Abstract
This study investigates the risk to contamination of groundwater in public water supply wells in the Koprivnica-Križevci county (northwest Croatia). Five physicochemical parameters were monitored in all groundwater samples from 2008 to 2017 to identify major differences between the wells, assess temporal variations and understand the capacity for rainfall to alter groundwater pollution loadings. Multivariate discriminant analysis showed statistically significant differences between the six sampled wells based on the analyzed parameters (Wilks' lambda: 0.001; F = 26.2; p < 0.0000). Principal component analysis revealed two significant factors, including factor 1 which explained 32.8% of the variance (suggesting that the quality of the groundwater was mainly controlled by nitrate) and factor 2, accounting for 16.2% of the total variance (which corresponded to KMnO4/oxidizability and to a lesser extent, pH). The time series data showed disparate trends, with nitrate concentrations increasing, whereas pH and KMnO4 decreased, while electrical conductivity and chloride levels remained stable. Although rainfall can impact groundwater pollution loadings through dilution processes in aquifers, the resulting fluctuations in physicochemical parameters are complicated by variations in rainfall events and local topography, as well as from climate change. Therefore, it is important to predict the contamination of groundwater quality in the future using machine learning algorithms using artificial neural network or similar methods. Multivariate statistical techniques are useful in verifying temporal and spatial variations caused by anthropogenic factors and natural processes linked to rainfall. The resulting identified risks to groundwater quality would provide the basis for further groundwater protection, particularly for decisions regarding permitted land use in recharge zones.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The deterioration of groundwater quality is typically derived from the following causal factors: industrial, agricultural, urban (domestic) and overexploitation (Soomro et al. 2014; Miles et al. 1989; Elias 2008; Das Gupta and Puspor 1997; Sing et al. 2021). The type and extent of agriculture, as well as the density of residential land use within specified areas around wells, can be major predictors of the presence of a contaminant. Although agricultural activities are often the dominant cause of widespread contamination, iron or anorganic/organic chemicals from industry may result in point-source pollution (Eckhardt and Stackelberg 1995; Annaporna and Janardhana 2015; Rawat and Tripathi 2015; Gautam et al. 2021). Aside from land use/land cover (Srivastava et al. 2013), the risk of groundwater contamination also depends upon well construction and density (Kalinski et al. 1994; Nemčić-Jurec and Jazbec 2015). Considerable temporal and spatial variations in groundwater chemistry reflect complex soil and groundwater interactions, which largely depend on soil type, soil erosion, weathering of crustal rocks and geology (Yang et al. 2013; Hill and Neal 1997; Nemčić-Jurec et al. 2017; Sing et al. 2009). Alluvial groundwater is especially vulnerable to contamination due to surface water and groundwater interactions and their dynamic relationship. In particular, the distance from the river is a major determinant for groundwater quality in alluvial areas (Negrel et al. 2003; Trivedi et al. 2010).
Variation to groundwater quality is primarily controlled by natural processes, such as atmospheric inputs including runoff, infiltration and precipitation (Trivedi et al. 2010; Machiwal and Jha 2015). Models simulating different climatic time periods have been used to estimate the future impacts of climate change on surface water and groundwater interactions, as well as on groundwater levels (e.g., Scibek et al. 2007). In addition to climate-induced changes, groundwater configuration and fluxes are determined by topography. The relative importance of local flow systems increases with topographic relief. Indeed, topography contributes to regional groundwater movement, dictating rates of groundwater imports and exports to surface water bodies (Condon et al. 2015).
Excluding natural processes, the majority of physicochemical indicators typically measured in groundwaters are of anthropogenic origin. For example, groundwater electrical conductivity (EC) is typically controlled by the ionization of the dissolved inorganic solids and increases with growing concentrations of dissolved minerals in contact with the groundwater (Soomro et al. 2014; Machiwal and Jha 2015; Pu et al. 2011; Rawat et al. 2017). However, contributions from anthropogenic factors (e.g., industrial pollution, sewage irrigation, agriculture) can dramatically increase groundwater EC levels (Annaporna and Janardhana 2015; Soomro et al. 2014; Sing et al. 2015).
Groundwaters occur within different types of aquifers under various geological conditions, with natural factors governing the pH (Zhou et al. 2015), buffered by inputs of carbonate, bicarbonate and carbon dioxide (Rawat et al. 2012). The acidification of soil and groundwater is an ongoing process, which may occur as a result of either natural or anthropogenic processes, such as that occurring from pyrite oxidation in coastal acid sulfate soils (Kinsela and Melville 2004). Furthermore, very acidic groundwaters can solubilize soil components, increasing concentrations of heavy metals and other components into surface and groundwaters (Macdonald et al. 2004). Cultivation and the deposition of atmospheric emissions further exacerbate these processes (Hill and Neal 1997; Knuttson 1994; Anderson and Bowser 1986; Singh and Agrawal 2008).
The major natural sources of organic matter are carbonaceous and nitrogenous substances in the soil and water bodies, along with decaying plant matter which enters groundwater through leaching mechanisms (Oyem et al. 2014). Anthropogenic organic contaminants such as pesticides, pharmaceutical active compounds, drugs, estrogens, personal care products, selected industrial compounds and lifestyle compounds have also been found in rural and urban groundwaters (Jurado et al. 2012). Organic contaminants enter aquifers mainly through wastewater effluents. It is important to mention that compared to the other water bodies (especially rivers), groundwaters are considerably less contaminated, indicative of the natural attenuation capacity of aquifers and infiltration processes (Jurado et al. 2012).
