Abstract
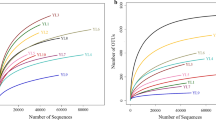
The current study aimed to investigate the association of microbial characteristics in the stomach of Sinanodonta woodiana (S. woodiana) and its cultured soil. S. woodiana stomach and its cultured soil were collected in winter 2019. The V3 + V4 regions of the 16S rRNA gene were sequenced to detect microbial diversity. The differences in the dominant species between S. woodiana and soil were also discussed. The results showed that the diversity and richness indices in S. woodiana were significantly higher than those in cultured soil (P < 0.01). At the phylum level, the dominant bacterial community compositions in the two groups were similar and included Proteobacteria, Firmicutes, Bacteroidetes, Cyanobacteria, Fusobacteria, and Acidobacteria. However, the abundance of these phyla was significantly different between the two groups (P < 0.05 or P < 0.01), and Proteobacteria was the most abundant phylum. At the genus level, a total of 1001 genera were identified in the two groups, of which 890 genera were detected in the cultured soil and 549 were detected in the S. woodiana stomach; 452 were unique to the cultured soil, and 111 were unique to the S. woodiana stomach. This result indicated great differences in the bacterial genera between the two groups. Moreover, 17 out of the 20 dominant bacterial genera exhibited statistically significant differences between the two groups (P < 0.05 or P < 0.01). Cupriavidus was the absolute dominant genus in the stomach of S. woodiana, followed by Sphingomonas and Burkholderia. The microorganisms in the stomach of S. woodiana formed a certain inherent bacterial system and were affected by the environment, which is beneficial to nutrition and health. In conclusion, the bacterial compositions were mostly the same between the stomach of S. woodiana and cultured soil, but the relative abundances of the bacterial communities were different. This study will greatly enhance the understanding of the microbial characteristics between the stomach of S. woodiana and the cultured environment, and provide guidance for healthy aquaculture in freshwater.






Similar content being viewed by others
References
Aceves AK, Johnson P, Bullard SA, Lafrentz S, Arias CR (2018) Description and characterization of the digestive gland microbiome in the freshwater mussel Villosa nebulosa (Bivalvia: Unionidae). J Mollus Stud 84(3):240–246. https://doi.org/10.1093/mollus/eyy014
Blaxter M, Mann J, Chapman T, Thomas F, Whitton C, Floyd R, Abebe E (2005) Defining operational taxonomic units using DNA barcode data. Philos Trans R Soc Lond B Biol Sci 360(1462):1935–1943. https://doi.org/10.1098/rstb.2005.1725
Burgos-Aceves MA, Cohen A, Smith Y, Faggio C (2018) MicroRNAs and their role on fish oxidative stress during xenobiotic environmental exposures. Ecotoxicol Environ Saf 148:995–1000. https://doi.org/10.1016/j.ecoenv.2017.12.001
Candela M, Biagi E, Maccaferri S, Turroni S, Brigidi P (2012) Intestinal microbiota is a plastic factor responding to environmental changes. Trends Microbiol 20(8):385–391. https://doi.org/10.1016/j.tim.2012.05.003
Clarke G, Stilling RM, Kennedy PJ, Stanton C, Cryan JF, Dinan TG (2014) Minireview: gut microbiota: the neglected endocrine organ. Mol Endocrinol 28(8):122–1238. https://doi.org/10.1210/me.2014-1108
Cole JR, Wang Q, Cardenas E, Fish J, Chai B, Farris RJ, Kulam-Syed-Mohideen AS, McGarrell DM, Marsh T, Garrity GM, Tiedje JM (2009) The ribosomal database project: improved alignments and new tools for rRNA analysis. Nucleic Acids Res 37(database issue):141–145. https://doi.org/10.1093/nar/gkn879
Del’Duca A, Cesar DE, Abreu PC (2015) Bacterial community of pond’s water, sediment and in the guts of tilapia (Oreochromis niloticus) juveniles characterized by fluorescentin situhybridization technique. Aquac Res 46(3):707–715. https://doi.org/10.1111/are.12218
DeSantis TZ, Hugenholtz P, Larsen N, Rojas M, Brodie EL, Keller K, Huber T, Dalevi D, Hu P, Andersen GL (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72(7):5069–5072. https://doi.org/10.1128/AEM.03006-05
