Abstract
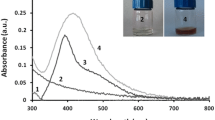
Silver nanoparticles (AgNPs) have been recognized for their unique physical and chemical properties, silver nanoparticles (AgNPs) have identified themselves as one of the most shows potential nanoparticles in biomedical and biological applications, including anticancer, drug delivery, and antimicrobial agents. The release of [Ag +], which is toxic to microorganisms, could be responsible for AgNPs’ bactericidal properties. Synthesis of silver nanoparticles (NPs) using lactic acid bacteria could be a good environmentally friendly substitute for physical and chemical methods. Lactobacillus acidophilus was identified by biochemical and molecular methods and rummage sale for the green mixture of AgNPs. The AgNPs produced within the L. acidophilus culture medium were characterized by color change after L. acidophilus development by 1 mM AgNO3 at 37 °C. The NPs configuration was established by UV–visible at 434 nm. Characterization by transmission electron microscopy (TEM) proved that the sphere-shaped poly-dispersed AgNPs were in the scope variety of 19–25 nm. Fourier-transform infrared spectroscopy showed high levels of AgNP-steadying proteins and other subordinate metabolites. X-ray deflection exposed a face-centered cubic deflection spectrum by a crystal-like countryside. The antimicrobial action of AgNPs was verified against Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, and Salmonella enterica), Gram-positive bacteria (Staphylococcus aureus and Bacillus subtilis), and a fungus (Candida albicans). The objective of this study was to investigate the biosynthesis of silver NPs using Lactobacillus and it is antibacterial and cytotoxicity activities of synthesized nanoparticles. The E. coli and S. enterica were highly susceptible, which were (21 and 20 mm), whereas S. aureus and C. albicans were the least susceptible, which were (10 and 9 mm). The findings on the cytotoxicity of AgNPs against Caco, A549, and HepG2 cell lines of cancer showed that the concentrations giving half-maximal responses were 5, 15, and 30 mg/ml, respectively. This study presents a well-organized and rapid method of synthesizing stable AgNPs using L. acidophilus with high antimicrobial and anticancer activities.






Similar content being viewed by others
References
Abdel-Mohsen AM, Hrdina R, Burgert L, Abdel-Rahman RM, Hašová M, Šmejkalová D, Kolář M, Pekar M, Aly AS (2013) Antibacterial activity and cell viability of hyaluronan fiber with silver nanoparticles. Carbohydr Polym 92:1177–1187
Ahmad A, Mukherjee P, Senapati S, Mandal D, Khan MI, Kumar R, Sastry M (2003) ‘Extracellular biosynthesis of silver nanoparticles using the fungus Fusariumoxysporum. Colloids Surf 28:313–318
Ahn YT, Lim KL, Ryu JC, Kang DK, Ham JS, Jang YH, Kim HU (2002) Characterization of Lactobacillus acidophilus isolated from piglets and chicken. Asian Aust J Anim Sci 15:1790–1797
Alani F, Moo-Young M, Anderson W (2012) Biosynthesis of silver nanoparticles by a new strain of Streptomyces sp. compared with Aspergillusfumigatus’. World J Microbiol Biotechnol 28:1081–1086
Alhayani B, Abdallah AA (2020) Manufacturing intelligent Corvus corone module for a secured two way image transmission under WSN. Eng Comput 37(9):1–17
Alhayani B, Ilhan H (2020a) Efficient cooperative imge transmission in one-Way mult-hop sensor network. Internat J Electrical Eng Educ 57(2):321–339
