Abstract
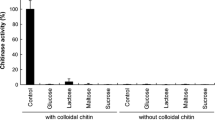
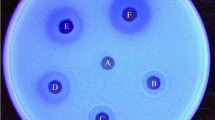
The filamentous fungus Aspergillus niveus produced extracellular antifungal chitinase when cultured under submerged fermentation (SbmF) using crab shells as the carbon source. Maximal chitinase production was achieved at 192 h of cultivation using minimal medium containing 1% chitin. The enzyme was purified 1.97-fold with 40% recovery by ammonium sulfate precipitation and Sephadex G-100 gel filtration. The molecular mass was estimated to be 44 kDa by both 12% SDS-PAGE and Sepharose CL-6B gel filtration. Maximal A. niveus chitinase activity was obtained at 65 °C and pH 5.0. The enzyme was fully stable at 60 °C for up to 120 min and the enzymatic activity was increased by Mn2+. In the presence of reducing and denaturing compounds, the enzyme activity was not drastically affected. The chitinase was able to hydrolyze colloidal chitin, azure chitin, and 4-nitrophenyl N-acetyl-β-D glucosaminide; for the latter, the K0.5 and maximal velocity (Vmax) were 3.51 mM and 9.68 U/mg of protein, respectively. The A. niveus chitinase presented antifungal activity against Aspergillus niger (MIC = 84 µg/mL), A. fumigatus (MIC = 21 µg/mL), A. flavus (MIC = 24 µg/mL), A. phoenicis (MIC = 24 µg/mL), and Paecilomyces variotii (MIC = 21 µg/mL). The fungus A. niveus was able to produce a thermostable and denaturation-resistant chitinase able to inhibit fungal development, signaling its biotechnological potential.




Similar content being viewed by others
References
Adams PR (1990) Mycelial amylase activities of thermophilic species of Rhizomucor, Humicola and Papulaspora. Mycopathologia 112:35–37. https://doi.org/10.1007/BF01795178
Berini F, Presti I, Beltrametti F, Pedroli M, Varum KM, Pellegioni L, Sjöling S, Marinelli F (2017) Production and characterization of a novel antifungal chitinase identified by functional screening of a suppressive-soil metagenome. Microb Cell Fact 16:16. https://doi.org/10.1186/s12934-017-0634-8
Binod P, Pusztaheyi T, Nagy N, Sandhya C, Szakács G, Pócsi I, Pandey A (2005) Production and purification of extracellular chitinases from Penicillium aculeatum NRRL 2129 under solid-state fermentation. Enzyme Microbial Technol 36:880–887. https://doi.org/10.1016/j.enzmictec.2004.12.031
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilization the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Duong-Ly KC, Gabelli SB (2014) Salting out of proteins using ammonium sulfate precipitation. Methods Enzymol 541:85–94. https://doi.org/10.1016/B978-0-12-420119-4.00007-0
Farag MA, Al-Nusarie ST (2014) Production, optimization, characterization and antifungal activity of chitinase produced by Aspergillus terreus. Afr J Biotechnol 13:1567–1578. https://doi.org/10.5897/AJB2014.13628
Farag AM, Abd-Elnabey HM, Ibrahim HAH, El-Shenawy M (2016) Purification, characterization and antimicrobial activity of chitinase from marine derived Aspergillus terreus. Egypt J Aquat Res 42:185–192. https://doi.org/10.1016/j.ejar.2016.04.004
Gun Lee D, Shin SY, Maeng CY, Jin ZZ, Kim KL, Hahm K-S (1999) Isolation and characterization of a novel antifungal peptide from Aspergillus niger. Biochem Biophys Res Commun 263:646–651. https://doi.org/10.1006/bbrc.1999.1428
Guo R-F, Shi B-S, Li D-C, Ma W, Wei Q (2008) Purification and characterization of a novel thermostable chitinase from Thermomyces lanuginosus SY2 and cloning of its encoding gene. Agric Sci China 7(12):1458–1465
Hameed AAA, Ayesh AM, Mohamed MAR, Mawla HFA (2012) Fungi and some mycotoxins producing species in the air of soybean and cotton mills: a case study. Atmos Pollut Res 3:126–131
Hill TW, Kafer E (2001) Improved protocols for Aspergillus minimal medium: trace element and minimal medium salt stock solutions. Fungal Genet Newsl 48:20–21. https://doi.org/10.4148/1941-4765.1173
Khan MA, Hamid R, Ahmad M, Abdin MZ, Javed S (2010) Optimization of culture media for enhanced chitinase production from a novel strain of Stenotrophomonas maltophilia using response surface methodology. J Microbiol Biotechnol 20(11):1597–1602. https://doi.org/10.4014/jmb.0909.09040
Khanna P, Sundari SS, Kumar NJ (1995) Production, isolation and partial purification of xylanase from Aspergillus sp. World J Microbiol Biotechnol 11:242–243. https://doi.org/10.1007/BF00704661
Kumar P, Mahato DK, Kamle M, Mohanta TK, Kang SG (2017) Aflatoxins: a global concern for food safety, human health and their management. Front Microbiol 7:2170. https://doi.org/10.3389/fmicb.2016.02170
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Lee YG, Chung KC, Wi SG, Lee JC, Bae HJ (2009) Purification and properties of a chitinase from Penicillium sp. LYG 0704. Protein Expr Purif 65:244–250. https://doi.org/10.1016/j.pep.2008.12.004
Li D-C (2006) Review of fungal chitinases. Mycopathologia 161:345–360. https://doi.org/10.1007/s11046-006-0024-y
Lodhi G, Kim YS, Hwang JW, Kim SK, Jeon YD, Je JY, Ahn BB, Moon SH, Jeon BT, Park PJ (2014) Chitooligosaccharide and its derivatives: preparation and biological applications. Biomed Res Int 2014:1–13. https://doi.org/10.1155/2014/654913 (Article ID 654913).