Fan LF, Wang ZL, Chen MS, Qu YX, Li JY, Zhou AG, Xie SL, Zeng F, Zou JX (2019) Microbiota comparison of Pacific white shrimp intestine and sediment at freshwater and marine cultured environment. Sci Total Environ 657:1194–1204. https://doi.org/10.1016/j.scitotenv.2018.12.069
Forouhar Vajargah M, Mohamadi Yalsuyi A, Hedayati A, Faggio C (2018) Histopathological lesions and toxicity in common carp (Cyprinus carpio L. 1758) induced by copper nanoparticles. Microsc Res Tech 81(7):724–729. https://doi.org/10.1002/jemt.23028
Gou M, Qu YY, Yang H, Zhou JT, Li A, Guan XY, Ai FF (2008) Sphingomomas sp.: a novel microbial resource for biodegradation of aromatic compounds. Chin J Appl Environ Biol 14(2):276–282. https://doi.org/10.3321/j.issn:1006-687X.2008.02.027
Guzzetti E, Sureda A, Tejada S, Faggio C (2018) Microplastic in marine organism: environmental and toxicological effects. Environ Toxicol Pharmacol 64:164–171. https://doi.org/10.1016/j.etap.2018.10.009
Huang RL, Zhang N, Sun B, Liang YT. (2019) Community structure of Burkholderiales and its diversity in typical maize rhizosphere soil. Acta Pedological Sinica. https://kns.cnki.net/kcms/detail/32.1119.P.20190509.0901.002.html. https://doi.org/10.11766/trxb201901040008.
Kang Y. (2019) Study on mechanisms of enhanced pollutants removal in constructed wetland added with typical benthic fauna. Dissertation, Shandong University.
Klase G, Lee S, Liang S, Kim J, Zo YG, Lee J (2019) The microbiome and antibiotic resistance in integrated fish farm water: implications of environmental public health. Sci Total Environ 649:1491–1501. https://doi.org/10.1016/j.scitotenv.2018.08.288
Ley RE, Turnbaugh PJ, Klein S, Gordon JI (2006) Microbial ecology: human gut microbes associated with obesity. Nature 444(7122):1022–1023. https://doi.org/10.1038/4441022a
Li YK (2010) Histology of stomach of Anodonta woodiana elliptica and effects of heavy metal copper on their structures. Ecological Sci 29(5):461–466. https://doi.org/10.3969/j.issn.1008-8873.2010.05.011
Li RD, Li J, Zeng A, Cai GX, Tan ZJ (2012) The dynamic change of microbes in stomach by ultra-micro Wuji pills in mice. Chin J Microecol 24(8):711–714. https://doi.org/10.13381/j.cnki.cjm.2012.08.014
Lopes-Lima M, Burlakova LE, Karatayev AY, Mehler K, Seddon M, Sousa R (2018) Conservation of freshwater bivalves at the global scale: diversity, threats and research needs. Hydrobiologia 810(1):1–14. https://doi.org/10.1007/s10750-017-3486-7
Lozupone C, Knight R (2005) UniFrac: a new phylogenetic method for comparing microbial communities. Appl Environ Microbiol 71(12):8228–8235. https://doi.org/10.1128/AEM.71.12.8228-8235.2005
Meng XL, Li WJ, Nie GX (2019) Effect of different factors on the fish intestinal microbiota. J Fisheries China 43(1):143–155. https://doi.org/10.11964/jfc.20181011476
Ramette A (2007) Multivariate analyses in microbial ecology. FEMS Microbiol Ecol 62(2):142–160. https://doi.org/10.1111/j.1574-6941.2007.00375.x
Schloss PD, Gevers D, Westcott SL (2011) Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA-based studies. PLoS ONE 6(12):e27310. https://doi.org/10.1371/journal.pone.0027310
Semova I, Carten JD, Stombaugh J, Mackey LC, Knight R, Farber SA, Rawls JF (2012) Microbiota regulate intestinal absorption and metabolism of fatty acids in the zebrafish. Cell Host Microbe 12(3):277–288. https://doi.org/10.1016/j.chom.2012.08.003
Shu D, He Y, Yue H, Wang Q (2015) Microbial structures and community functions of anaerobic sludge in six full-scale wastewater treatment plants as revealed by 454 high-throughput pyrosequencing. Bioresour Technol 186:163–172. https://doi.org/10.1016/j.biortech.2015.03.072
Sousa R, Pilotto F, Aldridge DC (2011) Fouling of European freshwater bivalves (Unionidae) by the invasive zebra mussel (Dreissena polymorpha). Freshwater Biol 56(5):867–876. https://doi.org/10.1111/j.1365-2427.2010.02532.x
Sun ZY. (2017) Screening and degradation characteristics of a heterotrophic nitrification-aerobic denitrification bacterium cupriavidus sp.S1. Dissertation, Taiyuan University of Technology.