Alhayani B, Ilhan H (2020b) Image transmission over decode and forward based cooperative wireless multimedia sensor networks for Rayleigh fading channels in medical internet of things (MIoT) for remote health-care and health communication monitoring. J Med Imaging Health Inform 10:160–168
Baker MJ, Hussain SR, Lovergne L, Untereiner V, Hughes C, Lukaszewski RA, Thiéfin G, Sockalingum GD (2016) Developing and understanding biofluid vibrational spectroscopy: a critical review’. Chem Soc Rev 45:1803–1818
Burduel AC, Gherasim O, Grumezescu AM, Mogoantă L, Ficai A, Andronescu E (2018) Biomedical applications of silver nanoparticles: an up-to-date overview. Nanomater Basel 8:681 (Crossref. PMID: 30200373. PMCID: PMC6163202)
Busi S, Rajkumari J, Ranjan B, Karuganti S (2014) Green rapid biogenic synthesis of bioactive silver nanoparticles (AgNPs) using Pseudomonas sp”. ET Nanobiotechnol. https://doi.org/10.1049/iet-nbt.2013.0059
Das VL, Thomas R, Varghese R, Soniya EV, Mathew J, Radhakrishnan EK (2014) Extracellular synthesis of silver nanoparticles by the Bacillus strain CS 11 isolated from industrialized area. 3 Biotech Biotech 4(2):121–126. https://doi.org/10.1007/s13205-013-0130-8
Dhoondia ZH, Chakraborty H (2012) Lactobacillus mediated synthesis of silver oxide nanoparticles. Nanomater Nanotechnol 2:1–7
Durán N, Marcato PD, De Souza GIH, Alves OL, Esposito E (2007) Antibacterial effect of silver nanoparticles produced by fungal process on textile fabrics and their effluent treatment. J Biomed Nanotechnol 3(2):203–208
Eckhardt S, Brunetto PS, Gagnon J, Priebe M, Giese B, Fromm KM (2013) NanoBiosilver: its interactions with peptides and bacteria, and its uses in medicine. Chem Rev 113(7):4708–4754
Fayaz AM, Balaji K, Girilal M, Yadav R, Kalaichelvan PT, Venketesan R (2010) ‘Biogenic synthesis of silver nanoparticles and their synergistic effect with antibiotics: a study against gram-positive and gram-negative bacteria. Nanomedicine 6:103–109
Feng QL, Wu J, Chen GQ, Cui FZ, Kim TN, Kim JO (2000) A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus’. J Biomed Mater Res 52:662–668
Foldbjerg R, Dang DA, Autrup H (2011) Cytotoxicity and genotoxicity of silver nanoparticles in the human lung cancer cell line A549’. Arch Toxicol 85:743–750. https://doi.org/10.1007/s00204-010-0545-5
Gandhi H, Khan S (2016) Biological synthesis of silver nanoparticles and its antibacterial activity. J Nanomed Nanotechnol 7(2):1–3. https://doi.org/10.4172/2157-7439.1000366
Gopinath V, Priyadarshini S, Loke MF, Arunkumar J, Marsili E, MubarakAli D, Velusamy P, Vadivelu J (2017) Biogenic synthesis, characterization of antibacterial silver nanoparticles and its cell cytotoxicity’. Arab J Chem 10:1107–1117
Govindappa M, Thanuja V, Hemashekhar B, Srinivas C, OmaimaNasif AP, Vinay BR (2021) Pomegranate fruit fleshy pericarp mediated silver nano particles possessing antimicrobial, antibiofilm formation, antioxidant, biocompatibility and anticancer activity. J Drug Delivery Sci Tech 61:102289
Hulkoti NI, Taranath TC (2014) Biosynthesis of nanoparticles using microbes-a review. Colloids Surf B Biointerfaces 121:474–483
Jones SA, Bowler PG, Walker M, Parsons D (2004) Controlling wound bioburden with a novel silver-containing Hydrofiber dressing. Wound Repair Regen 12:288–294
