Abstract
In 2019, 229 million cases of malaria were recorded worldwide. For epidemiologic surveillance and proper treatment of persons infected with Plasmodium spp., rapid detection of infections by Plasmodium spp. is critical. Thus, Plasmodium spp. diagnosis is one of the indispensable measures for malaria control. Although microscopy is the gold standard for diagnosis, it has restrictions related mainly to the lack of qualified human resources, which is a problem in many regions. Thus, this review presents major innovations in diagnostic methods as alternatives to or complementary to microscopy. Detection platforms in lateral flow systems, electrochemical immunosensors, molecular biology and, more recently, those integrated with smartphones, are highlighted, among others. The advanced improvement of these tests aims to provide techniques that are sensitive and specific, but also quick, easy to handle and free from the laboratory environment. In this way, the tracking of malaria cases can become increasingly effective and contribute to controlling the disease.

Adapted from Rebould et al. (2019) and within the recommendations of the Creative Commons Attribution (CC BY) (http://creativecommons.org/licenses/by/4.0/)


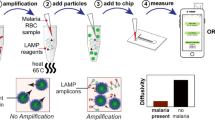
Adapted from Hemben et al. (Hemben et al. 2017) and within the recommendations of the Creative Commons Attribution (CC BY) (http://creativecommons.org/licenses/by/4.0/)

Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Ahmad A, Verma AK, Krishna S et al (2019) Plasmodium falciparum glutamate dehydrogenase is genetically conserved across eight malaria endemic states of India: exploring new avenues of malaria elimination. PLoS ONE 14:1–13. https://doi.org/10.1371/journal.pone.0218210
Amaral LC, Robortella DR, Guimarães LFF et al (2019) Ribosomal and non-ribosomal PCR targets for the detection of low-density and mixed malaria infections. Malar J 18:154. https://doi.org/10.1186/s12936-019-2781-3
Anfossi L, Di Nardo F, Profiti M et al (2018) A versatile and sensitive lateral flow immunoassay for the rapid diagnosis of visceral leishmaniasis. Anal Bioanal Chem 410:4123–4134. https://doi.org/10.1007/s00216-018-1067-x
Barber BE, William T, Grigg MJ et al (2013) Limitations of microscopy to differentiate Plasmodium species in a region co-endemic for Plasmodium falciparum, Plasmodium vivax and Plasmodium knowlesi. Malar J 12:1–6. https://doi.org/10.1186/1475-2875-12-8
Bauer WS, Richardson KA, Adams NM, et al (2017) Rapid concentration and elution of malarial antigen histidine-rich protein II using solid phase Zn(II) resin in a simple flow-through pipette tip format. Biomicrofluidics 11:. https://doi.org/10.1063/1.4984788
Berzosa P, De Lucio A, Romay-Barja M et al (2018) Comparison of three diagnostic methods (microscopy, RDT, and PCR) for the detection of malaria parasites in representative samples from Equatorial Guinea 11 Medical and Health Sciences 1108 Medical Microbiology. Malar J 17:1–12. https://doi.org/10.1186/s12936-018-2481-4
Birch CM, Hou HW, Han J, Niles JC (2015) Identification of malaria parasite- infected red blood cell surface aptamers by inertial microfluidic SELEX (I-SELEX). Nat Publ Gr 1–16. https://doi.org/10.1038/srep11347
BRASIL (2020) Guia de tratamento da malária no Brasil/Ministério da Saúde. Secretároa de Vigilância em Saúde. Departamento de imunização e Doenças Transmissíveis - Brasília; Ministério da Saúde, 2020., 1a. Brasília
Brasil P, Zalis MG, de Pina-Costa A et al (2017) Outbreak of human malaria caused by Plasmodium simium in the Atlantic Forest in Rio de Janeiro: a molecular epidemiological investigation. Lancet Glob Heal 5:e1038–e1046. https://doi.org/10.1016/S2214-109X(17)30333-9