Chloride concentrations in groundwaters also depends on natural processes, however, elevated levels may indicate the presence of organic waste, particularly of animal origin (e.g., agriculture). Increased chloride of 280 mg/l (exceed maximal level prescribed by WHO which is 250 mg/l) concentrations from the discharge of municipal (human sewage, roads) and industrial waste have also been reported (Soomro et al. 2014; Trivedi et al. 2010; Eriksson et al. 2007). Again, alluvial aquifers are particularly vulnerable to the major inorganic anthropogenic inputs, such as chloride, especially in agricultural areas (Negrel et al. 2003; Scanion, et al. 2005). Due to its high solubility as well as the properties of the aquifer (type, position, recharge conditions), chlorides can be rapidly introduced into groundwaters across catchment areas, potentially contributing to pollutant variability in the groundwater (Sakson and Brzezinska 2018).
The degradation of groundwater quality from nitrate has been widely reported both in rural and in urban areas (Elisante and Muzuka 2017; Pociene and Pocius 2005; Pu et al. 2011). Nitrate leaching into the groundwater is related to the soil type, hydrogeology and land use. Well depth is also an important determinant, with previous research that included physicochemical indicators and nitrate concentrations in groundwater assessments showed that higher nitrate levels (up to 186 mg/l) correlated with shallow wells (Nemčić-Jurec and Jazbec 2015; Nemčić-Jurec et al. 2013, 2017) The major sources of nitrate in urban aquifers are related to the disposal of wastewater and solid waste, as well as from industrial activities (Elisante and Muzuka 2017; Jurado et al 2012). Elevated concentrations have been attributed to sanitation in the urban centers and to agricultural activities (Pociene and Pocius 2005; Pu et al.2011). Nitrates have been found in drinking waters from agricultural and residential areas (Nemčić-Jurec and Jazbec 2013), where they have been shown to be derived from both diffuse and point sources (Nemčić-Jurec et al. 2015). Although the contamination of groundwater with nitrate represents a global problem, the contaminant source(s), as well as the spatial and temporal distributions, remain large knowledge gaps in certain areas. Groundwater is an important freshwater resource and a significant moderator of hydrologic processes.
Groundwater configuration and spatial patterns in groundwater flux influence the residence time and location of subsurface contaminants (Condon and Maxwell 2015). The water table aquifer at the location of this study in Podravina (northwest Croatia), is overlain by a highly permeable material, making it susceptible to contaminant infiltration from the land surface (Nemčić-Jurec et al. 2017). Considering its proximity to urban and agricultural areas, and therefore its relevance for drinking and irrigation water quality, it is important to explore and predict the penetration and dynamics of contaminants into the public supply wells. One approach to investigate this issue could be through distinguishing variations between wells coupled with groundwater flow direction and other relevant physicochemical (natural and anthropogenic) water quality parameters.
The primary aim of this research was to assess the risk of groundwater contamination in public supply wells in Podravina, Croatia. Previous research has shown that temporal processes and variations may occur over a multitude of timescales, varying daily through to yearly and beyond, depending on prevailing rainfall (Nemčić-Jurec and Jazbec 2015; Hill and Neal 1997; Nemčić-Jurec et al. 2013). Therefore, the second aim of this paper was to determine the behavior of individual parameters (variability and trends over time) across a longer period (10 years) and to assess the impact of rainfall and floods on their concentration in groundwaters. These findings will provide important information to decision makers (e.g., the government, water utilities) for predicting how future climate and rainfall events may impact upon levels of groundwater pollution. Future research should utilize and expand upon the information presented in this manuscript to predict the likelihood of groundwater contamination using emerging technologies, such as machine learning algorithms (artificial neural network) (Kouadri et al. 2021).
Materials and methods
Study area
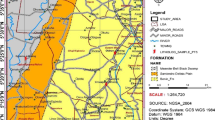
The Ivanščak well field is located in the catchment area of central Podravina, in the Koprivnica-Križevci county of northwestern Croatia (Fig. 1). The study area belongs to the Drava River water catchment and is composed of quaternary gravel–sand aquifer (Duić and Urumović 2007). The area of the old alluvial terraces composed of layers of the Drava River and its tributaries, and terraced sediments. Hydrogeological characteristics are closely related to the aquifer geological materials. The quaternary gravel–sand aquifer reaches a thickness up to 50 m (area where the wells are located) and includes a roof that is composed of dust, sand and clay (10–20 m) with a significant occurrence of “live sand” and marsh loess prevalent in the southern and eastern parts of the river basin district (Nemčić-Jurec et al. 2017). The thickness of this layer (the roof) and its texture strongly influence the permeability of substances from surface layers into the aquifer. The soil is freely drained in the sandstone dominating the catchment.
The aquifer is recharged by infiltration of rainwater through the low permeable cover (into high permeable layers) and by seepage from the Drava River bed in the upstream portion of the study area. The groundwater hydraulic conductivity of the aquifer ranges from 1 to 1.5 × 10–3 m/s, while the effective porosity of the aquifer was estimated to be 0.1. The Ivanščak source water is supplied with groundwater through 6 public supply wells: Z1, B1, B2, B3, B4 and B5 which are arrayed in an exploitation line spaced approximately 125 m apart (Fig. 1c). The aquifer is recharged through two flows: Primary flow (northwest–southeast direction) in the higher proportion recharges the aquifer while secondary flow (southwest–northeast direction) in a lesser proportion recharges the aquifer (Fig. 1). The recharge/discharge dynamics of flows greatly influences groundwater levels and water quality parameters in the wells. Figure 1 shows the locations of piezometers: KPI 1 (E/N-524507.91/5115150.42), KPI 3 (E/N-525055.8/5116474.40), KPI 4 (E/N-525853.63/5116072.73) and P 12 (E/N 524,919.71/5114807.54) which were used to determine the composition of the aquifer flow.
Land use of the study area
The Ivanščak well field supplies Koprivnica city (46°09´40″N; 16°50´14″E) and surrounding settlements. Koprivnica, the biggest city in the county, has a total area of 91 km2 with about 31,000 inhabitants, 77% of which live in the urban and 33% in rural areas (eight surrounding settlements) (“Koprivničke vode”; https://koprivnica.hr/o-koprivnici/misija-vizija-i-ciljevi/).
