Abstract
The quality of groundwater standards in Muttom–Mandaikkadu coastal stretch is the focus of the present study, whose coastal aquifers are particularly at risk due to intrusion of marine water. Thirty groundwater samples were scrutinized for the assessment of physical and chemical parameters during January and June. Hydrochemical characteristics were spatially depicted to understand the spatial variations such as (pH, EC, TDS, Na+, K+, Ca2+ and Mg2+, SO42−, HCO3−, and Cl−). Drinking water quality index based on those 11 parameters and irrigation water quality index based on EC, Na%, sodium adsorption ratio and permeability index was used to assess the water quality for drinking and irrigation, respectively. These results demonstrate that dominant hydrochemical facies for groundwater in both months are Na-K-Cl-SO4 type and Ca-Mg-Cl-SO4 type. The USSL diagram endorses that most of the water samples belong to low-medium salinity with low sodium hazards. Cl−/HCO3− ratio indicates that the majority of the samples show low to moderate seawater intrusion in the study area. Additionally, six vertical electrical sounding measuring points (Schlumberger array) were carried out in order to determine the number of the underlying layers, aquifer depths and their thicknesses as well as its influence by the marine water. The geophysical self-potential measurements suggest that the groundwater in the Manavalakurichi area is prone to contamination by seawater intrusion, confirmed by the use of Schlumberger vertical electrical sounding. For better understanding, the subsurface layers were shown in a 2D model using the constructed geoelectrical cross section.
Similar content being viewed by others
Introduction
In India, groundwater suites the drinking and domestic needs of nearly 90% of the rural and 30% of the urban population (Bhunia 2020). Usually, groundwater is considered a safer resource than surface water. Still, several factors such as uncontrolled discharge of industrial, agricultural and domestic effluents, improper land use, certain geological formations, shortage of rainfall, and low infiltration rate affect groundwater quality (Adimalla 2019). Increasing demand for water during these days makes the problem of water contamination a major concern and has also caused water shortage in different parts of the world. According to WHO reports, 65% of rural and 36% of urban inhabitants do not consume safe drinking water in India (Sexenal and Mishra 2011).
On the other hand, deprived quality of groundwater also affects agriculture as it constitutes 53% of the country's irrigation potential (Wilcox 1948; Hem 1991; FAO 2003; WHO 2004). If the groundwater in the aquifer is contaminated, it persists for several years and it is not easy to retrieve the quality (Jakeman et al. 2016). So, there is a need to monitor and assess the quality of groundwater. To understand the suitability for drinking as well as irrigation purposes, a hydrochemical assessment of the groundwater must be carried out (Nakhaei et al. 2016). Figuring of several physico-chemical criteria like pH, electrical conductivity (EC), total dissolved solids (TDS), Ca2+, Mg2+, K+, Na+, Cl−, HCO3− and SO42− and their comparison with existing firm standards disclose the quality of water and its suitability for drinking and irrigation needs. Several hydrochemical appraisal studies were carried out in recent days worldwide as well as India to understand the nature of the groundwater (Srinivas et al. 2014; Tiwari et al. 2017; Adimalla et al. 2018; He et al. 2018; Li et al. 2018; Chidambaram et al. 2018; Kumari and Rai 2020; Shaikh et al. 2020). Golchin and Azhdary Moghaddam (2016) in their study named hydrogeochemical characteristics and groundwater quality assessment in Iranshahr plain aquifer, Iran, obtained results such that the groundwater salinity hazard is low to high, but the Na hazard is low to medium, and in regard to irrigation water, the quality is low to medium. Ghalib (2017) had carried out a study that focused on assessing the groundwater quality of the shallow aquifer in the northeastern Wasit Governorate, Iraq. The studied groundwater samples were found to be oversaturated with carbonate minerals and undersaturated with evaporated minerals. A comparison of groundwater quality concerning drinking water standards showed that most groundwater samples were unsuitable for drinking purposes. A study was carried out by Mostafa et al (2017) to understand the hydrogeochemistry and groundwater quality in the Rajshahi City of Bangladesh. This study demonstrated that the rock–water interaction was the major geochemical process controlling groundwater chemistry in the study area. The study results also revealed that the groundwater quality in Rajshahi City area was of great concern and not suitable for human consumption without adequate treatment. The research paper by Saraswat et al (2018) provides an exploratory investigation in the hydrogeochemical characteristics of groundwater and thereby assesses the suitability of groundwater as an alternative and reliable resource for public water supply in the Indian city of Surat. The results of this study outline the unsustainability of groundwater for direct consumption, especially without any improved on-site water treatment, but it is appropriate for irrigation purposes. Mahanta et al (2019) had authored a study that aims to approach the analysis of groundwater samples of the Western part of Odisha, i.e., Maneswar Block of Sambalpur District, to assess the quality of groundwater in terms of drinking and irrigation purposes.