CDC (2017) CDC—Malaria—About Malaria—History—Laveran and the Discovery of the Malaria Parasite. https://www.cdc.gov/malaria/about/history/laveran.html. Accessed 4 Sep 2021
CDC—Malaria—Diagnostic Tools (2020) CDC—Malaria—Diagnostic Tools. https://www.cdc.gov/malaria/diagnosis_treatment/diagnostic_tools.html. Accessed 23 Aug 2021
Cho SJ, Lee J, Lee HJ et al (2016) A novel malaria Pf/Pv ab rapid diagnostic test using a differential diagnostic marker identified by network biology. Int J Biol Sci 12:824–835. https://doi.org/10.7150/ijbs.14408
De Souza CM, Laube T, Yamanaka H et al (2011) Magneto immunoassays for Plasmodium falciparum histidine-rich protein 2 related to malaria based on magnetic nanoparticles. Anal Chem 83:5570–5577. https://doi.org/10.1021/ac200573s
Dutta G, Lillehoj PB (2018) Wash-free, label-free immunoassay for rapid electrochemical detection of PfHRP2 in whole blood samples. Sci Rep 8:17129. https://doi.org/10.1038/s41598-018-35471-8
Dutta G, Nagarajan S, Lapidus LJ, Lillehoj PB (2017) Enzyme-free electrochemical immunosensor based on methylene blue and the electro-oxidation of hydrazine on Pt nanoparticles. Biosens Bioelectron 92:372–377. https://doi.org/10.1016/j.bios.2016.10.094
Dzakah EE, Kang K, Ni C et al (2013) Plasmodium vivax aldolase-specific monoclonal antibodies and its application in clinical diagnosis of malaria infections in China. Malar J 12:1. https://doi.org/10.1186/1475-2875-12-199
Figueroa-Miranda G, Feng L, Shiu SC et al (2018) Aptamer-based electrochemical biosensor for highly sensitive and selective malaria detection with adjustable dynamic response range and reusability. Sensors Actuators B Chem 255:235–243. https://doi.org/10.1016/j.snb.2017.07.117
Fontecha G, Mejía RE, Banegas E et al (2018) Deletions of pfhrp2 and pfhrp3 genes of Plasmodium falciparum from Honduras, Guatemala and Nicaragua. Malar J 17:1–10. https://doi.org/10.1186/s12936-018-2470-7
Fuhad KMF, Tuba JF, Sarker MRA et al (2020) Deep learning based automatic malaria parasite detection from blood smear and its smartphone based application. Diagnostics 10:329. https://doi.org/10.3390/diagnostics10050329
Gopal NS, Raychaudhuri R (2017) Detection of Plasmodium aldolase using a smartphone and microfluidic enzyme linked immunosorbent assay. Malar Res Treat 2017:1–4. https://doi.org/10.1155/2017/9062514
Grignard L, Nolder D, Sepúlveda N et al (2020) A novel multiplex qPCR assay for detection of Plasmodium falciparum with histidine-rich protein 2 and 3 (pfhrp2 and pfhrp3) deletions in polyclonal infections. EBioMedicine 55:102757. https://doi.org/10.1016/j.ebiom.2020.102757
Hemben A, Ashley J, Tothill IE (2017) Development of an Immunosensor for Pf HRP 2 as a biomarker for malaria detection. Biosensors 7:28. https://doi.org/10.3390/bios7030028
Jaureguiberry G, Hatin I, D’Auriol L, Galibert G (1990) PCR detection of Plasmodium falciparum by oligonucleotide probes. Mol Cell Probes 4:409–414. https://doi.org/10.1016/0890-8508(90)90031-T
Kain KC, Brown AE, Mirabelli L, Webster HK (1993) Detection of Plasmodium vivax by polymerase chain reaction in a field study. J Infect Dis 168:1323–1326. https://doi.org/10.1093/infdis/168.5.1323
Kim J, Cao XE, Finkelstein JL et al (2019) A two-colour multiplexed lateral flow immunoassay system to differentially detect human malaria species on a single test line. Malar J 18:1–10. https://doi.org/10.1186/s12936-019-2957-x
Komaki-Yasuda K, Vincent JP, Nakatsu M et al (2018) A novel PCR-based system for the detection of four species of human malaria parasites and Plasmodium knowlesi. PLoS ONE 13:e0191886. https://doi.org/10.1371/journal.pone.0191886
Kori LD, Valecha N, Anvikar AR (2020) Glutamate dehydrogenase: a novel candidate to diagnose Plasmodium falciparum through rapid diagnostic test in blood specimen from fever patients. Sci Rep 10:6307. https://doi.org/10.1038/s41598-020-62850-x