Kanchana A, Agarwal I, Sunkar S, Nellore J, Namasivayam K (2011) Biogenic silver nanoparticles from Spinaciaoleracea and Lactuca sativa and their potential antimicrobial activity. Dig J Nanomater Biostruct 6:1741–1750
Karami S, Roayaei M, Hamzavi H, Bahmani M, Hassanzad-Azar H, Leila M, Rafieian-Kopaei M (2017) Isolation and identification of probiotic Lactobacillus from local dairy and evaluating their antagonistic effect on pathogens’. Int j Pharm Investig 7:137–141
Kaviya S, Santhanalakshmi J, Viswanathan B, Muthumary J, Srinivasan K (2011) ‘Biosynthesis of silver nanoparticles using citrus sinensis peel extract and its antibacterial activity. Spectrochim Acta Part A 79:594–598
Khan T, Abbas S, Fariq A, Yasmin A (2018) Microbes: Nature’s cell factories of nanoparticles synthesis. In exploring the realms of nature for nanosynthesis. Springer
Khan I, Saeed K, Khan I (2019a) Nanoparticles: properties, applications and toxicities. Arabian J Chem 12(7):908–931. https://doi.org/10.1016/j.arabjc.2017.05.011
Korbekandi H, Iravani S, Abbasi S (2012) Optimization of biological synthesis of silver nanoparticles using Lactobacillus casei subsp. casei. J ChemTechnolBiotechnol 87:932–937. https://doi.org/10.1002/jctb.3702
Li WR, Xie XB, Shi QS, Zeng HY, Ou-Yang YS, Chen YB (2010) Antibacterial activity and mechanism of silver nanoparticles on Escherichia coli. Appl Microbiol Biotechnol 85:1115–1122
Li X, Xu H, Chen ZS, Chen G (2011) Biosynthesis of nanoparticles by microorganisms and their applications. J Nanomater 3:1–16
Lok CN, Ho CM, Chen R, He QY, Yu WY, Sun H, Tam PK, Chiu JF, Che CM (2006) Proteomic analysis of the mode of antibacterial action of silver nanoparticles. J Proteome Res 5(4):916–924. https://doi.org/10.1021/pr0504079
Morones JR, Elechiguerra JL, Camacho A, Holt K, Kouri JB, Ramírez JT, Yacaman MJ (2005) The bactericidal effect of silver nanoparticles. Nanotechnology 16:2346–2353
Nangia Y, Wangoo N, Goyal N, Shekhawat G, Suri CR (2009) A novel bacterial isolateStenotrophomonasmaltophilia as living factory for synthesis of gold nanoparticles. Microb Cell Factories 8:39
Narayanan KB, Sakthivel N (2010) Biological synthesis of metal nanoparticles by microbes. Adv Colloid Interface Sci 156:1–13. https://doi.org/10.1016/j.cis.2010.02.001
Paul D, Sinha SN (2014) Extracellular synthesis of silver nanoparticles using Pseudomonas Aeruginosa KUPSB12 and Its antibacterial activity. Jordan J Biol Sci 7(4):245–250
Prasad R, Pandey R, Barman I (2016) Engineering tailored nanoparticles with microbes: Quo vadis? Wiley Interdiscip. Rev Nanomed Nanobiotechnol 8:316–330
Priyadarshini S, Gopinath V, Priyadharsshini NM, Mubarak AD, Velusamy P (2013) Synthesis of anisotropic silver nanoparticles using novel strain, Bacillus flexus and its biomedical application. Colloids Surf 102:232–237
Pucciarelli S, Devaraj RR, Mancini A, Ballarini P, Castelli M, Schrallhammer M, Petroni G, Miceli C (2015) Microbial consortium associated with the antarctic marine ciliate euplotesfocardii: an investigation from genomic sequences’. Microb Ecol 70:484–497
Rajesh S, Dharanishanthi V, Kanna AV (2015) Antibacterial mechanism of biogenic silver nanoparticles of Lactobacillus acidophilus. J Exp Nanosci 10:1143–1745. https://doi.org/10.1080/17458080.2014.985750
Ramalingam V, Rajaram R, PremKumar C, Santhanam P, Dhinesh P, Vinothkumar S, Kaleshkumar K (2014) Biosynthesis of silver nanoparticles from deep sea bacterium Pseudomonas aeruginosa JQ989348 for antimicrobial, antibiofilm, and cytotoxic activity. J Basic Microbiol 54:928–936