The local industry includes medium- and large-sized companies, belonging mainly to food, beverage, pharmaceutical, cardboard packing and footwear industries. Farming and animal husbandry is typical across the rural areas. The application of organic and mineral fertilizers across central Podravina is considered to be higher than average, when compared to that applied in other parts of Croatia (Nemčić-Jurec and Jazbec 2015). The diffuse sources of pollution of agricultural origin such as landfills containing organic and mineral fertilizers are situated in the vicinity of the Ivanščak well field (< 100 m). Additionally, the municipal waste landfill for northwest Croatia is located 6 kms downstream from Koprivnica and Ivanščak.
Therefore, the risk of groundwater contamination in the Drava River basin could be expected. Despite the implementation of catchments protection zones shown in Fig. 1c (III. zone—restriction and control zone, II. zone—zone of strict restriction and control and zone I.—zone of strict protection and supervision regime) after 45 years of environmental stress, the deterioration due to urbanization and agricultural land use has resulted in an appreciable decline in groundwater quality. Because of that, groundwater is sampled to identify and quantity the occurrence of the contaminants in groundwater and to investigate the procesess around the contamination event(s). Regular checks are therefore required to identify future risks in time, in order to remedial measures to be taken.
Climate
The investigated area exhibits a moderately warm climate with an average annual temperature range of between 10.9 and 11.2 °C, and has an average rainfall of approximately 803 mm/year. The mean daily maximum temperature is 38.2 °C (in July) and the mean daily minimum is − 22.6 °C (in January). The relative humidity in the study area ranges between 80 and 96% in the rainy and cold season, and between 15 and 20% in the dry and hot season (Nemčić-Jurec et al. 2017).
Sampling and analysis
Groundwater samples (N = 720) were collected from 6 wells (Z1, B1, B2, B3, B4 and B5) at the Ivanščak well field, during their full working capacity, at monthly intervals between 2008 and 2017. Clean 1-L polyethylene bottles were rinsed with sampled water, filled, sealed and labeled after collection. The samples were stored in a portable fridge, in the dark at the temperature of 1–5 °C for transportation to the laboratory, where samples were stored at the same temperature until the analysis (within 24 h) according to the standard method for water sampling (HRN ISO 5667–5:2000).
The samples were analyzed for 5 physicochemical parameters and standard water analysis methods were used (Clesceri et al. 1998; Rice et al. 2012). pH and electrical conductivity (EC) were analyzed by electrometry multimeter method (HACH HQ 40d), nitrate (NO3−) and chloride (Cl−) by spectrophotometry method (SHIMADZU UV-1800) and permanganate oxidation (KMnO4) was analyzed by Titration/Titronic 300 method (SI Analytics).
Statistical data analysis
Descriptive statistics were applied for the classification, interpretation and modeling of the large dataset (5 analyzed parameters in 720 groundwater samples). Data normality was tested by Shapiro–Wilk's W test. Homogeneity of variance for each variable was tested by Levene's test. Differences between groups were assessed by the Newman–Keuls test.
Testing the differences among the six sampled public supply wells was undertaken by multivariate discriminant analysis (MDA). MDA performs forward stepwise analyses of variables (i.e., water quality parameter-independent variable, sampling location-grouping variable) into a model, which, in turn, calculates Wilks' lambda ranging from 0—perfect discrimination among the groups (sampling locations)—to 1—no discrimination among the groups (sampling locations)—along with p-values which correspond to the significance level of the analysis. The analysis also reports the raw and cumulative eigenvalues for all roots (weighted sums of variables), and their p-values, the raw and standardized discriminant (canonical) function coefficients, the structure coefficient matrix (of factor loadings), the means for the discriminant functions and the discriminant scores for each case. Five successive statistically significant roots were extracted, but only the first two were used for data interpretations due to their highest contribution to the total variance.
A chemometric analysis of the groundwater dataset was performed using principal component analysis (PCA) in order to further interpret the complex data set and characterize the public supply wells based on their physicochemical parameters. PCA provided information about the variables responsible for spatial differences in the groundwater quality and showed a degree of correlation among the water quality parameters for all samples.
Trends in the groundwater quality were used for monitoring temporal variations of groundwater quality and assessing the risk of contamination to public supply wells. The linear trends for water quality parameters across the Ivanščak water catchment were determined using box plots of values calculated as an average value of all six wells.
The significance of the sampling time (month and year when a sample was taken from the well; predictor variable) to the water quality parameters was analyzed by multiple regression analysis (MRA); which was significantly different at p < 0.05.
All statistical analysis was performed using the software STATISTICA 12.0 (StatSoft, Inc., USA 2013).
Results and discussion
Groundwater quality assessment: Well position differences
The results of the multivariate discriminant analysis (MDA) testing examining the differences among the six sampling sites are shown in Fig. 2. The results showed statistically significant differences between the six sampling sites (Wilks' lambda: 0.001; F = 26.2; p < 0.0000). All the variables reflecting water quality parameters were used in this model, of which pH, NO3− and Cl− had a statistically significant influence. It was visually apparent that the Z1 well was significantly different to all other wells, while the largest similarity was obtained between the B4 and B5 wells, as well as between the B1 and B2 wells. While wells B1, B2, B3, B4 and B5 were positioned along a line primarily exposed to primary groundwater flow, the more peripheral well, Z1, was likely influenced by secondary groundwater movement (Fig. 1d). The chemical composition of groundwater flows primary (main recharge) and secondary (additional recharge) with respect to the measured parameters in 2017 is shown in Table 1. The intensity of primary and secondary recharge flow influences the accumulation and washing out of anthropogenic pollutants, patterns for which have altered due to recent climate extremes (wet and dry season fluctuations). Well Z1 is in the vicinity of the city and exposed to urban influences, including solid waste dumps, household sullage and/or stagnant surface waters. These inputs may in some settings represent a greater pollution load compared to on-site (point) sources. Domestic waste in this region is often discharged into open ditches or channels and it may represent a significant source of groundwater contamination. Stagnant water pools may directly contaminate groundwater through infiltration or inundation of the sources when rainfall occurs. All of these impacts can contribute to the quality of recharge groundwater (secondary flow direction) outside the sanitary protection zones. Given that previous research has shown chemical pollution in cities and the surrounding areas impacts aquifer water quality (Erah et al. 2002; Ramesh and Elango 2012; Szymanski et al. 2018), it is of future importance to determine the concentration of harmful urban contaminants at the Ivanščak well field.
