Abstract
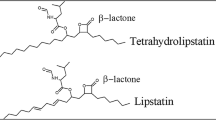
In the present study, lipstatin production was studied from different mutants of Streptomyces toxytricini which were developed using ultraviolet radiation (exposure time 30 s, 1, 2, 5 and 10 min), ethyl methane sulfonte, methyl methane sulfonate (MMS) and N-methyl-N′-intro-N-nitrosoguanidine (NTG) treatments (50, 100, 200, 500, 1000 µM, respectively). Highest yielding mutants were provided precursor supplementation of citric acid, thiamine and biotin (each 1 g/L) at idiophase for further enhancement in the production of lipstatin. Screened mutants produced biomass in the range of 5.8–7.16 g/L which were lesser than control. Screened mutants also exhibited pellet morphology in submerged culture. Out of these mutants, NTG8 mutant produced highest amount of lipstatin (1383.25 mg/L) with 9.606 mg/L/h productivity. Precursor supplementation to this mutant further increased the production to 2387.81 mg/L. Mutant was validated in 5 L bioreactor and lipstatin production was enhanced to 2519.34 mg/L.





Similar content being viewed by others
References
Baltz RH (1998) New genetic methods to improve secondary metabolite production in Streptomyces. J Ind Microbiol Biotechnol 20:360–363. https://doi.org/10.1038/sj.jim.2900538
Baltz RH (2001) Genetic methods and strategies for secondary metabolite yield improvement in actinomycetes. Antonie Van Leeuwenhoek 79:251–259
Baltz RH, Stonesifer J (1985) Adaptive response and enhancement of N-methyl-N’-nitro-N-nitrosoguanidine mutagenesis by chloramphenicol in Streptomyces fradiae. J Bacteriol 164(2):944–946
Barrios-Gonzalez J, Fernandez FJ, Tomasini A (2003) Microbial secondary metabolites production and strain improvement. Indian J Biotechnol 2:322–333
Berg JM, Tymoczko JL, Stryer L (2002) Biochemistry, 5th edn. W. H. Freeman and Company, New York
Celler K, Picioreanu C, van Loosdrecht MCM, van Wezel GP (2012) Structured morphological modeling as a framework for rational strain design of Streptomyces species. Antonie Van Leeuwenhoek 102:409–423. https://doi.org/10.1007/s10482-012-9760-9
Coulondre C, Miller JH (1977) Genetic studies of the lac repressor: IV. Mutagenic specificity in the lacl gene of E. coli. J Mol Biol 117(3):577–606. https://doi.org/10.1016/0022-2836(77)90059-6
Dhingra S, Dubey KK, Bhushan G (2014) Enhancement in Jatropha-based biodiesel yield by process optimisation using design of experiment approach. Int J Sustain Energ 33(4):842–853. https://doi.org/10.1080/14786451.2013.777335
Eisenreich W, Kupfer E, Weber W, Bacher A (1997) Tracer studies with crude U-13C-lipid mixtures. Biosynthesis of the lipase inhibitor lipstatin. J Biol Chem 272(2):867–874. https://doi.org/10.1074/jbc.272.2.867
Gao H, Liu M, Zhuo Y, Zhou X, Liu J, Chen D, Zhang W, Gou Z, Shang P, Zhang L (2010) Assessing the potential of an induced-mutation strategy for avermectin overproducers. Appl Environ Microbiol 76(13):4583–4586. https://doi.org/10.1128/AEM.01682-09
Goese M, Eisenreich W, Kupfer E, Stohler P, Weber W, Leuenberger HG, Bacher A (2001) Biosynthesis of lipstatin. Incorporation of multiply deuterium-labeled (5Z,8Z)-tetradeca-5,8-dienoic acid and octanoic acid. J Org Chem 66(13):4673–4678. https://doi.org/10.1021/jo010230b
Hopkins L, Bond MA, Tobin AK (2002) Ultraviolet-B radiation reduces the rates of cell division and elongation in the primary leaf of wheat (Triticum aestivum L. cv Maris Huntsman). Plant Cell Environ 25:617–624. https://doi.org/10.1046/j.0016-8025.2001.00834.x
Hörtnagl P, Pérez MT, Sommaruga R (2011) Contrasting effects of ultraviolet radiation on the growth efficiency of freshwater bacteria. Aquat Ecol 45(1):125–136. https://doi.org/10.1007/s10452-010-9341-9
Ikehata H, Ono T (2011) The mechanisms of UV mutagenesis. J Radiat Res 52(2):115–125. https://doi.org/10.1269/jrr.10175
Koebsch I, Overbeck J, Piepmeyer S, Meschke H, Schrempf H (2009) A molecular key for building hyphae aggregates: the role of the newly identified Streptomyces protein HyaS. Microb Biotechnol 2(3):343–360. https://doi.org/10.1111/j.1751-7915.2009.00093.x
Kumar AK (2015) UV mutagenesis treatment for improved production of endoglucanase and β-glucosidase from newly isolated thermotolerant actinomycetes, Streptomyces griseoaurantiacus. Bioresour Bioprocess 2:22. https://doi.org/10.1186/s40643-015-0052-x
Kumar P, Dubey KK (2015) Current trends and future prospects of lipstatin: a lipase inhibitor and pro-drug for obesity. RSC Adv 5:86954–86966. https://doi.org/10.1039/C5RA14892H
