovery yield of 90%. This study developed an efficient bioprocess for 2-CA with great potential for industrial application.







Similar content being viewed by others
References
Chen Z, Wang H, Yang L et al (2021) Significantly enhancing the stereoselectivity of a regioselective nitrilase for the production of (S)-3-cyano-5-methylhexanoic acid using an MM/PBSA method. Chem Commun 57:931–934. https://doi.org/10.1039/d0cc07106d
Du XH, Ge LD, Chen SY, Zhao JB, ZHong ZK, Wang JL, Xu ZY (2009) Patent. CN200910152436.1
Gong JS, Dong TT, Gu BC et al (2017) Semirational engineering accelerates the laboratory evolution of nitrilase catalytic efficiency for nicotinic acid biosynthesis. ChemCatChem 9:3395–3401. https://doi.org/10.1002/cctc.201700665
Gong JS, Zhang Q, Gu BC et al (2018) Efficient biocatalytic synthesis of nicotinic acid by recombinant nitrilase via high density culture. Bioresour Technol 260:427–431. https://doi.org/10.1016/j.biortech.2018.03.109
Hauer B (2020) Embracing nature’s catalysts: a viewpoint on the future of biocatalysis. ACS Catal 10:8418–8427. https://doi.org/10.1021/acscatal.0c01708
Jin LQ, Li YF, Liu ZQ et al (2011) Characterization of a newly isolated strain Rhodococcus erythropolis ZJB-09149 transforming 2-chloro-3-cyanopyridine to 2-chloronicotinic acid. N Biotechnol 28:610–615. https://doi.org/10.1016/j.nbt.2011.04.004
Lauder K, Anselmi S, Finnigan JD et al (2020) Enantioselective synthesis of α-thiocarboxylic acids by nitrilase biocatalysed dynamic kinetic resolution of α-thionitriles. Chem - A Eur J 26:10422–10426. https://doi.org/10.1002/chem.202001108
Liu JF, Zhang ZJ, Li AT et al (2011) Significantly enhanced production of recombinant nitrilase by optimization of culture conditions and glycerol feeding. Appl Microbiol Biotechnol 89:665–672. https://doi.org/10.1007/s00253-010-2866-y
Quevedo CE, Bavetsias V, McDonald E (2009) Microwave-assisted synthesis of 2-aminonicotinic acids by reacting 2-chloronicotinic acid with amines. Tetrahedron Lett 50:2481–2483. https://doi.org/10.1016/j.tetlet.2009.03.034
Sharma NN, Sharma M, Bhalla TC (2011) An improved nitrilase-mediated bioprocess for synthesis of nicotinic acid from 3-cyanopyridine with hyperinduced Nocardia globerula NHB-2. J Ind Microbiol Biotechnol 38:1235–1243. https://doi.org/10.1007/s10295-010-0902-7
Shen Q, Yu Z, Lv P et al (2020) Engineering a Pichia pastoris nitrilase whole cell catalyst through the increased nitrilase gene copy number and co-expressing of ER oxidoreductin 1. Appl Microbiol Biotechnol 104:2489–2500. https://doi.org/10.1007/s00253-020-10422-4
Shen JD, Cai X, Liu ZQ, Zheng YG (2021) Nitrilase: a promising biocatalyst in industrial applications for green chemistry. Crit Rev Biotechnol 41:72–93. https://doi.org/10.1080/07388551.2020.1827367
Tang XL, Jin JQ, Wu ZM et al (2018) Structure-based engineering of amidase from Pantoea sp. for efficient 2-chloronicotinic acid biosynthesis. Appl Environ Microbiol 85:e02471-e2518. https://doi.org/10.1128/AEM.02471-18
Teepakorn C, Zajkoska P, Cwicklinski G et al (2021) Nitrilase immobilization and transposition from a micro-scale batch to a continuous process increase the nicotinic acid productivity. Biotechnol J 16:e2100010. https://doi.org/10.1002/biot.202100010
