Abstract
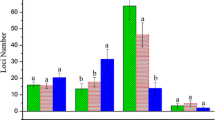
The epigenetic diversity of six genotype groups (commercial cultivars, S. officinarum, S. spontaneum, S. robustum, S. barberi, and Erianthus sp.) was assessed through methylation-sensitive amplification polymorphism (MSAP). A total of 1341 MSAP loci were analyzed, of which 1117 (83.29%) were susceptible to cytosine methylation and responsible for a higher proportion of overall diversity among genotypes. The MSAP selective primer combinations captured different proportions of internal and external cytosine methylation loci across genotype groups, while the average external cytosine frequency was higher for all genotype groups. The genotypes were divided into two subpopulations with a high differentiation index (φst = 0.086) based on epigenetic loci. The genotypes were clustered in three subgroups for both methylated and unmethylated loci, considering dissimilarity values. Four methylated fragments (MFs) were randomly selected and subsequently sequenced and compared with sugarcane public databases using BLASTN. MF alignments suggest that cytosine methylation occurs in sugarcane near CpG islands and tandem repeats within transcribed regions and putative cis-regulatory sequences, which assigned functions are associated with stress adaptation. These results provide the first insights about the distribution of this epigenetic mark in sugarcane genome, and suggest a biological relevance of methylated loci.




Similar content being viewed by others
References
Abuzayed M, El-Dabba N, Frary A, Doganlar S (2017) GDdom: an online tool for calculation of dominant marker gene diversity. Biochem Genet 55:155–157. https://doi.org/10.1007/s10528-016-9779-0
Afzal AJ, Wood AJ, Lightfoot DA (2008) Plant receptor-like serine threonine kinases: roles in signaling and plant defense. Mol Plant Microbe Interact 21:507–517. https://doi.org/10.1094/MPMI-21-5-0507
Aitken K, McNeil M (2010) Diversity analysis. In: Henry R, Kole MC (eds) Genetics, genomics and breeding of sugarcane, 1st edn. Science Publishers, New York, pp 19–42
Aitken KS, Li JC, Jackson P, Piperidis G, McIntyre CL (2006) AFLP analysis of genetic diversity within Saccharum officinarum and comparison with sugarcane cultivars. Aust J Agric Res 57(11):1167–1184. https://doi.org/10.1071/AR05391
Akimoto K, Katakami H, Kim HJ, Ogawa E, Sano CM, Wada Y, Sano H (2007) Epigenetic inheritance in rice plants. Ann Bot 100:205–217. https://doi.org/10.1093/aob/mcm110
Ali A, Pan YB, Wang QN, Wang JD, Chen JL, Gao SJ (2019) Genetic diversity and population structure analysis of Saccharum and Erianthus genera using microsatellite (SSR) markers. Sci Rep 9:395. https://doi.org/10.1038/s41598-018-36630-7
Alonso C, Pérez R, Bazaga P, Medrano M, Herrera CM (2016) MSAP markers and global cytosine methylation in plants: a literature survey and comparative analysis for a wild-growing species. Mol Ecol Resour 16:80–90. https://doi.org/10.1111/1755-0998.12426
Arceneaux G (1967) Cultivated sugarcane of the world and their botanical derivation. Proc ISSCT 12:844–854
Artschwager E (1954) A taxonomic study of Saccharum sinense Roxb and S. barberi Jeswiet. USDA Tech. Bull. No. 1089, Washington, DC
Ashikawa I (2001) Gene-associated CpG islands in plants as revealed by analyses of genomic sequences. Plant J 26:617–625. https://doi.org/10.1046/j.1365-313x.2001.01062.x
Brown JS, Schnell RJ, Power EJ, Douglas SL, Kuhn DN (2007) Analysis of clonal germplasm from five Saccharum species: S. barberi, S. robustum, S. officinarum, S. sinense and S. spontaneum. A study of inter- and intra species relationships using microsatellite markers. Genet Resour Crop Evol 54:627–648. https://doi.org/10.1007/s10722-006-0035-z
Da Silva JA (2017) The importance of the wild cane Saccharum spontaneum for bioenergy genetic breeding. Sugar Tech 19(3):229–240. https://doi.org/10.1007/s12355-017-0510-1
