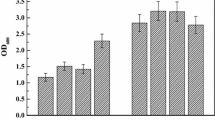
Abstract
Acetate is a potential low-cost carbon source that can be generated by biological and chemical processes. In this study, deletion of sdhAB encoding succinate dehydrogenase, iclR encoding the isocitrate lyase regulator, and maeB encoding the malic enzyme, and overexpression of acs encoding acetyl-CoA synthetase, gltA encoding citrate synthase, and acnB encoding aconitate hydratase in the wild-type Escherichia coli MG1655 strain yielded the recombinant E. coli strain WCY-7, which could synthesize succinate from acetate. After 48 h batch fermentation, this strain accumulated 11.23 mM succinate from 50 mM sodium acetate. This work indicates that microbial fermentation using acetate as the sole carbon source may be a suitable route to produce high yields of the valuable chemicals.



Similar content being viewed by others
References
Ahn JH, Jang YS, Lee SY (2016) Production of succinic acid by metabolically engineered microorganisms. Curr Opin Biotechnol 42:54–66
Amann E, Ochs B, Abel KJ (1988) Tightly regulated tac promoter vectors useful for the expression of unfused and fused proteins in Escherichia coli. Gene 69(2):301–315
Bhatia SK, Yang YH (2017) Microbial production of volatile fatty acids: current status and future perspectives. Rev Environ Sci Biotechnol 16(6):1–19
Bozell JJ, Petersen GR (2010) Technology development for the production of biobased products from biorefinery carbohydrates-the US Department of Energy’s “Top 10” revisited. Green Chem 4:539–554
Chen J, Li W, Zhang ZZ, Tan TW, Li ZJ (2018) Metabolic engineering of Escherichia coli for the synthesis of polyhydroxyalkanoates using acetate as a main carbon source. Microb Cell Fact 17(1):102
Cherepanov PP, Wackernagel W (1995) Gene disruption in Escherichia coli: TcR and KmR cassettes with the option of Flp-catalyzed excision of the antibiotic-resistance determinant. Gene 158(1):9–14
Datsenko KA, Wanner BL (2000) One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc Natl Acad Sci USA 97(12):6640–6645
Fernandez-Sandoval MT, Huerta-Beristain G, Trujillo-Martinez B, Bustos P, Gonzalez V, Bolivar F, Gosset G, Martinez A (2012) Laboratory metabolic evolution improves acetate tolerance and growth on acetate of ethanologenic Escherichia coli under non-aerated conditions in glucose-mineral medium. Appl Microbiol Biotechnol 96(5):1291–1300
Gibson DG, Young L, Chuang RY, Venter JC, Hutchison CA 3rd, Smith HO (2009) Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat Methods 6(5):343–345
Han K, Lim HC, Hong J (1992) Acetic acid formation in Escherichia coli fermentation. Biotechnol Bioeng 39(6):663–671
Huang JF, Zhang B, Shen ZY, Liu ZQ, Zheng YG (2018) Metabolic engineering of E. coli for the production of O-succinyl-l-homoserine with high yield. 3 Biotech 8(7):310
Kang Z, Gao C, Wang Q, Liu H, Qi Q (2010) A novel strategy for succinate and polyhydroxybutyrate co-production in Escherichia coli. Bioresour Technol 101(19):7675–7678
Lee PC, Lee WG, Lee SY, Chang HN (2001) Succinic acid production with reduced by-product formation in the fermentation of Anaerobiospirillum succiniciproducens using glycerol as a carbon source. Biotechnol Bioeng 72(1):41–48
Lerner CG, Inouye M (1990) Low copy number plasmids for regulated low-level expression of cloned genes in Escherichia coli with blue/white insert screening capability. Nucleic Acids Res 18(15):4631
Li Q, Yang M, Wang D, Li W, Wu Y, Zhang Y, Xing J, Su Z (2010) Efficient conversion of crop stalk wastes into succinic acid production by Actinobacillus succinogenes. Bioresour Technol 101(9):3292–3294
Li M, Gu P, Kang J, Wang Y, Wang Q, Qi Q (2012) Extending homologous sequence based on the single gene mutants by one-step PCR for efficient multiple gene knockouts. Folia Microbiol (Praha) 57(3):209–214
Li Y, Li M, Zhang X, Yang P, Liang Q, Qi Q (2013) A novel whole-phase succinate fermentation strategy with high volumetric productivity in engineered Escherichia coli. Bioresour Technol 149:333–340
Li Y, Huang B, Wu H, Li Z, Ye Q, Zhang YP (2016) Production of succinate from acetate by metabolically engineered Escherichia coli. ACS Synth Biol 5(11):1299–1307
Li J, Li Y, Cui Z, Liang Q, Qi Q (2017) Enhancement of succinate yield by manipulating NADH/NAD + ratio and ATP generation. Appl Microbiol Biotechnol 101(8):3153–3161
Lin H, Castro NM, Bennett GN, San KY (2006) Acetyl-CoA synthetase overexpression in Escherichia coli demonstrates more efficient acetate assimilation and lower acetate accumulation: a potential tool in metabolic engineering. Appl Microbiol Biotechnol 71(6):870–874
Majewski RA, Domach MM (1990) Simple constrained-optimization view of acetate overflow in E. coli. Biotechnol Bioeng 35(7):732–738
Pal P, Nayak J (2016) Acetic acid production and purification: critical review towards process intensification. Sep Purif Method 46(1):44–61
Song H, Kim TY, Choi BK, Choi SJ, Nielsen LK, Chang HN, Lee SY (2008) Development of chemically defined medium for Mannheimia succiniciproducens based on its genome sequence. Appl Microbiol Biotechnol 79(2):263–272
Wu QL, Guo WQ, Zheng HS, Luo HC, Feng XC, Yin RL, Ren NQ (2016) Enhancement of volatile fatty acid production by co-fermentation of food waste and excess sludge without pH control: the mechanism and microbial community analyses. Bioresour Technol 216:653–660
Xiao Y, Ruan Z, Liu Z, Wu SG, Varman AM, Liu Y, Tang YJ (2013) Engineering Escherichia coli to convert acetic acid to free fatty acids. Biochem Eng J 76(28):60–69
Xu X, Xie M, Zhao Q, Xian M, Liu H (2018) Microbial production of mevalonate by recombinant Escherichia coli using acetic acid as a carbon source. Bioengineered 9(1):116–123
Zeikus JG, Jain MK, Elankovan P (1999) Biotechnology of succinic acid production and markets for derived industrial products. Appl Microbiol Biotechnol 51(5):545–552
Zhang W, Zhu J, Zhu X, Song M, Zhang T, Xin F, Dong W, Ma J, Jiang M (2018) Expression of global regulator IrrE for improved succinate production under high salt stress by Escherichia coli. Bioresour Technol 254:151–156
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (31600066, 31100088), the Shandong Provincial Natural Science Foundation (ZR2016CB20, ZR2016CL02), State Key Laboratory of Microbial Technology Open Projects Fund (M2016-10), and the Shandong Province Science and Technology Development Plan (2013GSF12006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Niu, H., Li, R., Wu, J. et al. Production of succinate by recombinant Escherichia coli using acetate as the sole carbon source. 3 Biotech 8, 421 (2018). https://doi.org/10.1007/s13205-018-1456-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1456-z