Vaughan PA, Knowles CJ, Cheetham PSJ (1989) Conversion of 3-cyanopyridine to nicotinic acid by Nocardia rhodochrous LL100-21. Enzyme Microb Technol 11:815–823. https://doi.org/10.1016/0141-0229(89)90055-0
Wu Y, Wu C, Yan S, Hu B (2019) Solubility determination of 2-chloronicotinic acid and analysis of solvent effect. J Chem Eng Data 64:5578–5583. https://doi.org/10.1021/acs.jced.9b00661
Wu ZM, Hao CL, Tong T et al (2020) Purification of (S)-3-cyano-5-methylhexanoic acid from bioconversion broth using an acetone/ammonium sulfate aqueous two-phase system. Process Biochem 89:186–192. https://doi.org/10.1016/j.procbio.2019.10.023
Wu S, Snajdrova R, Moore JC et al (2021) Biocatalysis: enzymatic synthesis for industrial applications. Angew Chemie - Int Ed 60:88–119
Xue YP, Shi CC, Xu Z et al (2015) Design of nitrilases with superior activity and enantioselectivity towards sterically hindered nitrile by protein engineering. Adv Synth Catal 357:1741–1750. https://doi.org/10.1002/adsc.201500039
Yu S, Yao P, Li J et al (2019) Improving the catalytic efficiency and stereoselectivity of a nitrilase from: Synechocystis sp. PCC6803 by semi-rational engineering en route to chiral γ-amino acids. Catal Sci Technol 9:1504–1510. https://doi.org/10.1039/c8cy02455c
Yu S, Li J, Yao P et al (2021) Inverting the enantiopreference of nitrilase-catalyzed desymmetric hydrolysis of prochiral dinitriles by reshaping the binding pocket with a mirror-image strategy. Angew Chemie - Int Ed 60:3679–3684. https://doi.org/10.1002/anie.202012243
Zhang Q, Lu XF, Zhang Y et al (2020a) Development of a robust nitrilase by fragment swapping and semi-rational design for efficient biosynthesis of pregabalin precursor. Biotechnol Bioeng 117:318–329. https://doi.org/10.1002/bit.27203
Zhang XH, Wang CY, Cai X et al (2020b) Upscale production of (R)-mandelic acid with a stereospecific nitrilase in an aqueous system. Bioprocess Biosyst Eng 43:1299–1307. https://doi.org/10.1007/s00449-020-02326-4
Zheng RC, Jin JQ, Wu ZM et al (2018) Biocatalytic hydrolysis of chlorinated nicotinamides by a superior AS family amidase and its application in enzymatic production of 2-chloronicotinic acid. Bioorg Chem 76:81–87. https://doi.org/10.1016/j.bioorg.2017.11.001
Zheng RC, Dai AD, Zheng, YG (2020) Patent CN112063607-A
Zhu XY, Gong JS, Li H et al (2014) Bench-scale biosynthesis of isonicotinic acid from 4-cyanopyridine by Pseudomonas putida. Chem Pap 68:739–744. https://doi.org/10.2478/s11696-013-0521-7
Zou S, Hua D, Jiang Z et al (2021) A integrated process for nitrilase-catalyzed asymmetric hydrolysis and easy biocatalyst recycling by introducing biocompatible biphasic system. Bioresour Technol. https://doi.org/10.1016/j.biortech.2020.124392
Acknowledgements
This work was supported by the National Key R&D Program of China (2017YFE0129400) and Natural Science Foundation of Zhejiang Province (No. LR19B060001).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dai, AD., Wu, ZM., Zheng, RC. et al. Constitutive expression of nitrilase from Rhodococcus zopfii for efficient biosynthesis of 2-chloronicotinic acid. 3 Biotech 12, 50 (2022). https://doi.org/10.1007/s13205-022-03119-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-022-03119-0