Sun WW, Liu XJ, Wu RW, Wang WK, Wu YL, Ouyang S, Wu XP (2019) Declining freshwater mussel diversity in the middle and lower reaches of the Xin River Basin: threat and conservation. Ecol Evol 9(24):14142–14153. https://doi.org/10.1002/ece3.5849
Vaughn CC (2012) Life history traits and abundance can predict local colonisation and extinction rates of freshwater mussels. Freshwater Biol 57(5):982–992. https://doi.org/10.1111/j.1365-2427.2012.02759.x
Vaughn CC (2017) Ecosystem services provided by freshwater mussels. Hydrobiologia 810(1):15–27. https://doi.org/10.1007/s10750-017-3139-x
Wang CZ, Lin GR, Yan T, Zheng ZP, Chen B, Sun FL (2014) The cellular community in the intestine of the shrimp Penaeus penicillatus and its culture environments. Fisheries Sci 80(5):1001–1007. https://doi.org/10.1007/s12562-014-0765-3
Weingarten EA, Atkinson CL, Jackson CR (2019) The gut microbiome of freshwater Unionidae mussels is determined by host species and is selectively retained from filtered seston. PLoS ONE 14(11):e0224796. https://doi.org/10.1371/journal.pone.0224796
Wu H, Zhou SN, Guo C, Tan ZJ, Cai GX, Zeng A, Zhang HL (2012) A metagenome DNA extracting method of intestinal flora in mice for molecular diversity analysis based on PCR technology. Chin J Microecol 24(7):648–651. https://doi.org/10.13381/j.cnki.cjm.2012.07.003
Yin JX, Chen Y, Zhang CL (2004) Dynamic study on the population of microbiota in Hyriopsis cumingiiea. Reserv Fish 24(3):40–42. https://doi.org/10.15928/j.1674-3075.2004.03.016
Yin JX, Chen Y, Shao JZ, Liu XZ, Shou YY (2006) Study on the intestinal microflora of Hyriopsis cumingii. J Zhejiang Univ (Sci Edn) 33(2):211–215. https://doi.org/10.3321/j.issn:1008-9497.2006.02.022
Zhang J, Yang Y, Zhao L, Li Y, Xie S, Liu Y (2015) Distribution of sediment bacterial and archaeal communities in plateau freshwater lakes. Appl Microbiol Biotechnol 99(7):3291–3302. https://doi.org/10.1007/s00253-014-6262-x
Zhu TT, Tian CK (2018) Analysis on microbial diversity in the sediments and its relationship with environmental factors in a reservoir. Acta Scientiarum Naturalium Universitaties Pekinensis 54(3):625–632. https://doi.org/10.13209/j.0479-8023.2017.163
Acknowledgements
This work was supported by the Scientific Research Fund of Hunan Provincial Education Department (19A256).
Author information
Authors and Affiliations
Contributions
CX designed the study; CL analyzed the data; KT collected data and wrote the manuscript; CL and CX checked the paper. The decision to submit the manuscript for publication was made by all the authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests regarding the publication of this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tan, K., Xu, C. & Long, C. Association of microbiota in the stomach of Sinanodonta woodiana and its cultured soil. 3 Biotech 10, 319 (2020). https://doi.org/10.1007/s13205-020-02313-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02313-2