Daniels J, Roach BT (1987) Taxonomy and evolution. In: Heinz DJ (ed) Sugarcane improvement through breeding. Elsevier, Amsterdam, pp 7–14
D'Hont A, Paulet F, Glaszmann JC (2002) Oligoclonal interspecific origin of 'North Indian' and 'Chinese' sugarcanes. Chromosome Res 10:253–262. https://doi.org/10.1023/A:101520442
Earl DA, VonHoldt BM (2012) STRUCTURE HARVESTER: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4(2):359–361. https://doi.org/10.1007/s12686-011-9548-7
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620. https://doi.org/10.1111/j.1365-294X.2005.02553.x
Excoffier L, Lischer HE (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Fraga MF, Esteller M (2002) DNA methylation: a profile of methods and applications. Biotechniques 33:636–649. https://doi.org/10.2144/02333rv01
Francischini JHMB, Kemper EL, Costa JB, Manechini JRV, Pinto LR (2017) DNA methylation in sugarcane somaclonal variants assessed through methylation-sensitive amplified polymorphism. Genet Mol Res 16(2):gmr16029585. https://doi.org/10.4238/gmr16029585
Fulneček J, Kovařík A (2014) How to interpret methylation sensitive amplified polymorphism (MSAP) profiles? BMC Genet 15:2. https://doi.org/10.1186/1471-2156-15-2
Gachomo EW, Jimenez-Lopez JC, Baptiste LJ, Kotchoni SO (2014) GIGANTUS1 (GTS1), a member of Transducin/WD40 protein superfamily, controls seed germination, growth and biomass accumulation through ribosome-biogenesis protein interactions in Arabidopsis thaliana. BMC Plant Biol 14:37. https://doi.org/10.1186/1471-2229-14-37
Garsmeur O, Droc G, Antonise R, Grimwood J, Potier B, Aitken K, Jenkins J, Martin G, Charron C, Hervouet C, Costet L, Yahiaoui N, Healey A, Sims D, Cherukuri Y, Sreedasyam A, Kilian A, Chan A, Van Sluys MA, Swaminathan K, Town C, Bergès H, Simmons B, Glaszmann JC, van der Vossen E, Henry R, Schmutz J, D'Hont A (2018) A mosaic monoploid reference sequence for the highly complex genome of sugarcane. Nat Commun 9:2638. https://doi.org/10.1038/s41467-018-05051-5
Gent JI, Ellis NA, Guo L, Harkess AE, Yao Y, Zhang X, Dawe RK (2013) CHH islands: de novo DNA methylation in near-gene chromatin regulation in maize. Genome Res 23:628–637. https://doi.org/10.1101/gr.146985.112
Grativol C, Hemerly AS, Ferreira PC (2012) Genetic and epigenetic regulation of stress responses in natural plant populations. Biochim Biophys Acta 1819:176–185. https://doi.org/10.1016/j.bbagrm.2011.08.010
Guarino F, Cicatelli A, Brundu G, Heinze B, Castiglione S (2015) Epigenetic diversity of clonal white poplar (Populus alba L.) populations: could methylation support the success of vegetative reproduction strategy? PLoS ONE 10:e0131480. https://doi.org/10.1371/journal.pone.0131480
Hofmeister BT, Lee K, Rohr NA, Hall DW, Schmitz RJ (2017) Stable inheritance of DNA methylation allows creation of epigenotype maps and the study of epiallele inheritance patterns in the absence of genetic variation. Genome Biol 18:155. https://doi.org/10.1186/s13059-017-1288-x
Irvine JE (1999) Saccharum species as horticultural classes. Theor Appl Genet 98:186–194
Jackson P, Yuanhong F, Wei LQ, Haihua D, Piperidis G, Morgan T (2010) Use and improvement of sugarcane. ACIAR Final Rep. https://www.aciar.gov.au/node/11731. Accessed 17 Sep 2018
Ji L, Neumann DA, Schmitz RJ (2015) Crop epigenomics: identifying, unlocking, and harnessing cryptic variation in crop genomes. Mol Plant 8:860–870. https://doi.org/10.1016/j.molp.2015.01.021
Lei CP, Jiun KS, Choo CS, Singh R (2006) Analysis of tissue culture-derived regenerants using methylation sensitive AFLP. Asia Pac J Mol Biol Biotechnol 14(2):47–55
Li N, Xu C, Zhang A, Lv R, Meng X, Lin X, Gong L, Wendel JF, Liu B (2019) DNA methylation repatterning accompanying hybridization, whole genome doubling and homoeolog exchange in nascent segmental rice allotetraploids. New Phytol 223:979–992. https://doi.org/10.1111/nph.15820
