Abstract
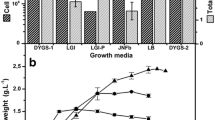
Use of agro-waste for production of value added products is a good alternative for developing low-cost carriers for formulation of Trichoderma-based bio-products. It provides avenues for safe utilization of wastes, while reducing cost and environment pollution load of waste disposal. The present study was undertaken to find suitable agro-waste for economical and higher mass production of Trichoderma lixii TvR1 under solid-state fermentation, optimizing culture conditions using mathematical model and assessing effect of formulated bio-product on growth of Spinach (Spinacia oleracea). Among various agro-wastes screened, sugarcane bagasse was observed to support maximum growth (20.08 × 107 spores/g) of T. lixii TvR1 which was significantly (p ≤ 0.05) higher than the others. The Response Surface Methodology (RSM) was used to optimize culture conditions using optimal point prediction analysis which predicted that maximum spore production of T. lixii TvR1 (19.1245 × 107 spores/g) will be obtained at 30 °C and 68.87% of moisture content after 31 days of incubation. Amendment of formulated bio-product of T. lixii TvR1 in soil at concentration 15% w/w promoted biomass, photosynthetic pigments, and protein content of spinach (significant at p ≤ 0.05). After 6 weeks of sowing the shoot length, root length, and photosynthetic pigments of plants irrigated daily and on alternate days were reported to be increased by 66.97, 185.03, and 82.80%; and 56.56, 71.36, and 74.64%, respectively; over the no amendment.


Similar content being viewed by others
References
Akladious SA, Abbas SM (2014) Application of Trichoderma harzianum T22 as a biofertilizer potential in Maize growth. J Plant Nutr 37(1):30–49. https://doi.org/10.1080/01904167.2013.829100
Bae H, Sicher RC, Kim MS, Kim SH, Strem MD, Melnick RL, Bailey BA (2009) The beneficial endophyte Trichoderma hamatum isolate DIS 219b promotes growth and delays the onset of the drought response in Theobroma cacao. J Exp Bot 60:3279–3295. https://doi.org/10.1093/jxb/erp165
Błaszczyk L, Siwulski M, Sobieralski K, Lisiecka J, Jedryczka M (2014) Trichoderma spp.—application and prospects for use in organic farming and industry. J Plant Prot Res 54(4):309–317. https://doi.org/10.2478/jppr-2014-0047
Box G, Behnken D (1960) Some new three level designs for the study of quantitative variables. Technometrics 2(4):455–475. https://doi.org/10.1080/00401706.1960.10489912
Cavalcante RS, Lima HLS, Pinto GAS, Gava CAT, Rodrigues S (2008) Effect of moisture on Trichoderma conidia production on corn and wheat bran by Solid State Fermentation. Food Bioprocess Tech 1(1):100–104. https://doi.org/10.1007/s11947-007-0034-x
Contreras-Cornejo HA, Macias-Rodriguez L, Cortes-Penagos C, Lopez-Bucio J (2009) Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in arabidopsis. Plant Physiol 149:1579–1592. https://doi.org/10.1104/pp.108.130369
Contreras-Cornejo HA, Macias-Rodriguez L, del-Val E, Larsen J (2016) Ecological functions of Trichoderma spp. and their secondary metabolites in the rhizosphere: interactions with plants. FEMS Microbiol Ecol. https://doi.org/10.1093/femsec/fiw036
Doni F, Isahak A, Zain CRCM, Yusoff WMW (2014) Physiological and growth response of rice plants (Oryza sativa L.) to Trichoderma spp. inoculants. AMB Express 4:45. https://doi.org/10.1186/s13568-014-0045-8
Eltem R, Sayit S, Sozer S, Sukan FV (2017) Production of Trichoderma citrinoviride micropropagules as a biocontrol agent by means of an economical process. US Patent 9551012
Garnica-Vergara A, Barrera-Ortiz S, Munoz-Parra E, Raya-Gonzalez J, Mendez-Bravo A, Macias-Rodriguez L, Ruiz-Herrera LF, López-Bucio J (2015) The volatile 6-pentyl-2H-pyran-2-one from Trichoderma atroviride regulates Arabidopsis thaliana root morphogenesis via auxin signaling and ethylene insensitive 2 functioning. N Phytol 209:1496–1512. https://doi.org/10.1111/nph.13725
Hyder S, Inam-ul-Haq MD, Bibi S, Malik AH, Ghuffar S, Iqbal S (2017) Novel potential of Trichoderma spp. as biocontrol agent. J Entomol Zool Stud 5(4):214–222
Kaya C, Ashraf M, Sonmez O, Aydemir S, Tuna AL, Cullu MA (2009) The influence of arbuscular mycorrhizal colonization on key growth parameters and fruit yield of pepper plants grown at high salinity. Sci 121(1):1–6. https://doi.org/10.1016/j.scienta.2009.01.001
Keswani C, Mishra S, Sarma BK, Singh SP, Singh HB (2014) Unraveling the efficient application of secondary metabolites of various Trichoderma. Appl Microbiol Biotechnol 98:533–544
Lichtenthaler HK, Wellburn AR (1983) Determinations of total carotenoids and chlorophylls a and b of leaf extracts in different solvents. Biochem Soc Trans 11:591–592. https://doi.org/10.1042/bst0110591