Nowadays, geographical information system (GIS) has been widely used as an effective tool to integrate various databases and develop solutions for assessing water quality, reducing water resources problems, preventing flooding and understanding the natural environment (Venkatramanan et al. 2019). Precautionary indicators of potential environment-related health problems include groundwater quality distribution maps and risk assessment maps (Arnous et al. 2011; Gnanachandrasamy et al. 2015; Ameen 2019; Li et al 2019).
In the Manavalakurichi and enclosing areas of India's southern apex, a total of 30 groundwater samples have been acquired for the current study. This study's primary focus is to delineate the level of spoilation in groundwater in the Muttom–Mandaikkadu coastal stretch. There is a possibility of groundwater deterioration due to saline water intrusion as the study area's southern boundary is surrounded by sea. So, the present study finds its importance in delineating the pollution in groundwater using water quality analysis.
Study area
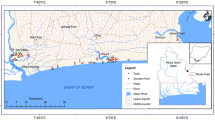
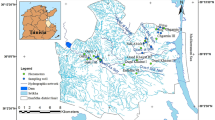
The study area Muttom to Mandaikkadu coastal zone located within the Kanyakumari district in the southern part of Tamil Nadu. Figure 1 shows the study area map with geology. The climate is humid with extremely hot summers; the southern part of the study area is bounded by the Indian Ocean and in the west by the Arabian Sea. The area being part of the west coast enjoys a typical monsoonal climate and receives an average annual rainfall of about 1465 mm. The maximum rain is received during April, May, June, and July (southwest monsoon) and in October, November (northeast monsoon). Rainfall forms the only source of aquifer replenishment of these aquifers. Physiographically, the region of study is almost outstretched with a gentle slope, generally facing south. Valliyar is the only river leading in the study area, and the drainage design is sub dendritic, a seasonal river. Manavalakurichi is one of the familiar coastal sectors for its mineral deposits. Several mining zones under the authority of Indian Rare Earth Limited (IREL) are present here (Srinivas et al. 2015). The typical temperature within the study area ranges between 22.8 °C and 33.6 °C. From the geological perspective, most of the part in the study area underlies Quaternary fluviomarine deposits. The Western Ghats Mountain occupies the entire north and northwestern region of the district with a maximum elevation of 1658 m. Sand dunes (Teri sands) and marshy swamps typically cover the coastal tract of the area. The dominant soil types of the study area are coastal sand, red soil, brown soil, and red lateritic soil.
The area is underlain by an Archaean Metamorphic Complex rock, comprising mainly gneisses and charnockites Pegmatite intruded and laterite cappings and quaternary deposits. In a geological perspective, the coastal part in the study area underlies Quaternary fluviomarine deposits. Borehole lithology reveals that the aquifer material consists of fine to coarse grain sand, sandy soil, clay sand and small lens patches of clay (Thamarai 2014). In the fluvial and shallow marine environments, the clay that occurs as thin lenses or small patches surrounded the charnockites and these charnokites that exist under this sandy formation act as impermeable strata. The charnockite group of rocks is well exposed around Muttom, Manavalakurichi and the Eastern part of the study area. The rock shows varying composition from acid to ultramafic. The topography is mild at places in the coastal belt, not exceeding 60 m height from sea level, where crystalline rocks are highly weathered to laterite (GSI 2005). Weathering is relatively higher in granite gneissic rock rather than charnockite.