Descriptive statistics of groundwater physicochemical parameters at public supply wells (Z1, B1, B2, B3, B4, B5) is presented in Table 2. EC is primarily an indicator of environmental influences on groundwater quality, with anthropogenic inputs typically only having a minor contribution. The results indicated that mean EC values varied from 490 µS/cm (B3) to 576 µS/cm (Z1). Overall, groundwater EC values were approximately related to their position in the landscape, in terms of both their average values and their variability (minimum and maximum values). The comparative studies (Oyem et al 2014) of hydrochemical data showed that EC range of 825–1416 µS/cm. The mean values recorded for EC were 1108 µS/cm. Mondal et al (2008) indicates that EC varies from 605 to 5770 µS/cm in groundwater resources. Hence, compared to previous researches, our results showed the average electrical conductivity of groundwater. According to a previous analysis of groundwater quality (by the public supply company, “Koprivnica waters—public water supply”; https://www.kcvode.hr) at the study area, it was determined that certain indicators (e.g., nitrates or EC) were present at higher values in the secondary flow of recharge groundwater than in public supply wells (Table 1). This supports the hypothesis that well Z1 was recharged mainly via the secondary flow pathway.
The average pH values for the studied groundwaters ranged from 6.87 (B3) to 7.07 (Z1), making them suitable for human use (pH from 6.5 to 8.5) and within the limits allowed by the Croatian Drinking water standards (National Newspaper 125 (2013) and WHO recommendations (2011). Again, variations in the pH values were likely due to their relative position, being influenced by urbanization (Z1) and agricultural (B3) inputs. The minimum pH value of 6.54 (at B3) almost reached the lower permissible limit, indicating a deterioration in water quality compared to previous findings, which noted pH values typically > 7.0 (Nemčić-Jurec et al. 2017). Although pH is predominantly influenced by natural conditions, anthropogenic modifications may impact upon future projections. Indeed, previous research has demonstrated the anthropogenic influence on pH values at this study site (Duarte et al. 2013).
Permanganate oxidation (KMnO4) concentrations ranged from 0.43 mg/L (B5) to 0.50 mg/L (B2) and showed a similar trend across all wells with minimal spatial variation. The values were substantially lower than the allowable limits (National Newspaper 125 2013, WHO 2011), suggesting a low organic content in the soil and groundwater. The possible contamination identified by other researchers (Jurado et al. 2012; Kauffman and Chapelle 2010) was found to be either absent or insignificant at this site.
Previous studies into the groundwater quality at the Ivanščak well field identified the presence of elevated nitrate concentrations in the groundwater sampled from shallow wells (Nemčić-Jurec and Jazbec 2015). Our findings support this previous work, with measured average nitrate values varying from 28.2 mg/L (Z1) to 32.7 mg/L (B5). All groundwater samples had nitrate concentrations below the upper permissible limit for drinking water determined by national (National newspaper 125, 2013) or international legislation (WHO 2011). However, the maximum nitrate concentration (42.2 mg/l in the B1 well) exceeded recommended limit value of 25 mg/L (European Union (EU) Council Directive (1991) and was the highest nitrate concentration across all other areas in the county (Nemčić-Jurec et al. 2017). Considering that nitrate is predominantly of anthropogenic origin, the influence of the well position was expected to be a critical determinant for this elevated measurement. The position of the B1 well was strongly influenced by urbanization, industry and agriculture, which collectively contributed to the highest nitrate concentration (Fig. 1d). The variability in nitrate concentrations at B1 was likely a result of the well recharging from two different directions (primary and secondary). Indeed, it would appear that B1 was the well which was most exposed to the combined influence of both groundwater flow directions. Agriculture predominantly contributes to elevated nitrate in the primary water flow direction, with animal feces, as well as the application of manure or fertilizers, representing a significant diffuse source of nitrate contamination over a long period of time. Intensive livestock farming may have a significant impact on nitrate contamination, potentially contributing to a more widespread aquifer contamination at the study site. Urbanization and industry (solid waste dumps, household sullage, stagnant surface waters, stagnant water pools, domestic waste, etc.) affects water quality in the secondary water flow direction (depending on environmental factors) (Scanion et al. 2005; Rawat and Tripathi 2015). Finally, nitrate concentrations at B1 will also be determined by the antecedent ratio of water from both primary and secondary directions, coupled with topographical variations.
In the groundwater samples of the study area, the average chloride concentrations were 23.9 mg/L (Z1), 25.2 mg/L (B1), 19.4 mg/L (B2), 17.3 mg/L (B3), 17.0 mg/L (B4) and 20.0 mg/L (B5). The highest concentrations of chloride in the Z1 and B1 wells were likely related to their position. As outlined above, these wells were subject to pollution pathways from municipal (human sewage, roads, waste) and industrial waste or wastewater via the secondary flow direction (KPI 1 and P 12 piezometers; Table 1). The increase in chloride concentration (exceed maximal levels prescribed by WHO of 250 mg/l) due to discharge of municipal and industrial waste has been reported in earlier studies (Soomro et al. 2014; Trivedi et al. 2010; Eriksson et al. 2007; Liu et al. 2008). Conversely, the impact of agriculture may also contribute to elevated chloride levels via the primary flow direction. However, the measured chloride concentrations were far below regulatory limits, suggesting that anthropogenic impacts were not significant. Previous research (Negrel et al. 2003; Scanion, et al. 2005; Sakson and Brzezinska 2018) has identified that alluvial aquifers are particularly vulnerable to anthropogenically induced changes in major ions, such as chloride, especially in agricultural areas. Considering the alluvial nature of the Podravina region, chloride could easily be introduced into the groundwater, contributing to the variability of the pollutants in the aquifer. This may well be the reason for the large range between minimum and maximum chloride values in our study area (Table 2).