Lowry OH, Rosembrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Martinez-Medina A, Roldan A, Albacete A, Pascual JA (2011) The interaction with arbuscular mycorrhizal fungi or Trichoderma harzianum alters the shoot hormonal profile in melon plants. Phytochemistry 72:223–229. https://doi.org/10.1016/j.phytochem.2010.11.008
Mastouri F, Björkman T, Harman GE (2010) Seed treatment with Trichoderma harzianum alleviates biotic, abiotic and physiological stresses in germinating seeds and seedlings. Phytopathology 100(11):1213–1221. https://doi.org/10.1094/phyto-03-10-0091
Mishra A, Salokhe VM (2011) Rice growth and physiological responses to SRI water management and implications for crop productivity. Paddy Water Environ 9:41–52. https://doi.org/10.1007/s10333-010-0240-4
Mohiddin FA, Bashir I, Padder SA, Hamid B (2017) Evaluation of different substrates for mass multiplication of Trichoderma species. J Pharmacogn Phytochem 6(6):563–569
Pang PK, Darah I, Poppe L, Szakacs G, Ibrahim CO (2006) Xylanase production by a local isolate, Trichoderma spp. FETL c3-2 via solid state fermentation using agricultural wastes as substrates. Malays J Microbiol 2(1):7–14
Rajput AQ, Shahzad S (2015) Growth and sporulation of Trichoderma polysporum on organic substrate by addition of carbon and nitrogen sources. Pak J Bot 47(3):979–986
Rama SS, Singh HV, Singh P, Kaur J (2001) A comparison of different substrates for the mass production of Trichoderma. Ann Pl Protec Sci 9(2):248–253
Rini CR, Sulochana KK (2007) Substrate evaluation for multiplication of Trichoderma spp. J Trop Agric 45(1–2):58–60
Sachdev S, Singh RP (2016) Current challenges, constraints and future strategies for development of successful market for biopesticides. Clim Change Environ Sustain 4(2):129–136. https://doi.org/10.5958/2320-642X.2016.00014.4
Sachdev S, Singh RP (2018) Isolation, characterisation and screening of native microbial isolates for biocontrol of fungal pathogens of tomato. Clim Change Environ Sustain 6(1):46–58. https://doi.org/10.5958/2320-642X.2018.00006.6
Shahid M, Singh A, Srivastava M, Mishra RP, Biswas SK (2011) Effect of temperature, pH and media for growth and sporulation of Trichoderma longibrachiatum and self life study in carrier based formulations. Ann Pl Protec Sci 19(1):147–149
Shamshad A, Vinayak VP, Richa K, Ashwani K, Suresh BNK (2018) Optimization of nutrient stress using C. pyrenoidosa for lipid and biodiesel production in integration with remediation in dairy industry wastewater using response surface methodology. 3 Biotech 8:326. https://doi.org/10.1007/s13205-018-1342-8
Shoresh M (2010) Induced systemic resistance and plant responses to fungal biocontrol agents. Annu Rev Phytopathol 48:21–43. https://doi.org/10.1146/annurev-phyto-073009-114450
Shukla N, Awasthi RP, Rawat L, Kumar J (2012) Biochemical and physiological responses of rice (Oryza sativa L.) as influenced by Trichoderma harzianum under drought stress. Plant Physiol Biochem 54:78–88. https://doi.org/10.1016/j.plaphy.2012.02.001
Siddhartha NS, Amara KV, Ramya Mol KA, Saju KA, Harsha KN, Sharanappa P, Kumar KP (2017) Evaluation of substrates for mass production of Trichoderma harzianum and its compatibility with Chlorpyrifos + Cypermethrin. Int J Curr Microbiol App Sci 6(8):3628–3635. https://doi.org/10.20546/ijcmas.2017.608.437
Singh PC, Nautiyal CS (2012) A novel method to prepare concentrated conidial biomass formulation of Trichoderma harzianum for seed application. J Appl Microbiol 113(6):1442–1450. https://doi.org/10.1111/j.1365-2672.2012.05426.x
Subramanian KS, Santhanakrishnan P, Balasubramanian P (2006) Responses of field grown tomato plants to arbuscular mycorrhizal fungal colonization under varying intensities of drought stress. Sci Hortic 107:245–253. https://doi.org/10.1016/j.scienta.2005.07.006
Tewari L, Bhanu C (2004) Evaluation of agro-industrial waste for conidia based inoculum production of biocontrol agent: Trichoderma harzianum. J Sci Ind Res 63:807–812
Vitti A, Pellegrini E, Nali C, Lovelli S, Sofo A, Valerio M, Scopa A, Nuzzaci M (2016) Trichoderma harzianum T-22 induces systemic resistance in tomato infected by Cucumber mosaic virus. Front Plant Sci 7:1520. https://doi.org/10.3389/fpls.2016.01520
Yedidia I, Srivastva AK, Kapulnik Y, Chet I (2001) Effect of Trichoderma harzianum on microelement concentrations and increased growth of cucumber plants. Plant Soil 235(2):235–242. https://doi.org/10.1023/a:1011990013955
Acknowledgements
Authors gratefully acknowledge University Grant Commission, New Delhi, India for providing UGC Major Research Project; MRP-MAJOR-ENV-2013-11912 and UGC-Senior Research Fellowship grant to Swati Sachdev.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sachdev, S., Singh, A. & Singh, R.P. Optimization of culture conditions for mass production and bio-formulation of Trichoderma using response surface methodology. 3 Biotech 8, 360 (2018). https://doi.org/10.1007/s13205-018-1360-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1360-6