The entire Kanyakumari to Colachel area is covered by hard rock formations like charnockite and gneisses. The groundwater occurrence is limited to only the weathered mantle of the hard rock. The weathered layer thickness generally ranges from 10 to 35 m below ground level. The groundwater occurrence is also limited from 1 to 10 m above mean sea level (MSL) (Perumal 2008). The study area's general water level varies from 3 to 25 m below ground level (IWS 2000). The shallow alluvial aquifers along Thamirabarani and Valliyar rivers serve as significant drinking water irrigation development for the Kanyakumari district. Alluvium occurs as the upper layer and is characterized by sand, gravel, and sandy clay with thickness ranging from 1 to 20 m in the study area. The alluvium and weathered charnockites function as an unconfined aquifer system (Perumal et al. 2010). The discharge from the aquifer is mainly by the withdrawal of water by pumping for domestic supply, mostly for drinking purposes, and for industries, both private and government agencies.
Materials and methods
Thirty groundwater samples were acquired from dug and bore wells in the study area during January and June 2018. The samples were scrutinized to interpret the chemistry variations of water quality and interpret observed groundwater anomalies in the region. Specimens were obtained in high-density 1L polyethylene bottles. The acquired samples were subjected to evaluating water quality criteria such as pH, electrical conductivity (EC), major cations, and anions. In situ measurements were taken for pH, electrical conductivity (EC), and total dissolved solids (TDS) with portable water quality analysis instruments HANNA (HI-9828, USA). Distinct titration procedures valued the total hardness (TH), major cations, and anions. Simultaneously, the sodium and potassium were analyzed using a Flame photometer instrument (DEEP VISION, Model -381).
The amount of sulfate ions was found using the UV–Visible photometer. The spatial analyst tool is used to draw maps that alleviated the spatial analysis of hydrological parameters to describe the water quality of the study area. The water quality index analysis has been done with the help of the Canadian Water Quality index (CWQI) programmed excel software (John-Mark 2006). ArcGIS (version10.1) software was used for spatial analysis of various physico-chemical factors. An inverse distance weighted (IDW) technique was used to interpolate the data spatially and enumerated the value for each grid node by inspecting the encompassing data points that lie within a user-defined search area (Burrough 1998).
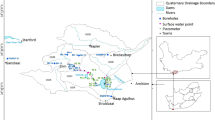
The self-potential signals are naturally occurring electric fields measured at the ground surface (Naudet et al. 2003) using gradient electrode configuration with a 4-m interval. The non-polarizing electrodes P1, P2 (Fig. 2) are filled by copper sulfate crystals and copper sulfate solution. The equipment used to measure the self-potential was a WDDS-2 resistivity meter, accompanied by two non-polarizable electrodes and connecting cables. A total of twenty-four points was collected throughout the study area using the Self-potential gradient method.
The vertical electrical sounding (VES) is carried out at 4 locations using the Schlumberger method by spacing the current electrode (AB) up to a maximum of 200 m in the study area. As the electrode spacing increases with unearthing the depth of investigation, it will always be less than the electrode spacing. The final results are produced by interpreting the observed data using one-dimensional software IPI2win.
Results and discussion
Drinking water quality
The statistical parameters (minimum, maximum and average) that help to understand and relate the concentration of different physico-chemical parameters such as pH, electrical conductivity (EC), total hardness (TH) and total dissolved solids (TDS) and major ions (Ca2+, Mg2+, Na+, K+, Cl−, HCO3−, SO42−) are given in Table 1. The number of samples from both months, which exceeds the standards for the quality of drinking water given by the World Health Organization (WHO 2011) and Bureau of Indian Standards (BIS 2000), is presented in Table 2.
In the study area, the pH values ranged between 5.5 and 7.8 with an average of 6 and from 5.4 to 7.2 with an average of 6.1 during June and January, respectively. Figure 3a shows the spatial distributions of pH for both the months in the study area. This indicates that the groundwater of the Mandaikkadu area is acidic (pH value of samples < 6.0) in nature. The presence of low-pH laterite soil is responsible for the acidity of groundwater in the Mandaikadu area. The electrical conductivity (EC value) varies from 93 to 2022 (µS/cm) in January, and it varies from 82 to 2163 (µS/cm) during June as shown in Fig. 3b. As per WHO 2011 standard, few water samples exceed the limit for drinking during January and June. Higher EC content in the samples endorses the enrichment of salts in the groundwater (Zhang et al. 2019). In the study area, the TDS value ranged from 60 to 1294 mg/l during January and varied from 53 to 1384 mg/l during June (Fig. 3c). As a result of saltwater intrusion and anthropogenic activities, the TDS value is also higher than the permitted value. It is noted that the complete Manavalakurichi area has recorded a high value of EC and TDS.