Risk assessment of groundwater contamination in public supply wells
The PCA results, including factor loading values and variance percentage, for the 5 wells are shown in Fig. 3. Eigenvalues provide a measure of the significance of individual factors, i.e., the factor with highest eigenvalue is the most important (Jacintha et al. 2017). The two primary factors extracted, Factors 1 and 2 (F1 and F2), explained 32.8% and 16.2% of the total variance, respectively. F1 correlated predominantly with nitrate concentrations suggesting that the groundwater quality was mainly controlled by this parameter. The high NO3− values highlight the potential role of manure or fertilizer application in agricultural areas or of urbanization (solid waste, sullage, surface water, broken sewage systems) upon aquifer contamination, a point which has been shown previously (Rawat and Tripathi 2015, Nemčić-Jurec et al. 2015). These observations suggest that anthropogenic sources contribute to the elevated NO3− concentrations in the study area. Low (negative) loadings of EC and chloride with F1, confirms that these parameters do not contribute to the variation in nitrate values. The second factor (F2) correlates predominantly with KMnO4 and pH values, indicating a second influence over the groundwater quality related to these parameters. As the degradation of organic material results in a pH decrease due to the formation of organic acids; F2 is may well be a degradation factor. Low (negative) loading of chloride and EC with F2, also suggests that their values were independent of pH or KMnO4. The low loading of EC and chloride would most likely be attributable to the natural solubility of aquifer minerals or natural sources of chloride.
The PCA results revealed that differences in groundwater quality were not restricted to any one or two measured parameters, indicative of both anthropogenic influences and natural mineralization. PCA results confirmed the descriptive statistics that nitrates and pH represent the greatest risk (as concentrations reach regulatory limits) for the deterioration of groundwater quality in public supply wells.
In order to cover all the geochemical processes affecting groundwater quality, groundwater monitoring was conducted for 10 years and the risk of contamination was assessed. The data collected from the study area and the results of current trends are shown in Fig. 4. Values used for the linear trend analysis represent mean concentrations measured monthly in all six wells for each of the water quality parameters.
The study area time series showed an increasing trend in nitrate, decreasing pH and permanganate oxidation, and steadily average concentrations for EC and chloride. Based on the theoretical assumptions and the temporal analysis as well as the variations and parameter trends (Hill and Neal 1997), the groundwater quality can be considered as predominantly natural (EC, pH, permanganate oxidation) and anthropogenic (nitrate, chloride) (Soomro et al. 2014; Nemčić-Jurec and Jazbec 2015; Nemčić-Jurec et al. 2013). An increasing trend of nitrate usage across the study area is a common response to increased human activities, resulting in human alteration of biogeochemical cycles, which has been demonstrated in other areas (Elisante and Muzuka 2017; Duarte et al. 2013). NO3− is very soluble in water and it is easily leached from soils into the groundwater. The elevated levels of nitrate in the groundwater may well have led to soil acidification. According to Elisante and Muzuka (2017), a significant negative correlation between the nitrate concentration and pH indicates the mineralization and nitrification of the organic nitrogen from animal manure. Our results show a negative correlation between pH (decreasing trend) and NO3− (increasing trend) over time. Given that the wells in the study area have been utilized for agricultural purposes for over 45 years, such results were largely expected. In terms of the study area, nitrate concentrations were lower (24 mg/L; KPI 3 and KPI 4 piezometers) from the primary recharge direction (west–northwest), when compared to average values at the public supply wells (sampling sites). Conversely, the groundwater from the secondary flow direction (southwest) nitrate concentrations were higher (39–51 mg/L; P 12 and KPI 1 piezometers) (Table 1). Consequently, the average nitrate concentration in the public supply wells was the result of mixing groundwaters from both directions. Considering that the secondary flow comes from the direction of Koprivnica city, the long-term effect of urbanization outside the sanitary protection zones is visually apparent (Fig. 1c).
The decreasing trend in permanganate oxidation (Fig. 4) during the study period may be the result of intensive agricultural activity, removal of crops and retention of soil organic matter and organic acids. The increasing use of mineral fertilizers instead of organic fertilizer (manure) at the study site, may also contribute to the reduction of organic matter in groundwater. Given that the concentrations of organic matter were low in all wells, even if there was an anthropogenic impact, they would appear to be insignificant toward the KMnO4 parameter.
Rainfall and sampling time influence on groundwater quality
Our results showed that a marked increase of nitrate, coupled with a decrease in pH and KMnO4 occurred in 2014 and 2015. In September 2014, the amount of rainfall was 38% (~ 1,300 mm/year) above the rainfall annual average in the study area (Crometeo 2014). It is conceivable that the more intense trends during this period were associated with the enhanced infiltration of contaminants (esp. nitrate) into the groundwater which consequently decreased pH values. Additional variability in pH values might well occur during perennial fluctuations in the climate. For example, it has been widely demonstrated that extremely low groundwater levels following dry periods promotes sulfide oxidation which increases the acidification of associated groundwaters (Knuttson 1994). Vidyalakshm et al. (2013) have shown that during heavy rainfall events, nutrients enter aquatic ecosystems more quickly, posing a higher risk for drinking water quality.