Mania D, Hilpert K, Ruden S, Fischer R, Takeshita N (2010) Screening for antifungal peptides and their modes of action in Aspergillus nidulans. Appl Environ Microbiol 76(21):7102–7108
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Pace CN, Huyghues-Despointes BMP, Fu H, Takano K, Scholtz JM, Grimsley GR (2010) Urea denatured state ensembles contain extensive secondary structure that is increased in hydrophobic proteins. Protein Sci 19:929–943. https://doi.org/10.1002/pro.370
Peralta RM, Terenzi HF, Jorge JA (1990) β-d-glycosidase activities of Humicola grisea var. thermoidea: biochemical and kinetic characterization of a multifunctional enzyme. Biochim Biophys Acta 1033:243–249. https://doi.org/10.1016/0304-4165(90)90127-l
Porter CM, Miller BG (2012) Cooperativity in monomeric enzymes with single ligand-binding sites. Bioorg Chem 43:44–50. https://doi.org/10.1016/j.bioorg.2011.11.001
Gómez Ramírez M, Rojas Avelizapaa LI, Rojas Avelizapab NG, Cruz Camarillo R (2004) Colloidal chitin stained with Remazol Brilliant Blue RR, a useful substrate to select chitinolytic microorganisms and to evaluate chitinases. J Microbiol Methods 56:213–219
Rathore AS, Gupta RD (2015) Chitinases from bacteria to human: properties applications, and perspectives. Enzyme Res 2015:1–8. https://doi.org/10.1155/2015/791907 (Article ID 791907)
Rattanakit N, Yano S, Plikomol A, Wakayama M, Tachiki T (2007) Purification of Aspergillus sp. S1-13 chitinases and their role in saccharification of chitin in mash of solid-state culture with shellfish waste. J Biosci Bioeng 103:535–541. https://doi.org/10.1263/jbb.103.535
Rizatti AC, Jorge JA, Terenzi HF, Rechia CG, Polizeli ML (2001) Purification and properties of a thermostable extracellular β-d-xylosidase produced by a thermotolerant Aspergillus phoenicis. J Ind Microbiol Biotechnol 3:156–160. https://doi.org/10.1038/sj/jim/7000107
Rojas-Avelizapa LI, Cruz-Camarillo R, Guerrero MI, Rodríguez-Vázquez R, Ibarra JE (1999) Selection and characterization of a proteo-chitinolytic strain of Bacillus thuringiensis, able to grow in shrimp waste media. World J Microbiol Biotechnol 15(2):299–308
Rustiguel CB (2014) Comparison of the biochemical properties of the chitinases produced by different isolates of Metarhizium anisopliae. Thesis, University of São Paulo, Brazil
Seidl V (2008) Chitinases of filamentous fungi: a large group of diverse proteins with multiple physiological functions. Fungal Biol Rev 22:36–42. https://doi.org/10.1016/j.fbr.2008.03.002
Shehataa AN, Abd El Aty AA, Darwishc DA, Wahabb WAA, Mostafa FA (2018) Purification, physicochemical and thermodynamic studies of antifungal chitinase with production of bioactive chitosan-oligosaccharide from newly isolated Aspergillus griseoaurantiacus KX010988. Int J Biol Macromol 107:990–999
Singh B, Singh S, Asif AR, Oellerich M, Sharma GL (2014) Allergic aspergillosis and the antigens of Aspergillus fumigatus. Curr Protein Pept Sci 15(5):403–423. https://doi.org/10.2174/1389203715666140512120605
Swiontek Brzezinska M, Jankiweicz U (2012) Production of chitinase by Aspergillus niger LOCK 62 and Its potential role in the biological control. Curr Microbiol 65:666–672. https://doi.org/10.1007/s00284-012-0208-2
Thimoteo SS, Glogauer A, Faoro H, Souza EM, Huergo LF, Moerschbacher BM, Pedrosa FO (2017) A broad pH range and processive chitinase from metagenome library. Braz J Med Biol Res 50(1):e5658. https://doi.org/10.0.6.54/1414-431x20165658
Wiseman A (1975) Industrial practice with enzymes. In: Wiseman A (ed) Handbook of enzyme biotechnology, 1rd edn. Horwood, Chichester, pp 243–272
Xia G, Jin CZ, Hou JY, Ang SZ, Hang S, Jin C (2001) A novel chitinase having a unique model of action from Aspergillus fumigatus YJ-407. Eur J Biochem 208:4079–4085. https://doi.org/10.1046/j.1432-1327.2001.02323.x
Acknowledgements
We thank Mauricio de Oliveira for technical assistance. The authors also kindly acknowledge the financial support from FAPESP (Process no. 2011/50880-1) and the research scholarships from Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). This manuscript is part of the doctoral thesis by T.B.A.
Author information
Authors and Affiliations
Contributions
LHSG and TBA designed the study; TBA performed the experiments on chitinase production, purification, and characterization; PHOO performed the experiments on antifungal activity; LHSG, TBA, AHCO, and JAJ analyzed the results; LHSG and TBA wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Rights and permissions
About this article
Cite this article
Alves, T.B., de Oliveira Ornela, P.H., de Oliveira, A.H.C. et al. Production and characterization of a thermostable antifungal chitinase secreted by the filamentous fungus Aspergillus niveus under submerged fermentation. 3 Biotech 8, 369 (2018). https://doi.org/10.1007/s13205-018-1397-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1397-6