Lima MLA, Garcia AAF, Oliveira KM, Matsuoka S, Arizono H, Souza JR, Souza AP (2002) Analysis of genetic similarity detected by AFLP and coefficient of parentage among genotypes of sugarcane (Saccharum spp.). Theor Appl Genet 104:30–38. https://doi.org/10.1007/s001220200003
Liu XL, Li XJ, Xu CH, Lin XQ, Deng ZH (2016) Genetic diversity of populations of Saccharum spontaneum with different ploidy levels using SSR molecular markers. Sugar Tech 18(4):365–372. https://doi.org/10.1007/s12355-015-0399-5
Ludwig DL, Chen F, Peterson SR, Nussenzweig A, Li GC, Chen DJ (1997) Ku80 gene expression is Sp1-dependent and sensitive to CpG methylation within a novel cis element. Gene 199:181–194. https://doi.org/10.1016/S0378-1119(97)00366-1
Luu PL, Schöler HR, Araúzo-Bravo MJ (2013) Disclosing the crosstalk among DNA methylation, transcription factors, and histone marks in human pluripotent cells through discovery of DNA methylation motifs. Genome Res 23:2013–2029. https://doi.org/10.1101/gr.155960.113
Ma KF, Song YP, Jiang XB, Zhang ZY, Li BL, Zhang D (2012) Photosynthetic response to genome methylation affects the growth of Chinese white poplar. Tree Genet Genomes 8:1407–1421. https://doi.org/10.1007/s11295-012-0527-2
Ma K, Song Y, Yang X, Zhang Z, Zhang D (2013) Variation in genomic methylation in natural populations of chinese white poplar. PLoS ONE 8:e63977. https://doi.org/10.1371/journal.pone.0063977
Nayak SN, Song J, Villa A, Pathak B, Ayala-Silva T, Yang X, Todd J, Glynn NC, Kuhn DN, Glaz B, Gilbert RA, Comstock JC, Wang J (2014) Promoting utilization of Saccharum spp. genetic resources through genetic diversity analysis and core collection construction. PLoS ONE 9:e110856. https://doi.org/10.1371/journal.pone.0110856
Ong-Abdullah M, Ordway JM, Jiang N, Ooi SE, Kok SY, Sarpan N, Azimi N, Hashim AT, Ishak Z, Rosli SK, Malike FA, Bakar NA, Marjuni M, Abdullah N, Yaakub Z, Amiruddin MD, Nookiah R, Singh R, Low ET, Chan KL, Azizi N, Smith SW, Bacher B, Budiman MA, Van Brunt A, Wischmeyer C, Beil M, Hogan M, Lakey N, Lim CC, Arulandoo X, Wong CK, Choo CN, Wong WC, Kwan YY, Alwee SS, Sambanthamurthi R, Martienssen RA (2015) Loss of Karma transposon methylation underlies the mantled somaclonal variant of oil palm. Nature 525:533–537. https://doi.org/10.1038/nature15365
Pérez-Figueroa A (2013) msap: a tool for the statistical analysis of methylation-sensitive amplified polymorphism data. Mol Ecol Resour 13:522–527. https://doi.org/10.1111/1755-0998.12064
Perrier X, Jacquemoud-Collet JP (2006) DAR-win software. https://www.darwin.cirad.fr/darwin. Accessed 10 Dec 2018
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Pritchard JK, Wen W, Falush D (2010) Documentation for STRUCTURE software: version 2.3. http://pritch.bsd.uchicago.edu/structure.html. Accessed 10 Dec 2018
Rombauts S, Florquin K, Lescot M, Marchal K, Rouzé P, van de Peer Y (2003) Computational approaches to identify promoters and cis-regulatory elements in plant genomes. Plant Physiol 132:1162–1176. https://doi.org/10.1104/pp.102.017715
Sahu PP, Pandey G, Sharma N, Puranik S, Muthamilarasan M, Prasad M (2013) Epigenetic mechanisms of plant stress responses and adaptation. Plant Cell Rep 32:1151–1159. https://doi.org/10.1007/s00299-013-1462-x
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Schübeler D (2015) Function and information content of DNA methylation. Nature 517:321–326. https://doi.org/10.1038/nature14192
Schulz B, Eckstein RL, Durka W (2013) Scoring and analysis of methylation-sensitive amplification polymorphisms for epigenetic population studies. Mol Ecol Resour 13:642–653. https://doi.org/10.1111/1755-0998.12100