Ramasamy KP, Telatin A, Mozzicafreddo M, Miceli C, Pucciarelli S (2019) Draft genome sequence of a new Pseudomonas sp Strain, ef1, associated with the psychrophilic antarctic ciliate Euplotesfocardii strain. Microbiol Resour Announc 8:8
Ravindra BK, Rajasab AH (2014) A comparative study on biosynthesis of silver nanoparticles using four different fungal species’. Int J Pharm Pharm Sci 6:372–376
Sahu SC, Zheng JW, Graham L, Chen L, Ihrie J, Yourick JJ, Sprando RL (2014) Comparative cytotoxicity of nanosilver in human liver HepG2 and colon CaCO2 cells in culture. J Appl Toxicol 34:1155–1166. https://doi.org/10.1002/jat.2994
Sastry M, Patil V, Sainkar SR (1998) Electrostatically controlled diffusion of carboxylic acid derivatized silver colloidal particles in thermally evaporated fatty amine films. J Phys Chem b 102(8):1404–1410. https://doi.org/10.1021/jp9719873
Sathishkumar G, Dhivyabharathi B, Prabukumar S, Pugazhendhi A, Rajkuberan Ch, Sivaramakrishnan S (2020) Chrysin-anchored silver and gold nanoparticle-reduced graphene oxide composites for breast cancer therapy, ACS. Appl Nano Mater 3(5):4574–4585. https://doi.org/10.1021/acsanm.0c00630
Shivakrishna P, Ram Prasad M, Krishna G, Singara-Charya MA (2013) Synthesis of silver nano particles from marine bacteria Pseudomonas aerogenosa. Octa j Biosci 1(2):108–114
Van BP, Wells JM, Kleerebezem M (2013) Regulation of intestinal homeostasis and immunity with probiotic lactobacilli. Trends Immunol 34:208–215. https://doi.org/10.1016/j.it.2013.01.005
Vasantharaj S, Sa S, Sripriya N, Jaya G, Thiruvengadam N, Pannerselvam B, Fahad A, Muthiah Sh, Palanisamy S, Arivalagan P (2021) Cytotoxic effects of silver nanoparticles on Ruellia tuberosa: Photocatalytic degradation properties against crystal violet and coomassie brilliant blue. J Envir Che Eng 9:105088
Wang K, Li W, Rui X, Chen X, Jiang M, Dong M (2014) Characterization of a novel exopolysaccharide with antitumor activity from Lactobacillus plantarum 70810. Int J BiolMacromol 63:133–139. https://doi.org/10.1016/j.ijbiomac.2013.10.036
Wang C, Kim YJ, Singh P, Mathiyalagan R, Jin Y, Yang DC (2015) Green synthesis of silver nanoparticles by Bacillus methylotrophicus, and their antimicrobial activity. Artif Cells Nanomed Biotechnol 6:1–6
You I, Kim EB (2020) Genome-based species-specific primers for rapid identification of six species of Lactobacillus acidophilus group using multiplex PCR. PLoS ONE 1:e0230550
Acknowledgements
The authors wish to thank University of Sadat City for funding this study and the Department of Microbial Biotechnology for providing access to a microbial biotechnology laboratory for the experiments. The work in a microbial biotechnology laboratory was supported by University of Sadat City Fund (project ID: 12).
Funding
These studies in Microbial Biotechnology Department were supported by University of Sadat City (USC) with funding through Project No. 12.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mohammed, A.B.A., Hegazy, A.E. & Salah, A. Novelty of synergistic and cytotoxicity activities of silver nanoparticles produced by Lactobacillus acidophilus. Appl Nanosci 13, 633–640 (2023). https://doi.org/10.1007/s13204-021-01878-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-021-01878-5