Krampa FD, Aniweh Y, Kanyong P, Awandare GA (2020) Recent advances in the development of biosensors for malaria diagnosis. Sensors 20:799. https://doi.org/10.3390/s20030799
Kumar B, Bhalla V, Bhadoriya RPS, et al (2016) Label-free electrochemical detection of malaria-infected red blood cells. ROAYAL Soc Chem 1–3. https://doi.org/10.1039/C6RA07665C
Lee GC, Jeon ES, Le DT et al (2011) Development and evaluation of a rapid diagnostic test for Plasmodium falciparum, P. vivax, and mixed-species malaria antigens. Am J Trop Med Hyg 85:989–993. https://doi.org/10.4269/ajtmh.2011.11-0265
Low YK, Chan J, Soraya G V, et al (2019) Development of an Ultrasensitive Impedimetric Immunosensor Platform for Detection of Plasmodium Lactate Dehydrogenase. Sensors 19:. https://doi.org/10.3390/s19112446
Martinsen Ellen, Perkins Susan L (2013) The diversity of Plasmodium and other haemosporidians: The intersection of taxonomy, phylogenetics and genomics | Request PDF. Malar Parasites Comp Genomics, Evol Mol Biol 1–15
Mfuh KO, Achonduh-Atijegbe OA, Bekindaka ON et al (2019) A comparison of thick-film microscopy, rapid diagnostic test, and polymerase chain reaction for accurate diagnosis of Plasmodium falciparum malaria. Malar J 18:1–8. https://doi.org/10.1186/s12936-019-2711-4
Mohon AN, Getie S, Jahan N et al (2019) Ultrasensitive loop mediated isothermal amplification (US-LAMP) to detect malaria for elimination. Malar J 18:1–10. https://doi.org/10.1186/s12936-019-2979-4
Murphy SC, Prentice JL, Williamson K et al (2012) Real-time quantitative reverse transcription PCR for monitoring of blood-stage Plasmodium falciparum infections in malaria human challenge trials. Am J Trop Med Hyg 86:383–394. https://doi.org/10.4269/ajtmh.2012.10-0658
Paul A, Padmapriya P, Natarajan V (2017) ScienceDirect Diagnosis of malarial infection using change in properties of optically trapped red blood cells. Biomed J 40:101–105. https://doi.org/10.1016/j.bj.2016.10.001
Pirnstill CW, Coté GL (2015) Malaria diagnosis using a mobile phone polarized microscope. Sci Rep 5:13368. https://doi.org/10.1038/srep13368
Rampazzo RCP, Graziani AC, Leite KK et al (2019) Proof of concept for a portable platform for molecular diagnosis of tropical diseases: on-chip ready-to-use real-time quantitative pcr for detection of Trypanosoma cruzi or Plasmodium spp. J Mol Diagnostics 21:839–851. https://doi.org/10.1016/j.jmoldx.2019.04.008
Reboud J, Xu G, Garrett A et al (2019) Paper-based microfluidics for DNA diagnostics of malaria in low resource underserved rural communities. Proc Natl Acad Sci USA 116:4834–4842. https://doi.org/10.1073/pnas.1812296116
Ricks KM, Adams NM, Scherr TF et al (2016) Direct transfer of HRPII-magnetic bead complexes to malaria rapid diagnostic tests significantly improves test sensitivity. Malar J 15:1–8. https://doi.org/10.1186/s12936-016-1448-6
Rosado L, da Costa J, Elias D, Cardoso J (2017) Mobile-based analysis of malaria-infected thin blood smears: automated species and life cycle stage determination. Sensors 17:2167. https://doi.org/10.3390/s17102167
Rougemont M, Van Saanen M, Sahli R et al (2004) Detection of Four Plasmodium species in blood from humans by 18S rRNA gene subunit-based and species-specific real-time PCR assays. J Clin Microbiol 42:5636–5643. https://doi.org/10.1128/JCM.42.12.5636-5643.2004
Sanjay M, Singh NK, Ngashangva L, Goswami P (2020) A smartphone-based fiber-optic aptasensor for label-free detection of Plasmodium falciparum glutamate dehydrogenase. Anal Methods 12:1333–1341. https://doi.org/10.1039/C9AY02406A
Sattabongkot J, Tsuboi T, Han E-T et al (2014) Loop-mediated isothermal amplification assay for rapid diagnosis of Malaria infections in an area of endemicity in Thailand. J Clin Microbiol 52:1471–1477. https://doi.org/10.1128/JCM.03313-13