However, individual indicators behave differently during rainfall. The significantly decrease in KMnO4 (organic matter) after elevated rainfall may also be indicative of aquifer dilution processes. The influence of elevated rainfall and associated flooding was evident during 2014–2015, when KMnO4 concentrations were at their lowest. Previous research has shown that a decrease in organic matter concentrations could be accelerated during heavy rainfalls (Oyem et al. 2014; Sakson and Brzezinska 2018; Mondal et al. 2008).
Groundwater EC and chloride concentrations showed inconsistent trends over the study period (Fig. 4). Chloride concentrations in the groundwater suggested the presence of organic waste, which could have contributed to the variability observed in chloride concentrations during rainfall events. As in previous studies (Pu et al. 2011; Mondal et al. 2008), it is obvious that the low concentrations of chloride recorded during 2014 and 2015 were connected to the heavy rainfall, causing dilution of the groundwater during flooding.
Diffuse sources were the likely major contributor to nitrate in groundwaters (fertilizers from agriculture and urbanization), which were leached from the soil during periods of high rainfall. Intuitively, the behavior of parameters such as nitrate depends on their solubility and leaching rate during precipitation, coupled with the amount of pollutant which is stored in the soil within the aquifer catchment. Therefore, establishing direct links between rainfall and parameters is problematic, and as such, no correlation was observed in our data. Furthermore, studies into the connections between topography and climate upon groundwater fluxes have shown that groundwater configuration is controlled by geology and climate, which influence the residence time and location of subsurface contaminants (Condon and Maxwell 2015).
Investigations were also made into the time dependence (month and year of sampling) on the groundwater quality, analyzed by multiple regression analysis (MRA). The correlations among the groundwater quality and the predictor variable sampling time are shown in Table 3. The predictor variable had a significant influence on the parameters: electrical conductivity (β = 0.33) and chloride concentrations (β = 0.23). Based on the values of the beta coefficients and their levels of significance, it would appear that sampling time had a strong influence on the permanganate oxidation levels (β = 0.17), whereas the values for nitrate (β = 0.13) and pH (β = 0.11) were less dependent on sampling time. This supports the previous assertion that nitrate concentrations and pH value were more dependent on environmental factors such as pollution sources and land use, pollution distance and rainfall. MRA confirmed that the duration of monitoring was a very important factor in the assessment of environmental impacts (e.g., rainfall) on overall groundwater quality.
Water quality prediction
An evaluation of aquifer water quality is critical to ensure the provision of safe drinking water and has a direct impact on public health and the environment. Therefore, it is important to assess and predict the contamination of groundwater quality in the future using machine learning algorithms (artificial neural network, ANN) and similar methods. Some studies have investigated the performance of artificial intelligence techniques for predicting water quality components. For example, it was found that using a group method of data handling (GMDH) and support vector machine (SVM) model were more reliable in comparison with ANN (Haghiabi et al. 2018; Condon and Maxwell 2015). It is likely that similar models will be the subject of further research with the aim of predicting water quality across sites such as the one studied here.
Conclusion
Excess chemical and pollution loadings in groundwater is a serious problem. Understanding spatial variations in contaminant distribution is an important first step for assessing pollution loading at the study site. The highest difference were found in well Z1, which was subject to secondary recharge flows. This well was closest to urban areas as well as being subject to agricultural inputs, placing it at high risk of contamination. The concentration of physicochemical parameters in public supply wells generally reflected a balance of different water sources, soils and anthropogenic activities. Overall, the PCA method revealed that groundwater quality depended primarily on nitrate concentrations that were found to increase over the study period (from 2008 to 2017). A decreasing trend for both pH and KMnO4 indicated the acidification of groundwater via degradation of organic matter. Chloride and electroconductivity values over the study period were variable, with no definitive trends evident. These parameters were most likely impacted upon by dilution due to rainfall as well as changes in land use and anthropogenic activities. The increasing prevalence of both agricultural and non-agricultural activities suggests that there is a possibility for even greater contamination in the future, elevating nitrate concentrations and/or decreasing pH values. The risk to contamination in the alluvium aquifer was deemed particularly high. If activities in agricultural and non-agricultural sectors (urbanization and industry) are not minimized, then risks will increase. Climate change and global warming, particularly the increasing prevalence of intense rainfall events, affect contaminant loading and infiltration processes from soil to groundwater. Understanding the impact of rainfall, climate change and topography on groundwater quality is an important step toward ensuring the sustainability of future groundwater resources. Therefore, it is important to assess and predict the contamination of groundwater quality in the future using machine learning algorithms (artificial neural network) and similar methods. Multivariate statistical techniques have been applied to evaluate groundwater quality, with the results useful for verifying temporal and spatial variations caused by anthropogenic factors and natural processes linked to rainfall. Our results contribute to the decision-making processes in terms of the land use, groundwater quality protection and resource management, which are collectively aimed at reducing the health risk of the population. As such, our research should be of value to other studies using similar analytical tools aimed at controlling source water contamination.