Kumar P, Dubey KK (2016) Modulation of fatty acid metabolism and tricarboxylic acid cycle to enhance the lipstatin production through medium engineering in Streptomyces toxytricini. Bioresour Technol 213:64–68. https://doi.org/10.1016/j.biortech.2016.01.133
Kumar P, Dubey KK (2017) Mycelium transformation of Streptomyces toxytricini into pellet: role of culture conditions and kinetics. Bioresour Technol 228:339–347. https://doi.org/10.1016/j.biortech.2017.01.002
Kumar AK, Parikh BP, Singh SP, Shah D (2015) Use of combined UV and chemical mutagenesis treatment of Aspergillus terreus D34 for hyper-production of cellulose-degrading enzymes and enzymatic hydrolysis of mild-alkali pretreated rice straw. Bioresour Bioprocess 2:35. https://doi.org/10.1186/s40643-015-0062-8
Luthra U, Kumar H, Dubey RC (2013a) Mutagenesis of the lipstatin producer Streptomyces toxytricini ATCC 19813. J Biotechnol Lett 4:68–71
Luthra U, Kumar H, Kulshreshtha N, Tripathi A, Trivedi A, Khadpekar S, Chaturvedi A, Dubey RC (2013b) Medium optimization for the production of lipstatin by Streptomyces toxytricini using full factorial design of experiment. Nat Sci 11:73–76
Malik A, Khan AU, Lal SK (2002) Chemoprotection profiles of sodium thiosulfate on methyl methanesulfonate-induced mutagenesis of bacteriophage T4. Med Sci Monit 8(6):BR212-20
Miller JH (1983) Mutational specificity in bacteria. Ann Rev Genet 17:215–238. https://doi.org/10.1146/annurev.ge.17.120183.001243
Mo S, Lee SK, Jin YY, Oh CH, Suh JW (2013) Application of a combined approach involving classical random mutagenesis and metabolic engineering to enhance FK506 production in Streptomyces sp. RM7011. Appl Microbiol Biotechnol 97(7):3053–3062. https://doi.org/10.1007/s00253-012-4413-5
Rodgers RJ, Tschöp MH, Wilding JP (2012) Anti-obesity drugs: past, present and future. Dis Model Mech 5(5):621–626. https://doi.org/10.1242/dmm.009621
Savergave LS, Gadre RV, Vaidya BK, Jogdand VV (2013) Two-stage fermentation process for enhanced mannitol production using Candida magnoliae mutant R9. Bioprocess Biosyst Eng 36:193–203. https://doi.org/10.1007/s00449-012-0775-4
Shockman GD, Barrett JF (1983) Structure, function, and assembly of cell walls of Gram-positive bacteria. Annu Rev Microbiol 37:501–527. https://doi.org/10.1146/annurev.mi.37.100183.002441
Stonesifer J, Baltz RH (1985) Mutagenic DNA repair in Streptomyces. Proc Natl Acad Sci USA 82:1180–1183
Thibessard A, Fernandez A, Gintz B, Leblond-Bourget N, Bernard Decaris B (2002) Effects of rodA and pbp2b disruption on cell morphology and oxidative stress response of Streptococcus thermophilus CNRZ368. J Bacteriol 184(10):2821–2826. https://doi.org/10.1128/JB.184.10.2821-2826.2002
van Dissel D, Claessen D, Roth R, van Wezel GP (2015) A novel locus for mycelial aggregation forms a gateway to improved Streptomyces cell factories. Microb Cell Fact 14:44. https://doi.org/10.1186/s12934-015-0224-6
Wakil SJ, Stoops JK, Joshi VC (1983) Fatty acid synthesis and its regulation. Annu Rev Biochem 52(1):537–579. https://doi.org/10.1146/annurev.bi.52.070183.002541
Weibel EK, Hadvary P, Hochuli E, Kupfer E, Lengsfeld H (1987) Lipstatin, an inhibitor of pancreatic lipase, produced by Streptomyces toxytricini I. Producing organism, fermentation, isolation and biological activity. J Antibiot 40(8):1081–1085. https://doi.org/10.7164/antibiotics.40.1081
Zhu T, Wang L, Wang W, Hu Z, Yu M, Wang K, Cui Z (2014) Enhanced production of lipstatin from Streptomyces toxytricini by optimizing fermentation conditions and medium. J Gen Appl Microbiol 60:106–111. https://doi.org/10.2323/jgam.60.106
Acknowledgements
Authors sincerely acknowledge Science and Engineering Research Board (Department of Science and Technology, Govt. of India, Grant number: SR/FT/LS-45/2012) for providing financial assistance. Maharshi Dayanand University Rohtak, Haryana, India for providing necessary facilities and Agricultural Research Service (NRRL) United States Department of Agriculture for providing strain used in this research work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Rights and permissions
About this article
Cite this article
Kumar, P., Dubey, K.K. Implication of mutagenesis and precursor supplementation towards the enhancement of lipstatin (an antiobesity agent) biosynthesis through submerged fermentation using Streptomyces toxytricini . 3 Biotech 8, 29 (2018). https://doi.org/10.1007/s13205-017-1049-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-017-1049-2