Sazed SA, Kibria MG, Alam MS (2021) An optimized real-time qPCR method for the effective detection of human malaria infections. Diagnostics 11:736. https://doi.org/10.3390/diagnostics11050736
Scherr TF, Gupta S, Wright DW, Haselton FR (2016) Mobile phone imaging and cloud-based analysis for standardized malaria detection and reporting. Sci Rep 6:28645. https://doi.org/10.1038/srep28645
Selvarajah D, Naing C, Htet NH, Mak JW (2020) Loop-mediated isothermal amplification (LAMP) test for diagnosis of uncomplicated malaria in endemic areas: a meta-analysis of diagnostic test accuracy. Malar J 19:211. https://doi.org/10.1186/s12936-020-03283-9
Sharma MK, Rao VK, Agarwal GS et al (2008) Highly sensitive Amperometric Immunosensor for detection of Plasmodium falciparum histidine-rich protein 2 in serum of humans with Malaria: comparison with a commercial kit. J Clin Microbiol 46:3759–3765. https://doi.org/10.1128/JCM.01022-08
Singh NK, Arya SK, Estrela P, Goswami P (2018) Capacitive malaria aptasensor using Plasmodium falciparum glutamate dehydrogenase as target antigen in undiluted human serum. Biosens Bioelectron 117:246–252. https://doi.org/10.1016/j.bios.2018.06.022
Stemple CC, Angus SV, Park TS, Yoon J (2014) Smartphone-based optofluidic lab-on-a-chip for detecting pathogens from blood. J Lab Autom 19:35–41. https://doi.org/10.1177/2211068213498241
Tran TV, Nguyen BV, Nguyen TTP et al (2019) Development of a highly sensitive magneto-enzyme lateral flow immunoassay for dengue NS1 detection. PeerJ 2019:1–18. https://doi.org/10.7717/peerj.7779
Wanja EW, Kuya N, Moranga C et al (2016) Field evaluation of diagnostic performance of malaria rapid diagnostic tests in western Kenya. Malar J 15:1–10. https://doi.org/10.1186/s12936-016-1508-y
Wataya Y, Arai M, Kubochi F et al (1993) DNA diagnosis of falciparum malaria using a double PCR technique: a field trial in the Solomon Islands. Mol Biochem Parasitol 58:165–167. https://doi.org/10.1016/0166-6851(93)90101-3
WHO (2019) World Malaria Report. https://www.who.int/malaria
WHO (2018a) WHO | The World malaria report 2018
WHO (2000) New perspectives: malaria diagnosis: report of a joint WHO/USAID INFORMAL CONSULTATION
WHO (2020) Global Malaria Programme. https://www.who.int/teams/global-malaria-programme/case-management/diagnosis/nucleic-acid-amplification-based-diagnostics. Accessed 26 Aug 2021
WHO (2018) Malaria rapid diagnostic test performance: results of WHO product testing of malaria RDTs: round 8 (2016–2018). World Health Organization, Geneva, p 2018
WHO (2021) Global technical strategy for malaria 2016–2030, 2021 update. Geneva
Acknowledgements
We thank Amy Grabner for the English edition of the manuscript.
Funding
The authors express their gratitude to Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado de Rondônia (FAPERO), Instituto Nacional de Ciência e Tecnologia, Epidemiologia da Amazônia Ocidental (INCT-EpiAmO) and Fundação Oswaldo Cruz (Fiocruz) for the financial support.
Author information
Authors and Affiliations
Contributions
NBdS: conceptualization, data curation, formal analysis, writing—original draft. LdNM: conceptualization, investigation, methodology. RMdOS: data curation. Sd SP: data curation, writing - review & editing. CBGT: data curation, writing—review & editing, visualization.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Rights and permissions
About this article
Cite this article
Brilhante-da-Silva, N., do Nascimento Martinez, L., de Oliveira Sousa, R.M. et al. Innovations in Plasmodium spp. diagnosis on diverse detection platforms. 3 Biotech 11, 505 (2021). https://doi.org/10.1007/s13205-021-03054-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-03054-6