References
Anderson MP, Bowser CJ (1986) The role of groundwater in delaying lake acidification. Water Resour Res 22(7):1101–1108
Annaporna H, Janardhana MR (2015) Assessment of groundwater quality for drinking purposes in rural areas surrounding a defunct cooper mine. Aquat Procedia 4:685–692
Clesceri LS, Greenberg AE, Eaton AD (1998) Standard Methods for the examination of water and waste water, 20th edn. American Public Health Association, Washington
Condon LE, Maxwell RM (2015) Evaluating the relationship between topography and groundwater using outputs from a continental-scale integrated hydrology model. Water Resour Res. https://doi.org/10.1002/2014WR016774-021-00102-3
Crometeo (2014) https://www.crometeo.hr/forum/viewtopic.php?f=50&t=9579
Das Gupta A, Puspar RO (1997) Sustainable groundwater resources development. Hydrol Sci J 42(4):565–582
Duarte CM, Hendriks IE, Moore TS, Olsen YS, Steckbauer A, Ramajo L, Cartensen J, Trotter JA, McCulloch M (2013) Is ocean acidification an open-ocean syndrome? Understanding antropogenic impacts on seawater pH. Estuaries Coasts 36:221–236
Duić Ž, Urumović K (2007) Influence of Legrad threshold structure on hydrogeological characteristics in Koprivnica area. Min Geol Petrol Proc 19:1–10
Eckhardt DAV, Stackelberg PE (1995) Relation of ground-water quality to land use on Long Island. New York Ground Water 33(6):1019–1995
Elias S (2008) Over-exploitation of groundwater resources and their environmental and socio-economic implications: case of Jordan. Water Int 33:55–68
Elisante E, Muzuka ANN (2017) Occurrence of nitrate in Tanzanian groundwater aquifers. A Review Appl Water Sci 7(1):71–87
Erah PO, Akuijeze CN, Oteze GE (2002) The quality of groundwater in Benin City: a baseline study on inorganic chemicals and microbial contaminants of health importance in boreholes and open wells. Trop J Pharm Res 1(2):75–82
Eriksson E, Baun A, Scholes L, Ledin A, Ahlman S, Revit M, Noutsopoulos C, Mikkelsen PS (2007) Selected stormwater priority pollutants-a European perspective. Sci Tot Environ 383(1–3):41–51
European Union(EU) (1991) Council Directive 91/676/EEC of 12 December 1991 concerning the protection of waters against pollution caused by nitrates from agricultural sources. Off J 375:0001–0008
Gautam S, Singh SK, Rawat KS (2021) Intrinsic vulnerability evaluation of groundwater nitrate pollution along a course of the Subarnarekha river in Jharkhand India. Water Conserv Sci Eng. https://doi.org/10.1007/s41101-021-00102-3
Haghiabi AH, Nasrolahi AH, Parsaie A (2018) Water quality prediction using machine learning methods. Water Qual Res J 53(1):3–13
Hill T, Neal C (1997) Spatial and temporal variation in pH, alkalinity and conductivity in surface runoff and groundwater for the upper river Severn catchment. Hydrol Earth Syst Sc 1(3):697–715
HRN ISO 5667–5:2000 Water quality-Sampling-Part 5: Guidance on sampling of drinking water and water used for food and beverage processing, https://www.kcvode.hr
Jacintha TGA, Rawat KS, Mishra A, Singh SK (2017) Hydrogeochemical characterization of groundwater of peninsular Indian region using multivariate statistical techniques. Appl Water Sci 7:3001–3013
Jurado A, Vazquez-Sune E, Carrera J, Lopez de Alda M, Pujades E, Barcelo D (2012) Emerging organic contaminants in groundwater in Spain: a review of sources, recent occurrence and fate in a European context. Sci Total Environ 440:82–94
Kalinski RJ, Kelly WE, Bogardi I, Ehrman RL, Yaniamoto PD (1994) Correlation between DRASTIC vulnerabilities and incidents of VOC contamination of municipal wells in Nebraska. Ground Water 32(1):31–34
Kauffman LJ, Chapelle FH (2010) Relative vulnerability of public supply wells to VOC contamination in hydrologically distinct regional aquifers. Ground Water Monit Remediat 30(4):54–63
Kinsela AS, Melville MD (2004) Mechanisms of acid sulfate soil oxidation and leaching under sugarcane cropping. Australian J Soil Res 42:569–578. https://doi.org/10.1071/SR03084
Knuttson G (1994) Acidification effects on groundwater – prognosis of the risks for the future. Future Ground Water Resour Risk 222:3–17
Kouadri S, Elbeltagi A, Islam ARMT, Kateb S (2021) Performance of machine learning methods in predicting water quality index based on irregular data set: application on Illizi region (Algerian southeast). Appl Water Sci. https://doi.org/10.1007/s13201-021-01528-9
Liu CQ, Lang YC, Satake H, Wu J, Li SL (2008) Identification of anthropogenic and natural inputs of sulfate and chloride into the karstic ground water of guiyang, SW China: combined δ37Cl and δ34S approach. Environ Sci Technol 42:5421–5427
Macdonald BCT, Smith J, Keene AF, Tunks M, Kinsela A, White I (2004) Impacts of runoff from sulfuric soils on sediment chemistry in an estuarine lake. Sci Tot Environ 329:115–130. https://doi.org/10.1016/j.scitotenv.2004.02.016
Machiwal D, Jha MK (2015) Identifying sources of groundwater contamination in a hard-rock aquifer system using multivariate statistical analyses and GIS-based geostatistical modelling techniques. J Hydrol Reg Stud 4:80–110
Miles JC, Lloyd JW, Witt JA (1989) An assessment of the effect of urban development on groundwater levels in a chalk aquifer. Environ Geol Water Sci 14(3):187–194
Mondal NC, Singh VS, Saxena VK (2008) Improvement of groundwater quality due to fresh water ingress in Potharlanka island, Krishna delta. India Environ Geol 55(3):595–603
Negrel P, Petelet-Giraud E, Barbier J, Gautier E (2003) Surface water-groundwater interactions in an alluvial plain: chemical and isotopic systematics. J Hydrol 277(3–4):248–267
Nemčić-Jurec J, Jazbec A (2015) Point source pollution and variability of nitrate concentrations in water from shallow aquifers. Appl Water Sci 7:1337–1348
Nemčić-Jurec J, Konjačić M, Jazbec A (2013) Monitoring of nitrates in drinking water from agricultural and residential areas of Podravina and Prigorje (Croatia). Environ Monit Assess 185:9509–9520