Seo JS, Joo J, Kim MJ, Kim YK, Nahm BH, Song SI, Cheong JJ, Lee JS, Kim JK, Choi YD (2011) OsbHLH148, a basic helix-loop-helix protein, interacts with OsJAZ proteins in a jasmonate signaling pathway leading to drought tolerance in rice. Plant J 65:907–921. https://doi.org/10.1111/j.1365-313X.2010.04477.x
Singh P, Singh SP, Tiwari AK, Sharma BL (2017) Genetic diversity of sugarcane hybrid cultivars by RAPD markers. 3 Biotech 7:222. https://doi.org/10.1007/s13205-017-0855-x
Singh RB, Singh B, Singh RK (2019) Development of potential dbEST-derived microsatellite markers for genetic evaluation of sugarcane and related cereal grasses. Ind Crops Prod 128:38–47. https://doi.org/10.1016/j.indcrop.2018.10.071
Song Q, Chen ZJ (2015) Epigenetic and developmental regulation in plant polyploids. Curr Opin Plant Biol 24:101–109. https://doi.org/10.1016/j.pbi.2015.02.007
Springer NM, Schmitz RJ (2017) Exploiting induced and natural epigenetic variation for crop improvement. Nat Rev Genet 18:563–575. https://doi.org/10.1038/nrg.2017.45
Stirnimann CU, Petsalaki E, Russell RB, Müller CW (2010) WD40 proteins propel cellular networks. Trends Biochem Sci 35:565–574. https://doi.org/10.1016/j.tibs.2010.04.003
Team RC (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna (ISBN 3-900051-07-0)
To TK, Saze H, Kakutani T (2015) DNA methylation within transcribed regions. Plant Physiol 168:1219–1225. https://doi.org/10.1104/pp.15.00543
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23:4407–4414. https://doi.org/10.1093/nar/23.21.4407
Wright S (1978) Evolution and genetics of populations: variability within and among natural populations. University of Chicago Press, Chicago
Xu W, Zhang N, Jiao Y, Li R, Xiao D, Wang Z (2014) The grapevine basic helix-loop-helix (bHLH) transcription factor positively modulates CBF-pathway and confers tolerance to cold-stress in Arabidopsis. Mol Biol Rep 41:5329–5342. https://doi.org/10.1007/s11033-014-3404-2
Zhang H, Lang Z, Zhu JK (2018a) Dynamics and function of DNA methylation in plants. Nat Rev Mol Cell Biol 19:489–506. https://doi.org/10.1038/s41580-018-0016-z
Zhang J, Zhang X, Tang H, Zhang Q, Hua X, Ma X, Zhu F, Jones T, Zhu X, Bowers J, Wai CM, Zheng C, Shi Y, Chen S, Xu X, Yue J, Nelson DR, Huang L, Li Z, Xu H, Zhou D, Wang Y, Hu W, Lin J, Deng Y, Pandey N, Mancini M, Zerpa D, Nguyen JK, Wang L, Yu L, Xin Y, Ge L, Arro J, Han JO, Chakrabarty S, Pushko M, Zhang W, Ma Y, Ma P, Lv M, Chen F, Zheng G, Xu J, Yang Z, Deng F, Chen X, Liao Z, Lin Z, Lin H, Yan H, Kuang Z, Zhong W, Liang P, Wang G, Yuan Y, Shi J, Hou J, Jin J, Cao P, Shen Q, Jiang Q, Zhou P, Xu R, Liu J, Zhou Y, Jia H, Ma Q, Qi R, Zhang Z, Fang J, Fang H, Song J, Wang M, Dong G, Chen Z, Ma T, Liu H, Dhungana SR, Huss SE, Yang X, Sharma A, Trujillo JH, Martinez MC, Hudson M, Riascos JJ, Schuler M, Chen LQ, Braun DM, Li L, Yu Q, Wang J, Wang K, Schatz MC, Heckerman D, Van Sluys MA, Souza GM, Moore PH, Sankoff D, VanBuren R, Paterson AH, Nagai C, Ming R (2018b) Allele-defined genome of the autopolyploid sugarcane Saccharum spontaneum L. Nat Genet 50:1565–1573. https://doi.org/10.1038/s41588-018-0237-2
Acknowledgements
Authors are grateful to the Fundação de Amparo a Pesquisa do Estado de São Paulo (FAPESP, Grant 2013/22500-5). AAM was supported by “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” CAPES.
Author information
Authors and Affiliations
Contributions
LRP was responsible for the design and conceptualization of the experiment, and further supervised the study. AAM performed the experiments and the statistical analysis. MFS performed the sequencing and alignment analysis. AAM, MFS, and LRP wrote and revised the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Martins, A.A., da Silva, M.F. & Pinto, L.R. Epigenetic diversity of Saccharum spp. accessions assessed by methylation-sensitive amplification polymorphism (MSAP). 3 Biotech 10, 265 (2020). https://doi.org/10.1007/s13205-020-02257-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02257-7