Nemčić-Jurec J, Singh SK, Jazbec A, Gautam SK, Kovač I (2017) Hydrochemical investigations of groundwater quality for drinking and irrigational purposes: two case studies of Koprivnica-Križevci County (Croatia) and district Allahabad (India). Sust Water Resour Manage. https://doi.org/10.1007/s40899-017-0200-x
National Newspaper 125 (2013) Regulation about the parameters of assessment and methods of analysis of water for human consumption. Zagreb, Croatia
Oyem HH, Oyem IM, Ezeweali D (2014) Temperature, pH, electrical conductivity, total dissolved solids and chemical oxygen demand of groundwater in Boji-BojiAgbor/Owa area and immediate suburb. Res J Environ Sci 8(8):444–450
Pociene A, Pocius S (2005) Relationship between nitrate amount in groundwater and natural factors. J Environ Eng Landscape Manage 13(1):23–30
Pu JB, Yuan DX, Hu ZY, Yang PH, Gou PF, He QF, Wang ZJ (2011) High-resolution research on the NO3-changes of karst groundwater and its responses to the outside environmental variations. Huan Jing Ke Xue 32(3):680–686
Ramesh K, Elango L (2012) Groundwater quality and its suitability for domestic and agricultural use in Tondiar river basin, Tamil Nadu. India Environmental Monitoring and Assessment 184(6):3887–3899
Rawat KS, Tripathi VKJ (2015) Hydro-chemical survey and quantifying spatial variations of groundwater quality in Dwarka, sub-city of Delhi. India J Inst Eng (india) 96(2):99–108
Rawat EKS, Mishra AK, Sehgal VK, Tripathi VK (2012) Spatial variability of ground water quality in Mathura district (Uttar Pradesh, India) with geostatistical method. Int J Remote Sens Appl 2:1–9
Rawat KS, Mishra AK, Singh SK (2017) Mapping of groundwater quality using Normalized Difference Dispersal Index of Dwarka sub-city at Delhi National Capital of India. ISH J Hydraul Eng 23(3):229–240. https://doi.org/10.1080/09715010.2016.1277795
Rice EW, Baird RB, Eaton AD, Clesceri LS (2012) Standard Methods for the examination of water and waste water, 22nd edn. American Public Health Association, Washington.
Sakson G, Brzezinska A (2018) Emission of heavy metals from an urban catchment into receiving water and possibility of its limitation on the example of Lodz city. Environ Monit Assess 190:281
Scanion BR, Reedy RC, Stonestrom DA, Prudic DE, Dennehy KF (2005) Impact of land use and land cover change on groundwater recharge and quality in the southwestern US. Glob Change Biol 11(10):1577–1593
Scibek J, Allena DM, Cannon AJ, Whitfield PH (2007) Groundwater-surface water interaction under scenarios of climate change using a high-resolution transient groundwater model. J Hydrol 333(2–4):165–181
Singh A, Agrawal M (2008) Acid rain and its ecological consequences. J Environ Biol 29(1):15–24
Singh S, Singh C, Kumar K, Gupta R, Mukherjee S (2009) Spatial-temporal monitoring of groundwater using multivariate statistical techniques in Bareilly district of Uttar Pradesh. India J Hydrol Hydromech 57:45–54
Singh SK, Srivastava PK, Singh D, Han D, Gautam SK, Pandey AC (2015) Modeling groundwater quality over a humid subtropical region using numerical indices, earth observation datasets, and X-ray diffraction technique: a case study of Allahabad district. India Environ Geochem Health 37(1):157–180. https://doi.org/10.1007/s10653-014-9638-z
Singh SK, Bharose R, Nemčić-Jurec J, Rawat KS, Singh D (2021) Irrigation water quality appraisal using statistical methods and WATEQ4F geochemical model. Agricultural water management. Elsevier, pp 101–138
Soomro A, Siyal A, Saffar MM, Wadhayo GA, Latif QA (2014) Seasonal variation of groundwater quality assessment for irrigation and drinking purpose in Phuleli canal command area (Sindh). Pakistan Int Water Technol J 4(4):222–237
Srivastava PK, Singh SK, Gupta M, Thakur JK, Mukherjee S (2013) Modeling impact of land use change trajectories on groundwater quality using remote sensing and GIS. Environ Eng Manage J 12:2343–2355
Statistica 12.0 (Electronic version): StatSoft, Inc (2013). Electronic statistics textbook. Tulsa, OK: StatSoft. WEB: https://statisticasoftware.wordpress.com/2013/05/15/statsoft-releases-version-12-of-statistica-software/. Accessed 7. May, 2013
Szymanski K, Janowska B, Izewska A, Sidelko R, Siebielska I (2018) Method of evaluating the impact of landfill leachate on groundwater quality. Environ Monit Assess 190:415
Trivedi P, Bajpai A, Thareja S (2010) Comparative study of seasonal variation in physico-chemical characteristics in drinking water quality of Kanpur, India with reference to 200 MLD filtration plant and ground water. Nat Sci 8(4):11–17
Vidyalakshm R, Balan B, Prashanthi D, Paneer S, Benedict R (2013) Determination of land use stress on drinking water quality in Tiruchirappalli, India using derived indices. Water Qual Expos Hea 5(1):11–29
WHO (2011). Guidelines for drinking-water quality, 4th ed. World Health Organization (WHO), Geneva.
Yang PH, Yuan DX, Ye XC, Xie SY, Chen XB, Lin ZQ (2013) Sources and migration path of chemical compositions in a karst groundwater system during rainfall events. Chinese Sci Bull 58(20):2488–2496
Zhou X, Shen Y, Zhang Y (2015) Hydrochemistry of the natural low pH groundwater in the coastal aquifers near Beihai, China. J Ocean U China 14: 475–483, https://koprivnica.hr/o-koprivnici/misija-vizija-i-ciljevi/
Funding
The author(s) received no specific funding for this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The author(s) comply with ethical policies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nemčić-Jurec, J., Ruk, D., Oreščanin, V. et al. Groundwater contamination in public water supply wells: risk assessment, evaluation of trends and impact of rainfall on groundwater quality. Appl Water Sci 12, 172 (2022). https://doi.org/10.1007/s13201-022-01697-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-022-01697-1