Following the value of total hardness, groundwater is classified according to Sawyer and McCarthy DL 1967 in Table 3 and the distribution is shown in Fig. 3d. During January, 57% of samples and 47% of samples fall in the hard to very hard water category during June. The presence of calcium-bearing minerals, dissolution of sulfate and carbonate minerals in sediments and anthropogenic activities adds hardness in the study area (Ayman and Mohamed 2011). Calcium and magnesium are the most abundant components in the surface and groundwater that prevail mostly as bicarbonates and in the form of sulfate and chloride in small amounts (Kumar et al 2015). The concentration of calcium varied between 2 to 72 mg/l with an average of 27 mg/l in January (Fig. 4a). Manavalakurichi samples show that the Calcium level is higher due to the presence of shell-rich marine mudstone and some carbonate rocks. In January, the concentration of Mg2+in the study area ranges between 2 and 178 mg/l with an average of 55 mg/l and in June, it ranges between 3 and 126 mg/l with an average value of 45 mg/l as shown in Fig. 4b. Reverse cationic exchange, i.e., the replacement of calcium and magnesium ions by sodium ions in the groundwater, results in a higher concentration of Ca2+ and Mg2+ in some areas (Srinivas et al. 2015).
The chloride ion concentration in groundwater originates from various sources like leaching of sedimentary rocks and soils, weathering, domestic and industrial waste discharges, and saltwater intrusion (Krishnakumar et al. 2009). In the study area, Cl-concentration ranges between 20–426 mg/l during January with an average of 161 mg/l, whereas in June, it ranges from 17 mg/l to 646 mg/l with an average of 141 mg/l. Cl- ion spatial distribution is shown in Fig. 4c. The sodium concentration in January ranges from 16 to 420 mg/l, with an average value of 105 mg/l. In June, it ranges from 23 to 470 mg/l, with an average of 125 mg/l and the distribution is shown in Fig. 4d, where few samples exceed the permissible limit. In the study area, about 40% of samples in January and 30% of samples in June exceed the permissible limit for potassium. High potassium ion concentration is the result of agricultural runoff and saline water intrusion (Alfarrah and Walraevens 2018) (Fig. 5a). In the case of bicarbonate (HCO3−) and sulfate (SO42−), no samples exceed the permissible limit of WHO. The spatial distribution of HCO3− and SO42− ions in the study area is depicted in Fig. 5b, c, respectively.
In our study, spatial distribution maps of Figs .3, 4 and 5 show good groundwater quality in the Mandaikadu region, while Manavalakurichi regions appear to pose contaminated groundwater. Generally, groundwater contamination is reasoned by both natural and human-induced chemicals. In our study area, probable causes for natural contamination include nearby seawater intrusion, backwater from Valliyar river estuary and geomorphic features. The main human activities in the study area contribute to contamination are agriculture and coir industries. The above analysis results will be used to calculate a weighted average to categorize the groundwater quality for suitability of drinking purposes.
Water quality index (WQI)
WQIs have been applied worldwide and are used to assess the overall water quality within a particular region quickly and effectively. The water quality index (WQI) was calculated to enumerate the impact of natural and anthropogenic activities (Bilgin 2018). Based on the WQI of British Columbia Canadian council, the Canadian Water Quality index (CWQI) was developed. To measure CWQI, a set of 11 physical and chemical parameters such as pH, EC, TDS, TH, Ca2+, Mg2+, Na+, K+, HCO3−, Cl− and SO42− were resolved. The water quality objective depends on three attributes of CWQI (Khan et al. 2004; Hurley et al. 2012; Gupta et al. 2017) and it can be calibrated using Eq. (1) (Lumb et al. 2006)
-
(i)
Scope–F1.
-
(ii)
Frequency–F2.
-
(iii)
Amplitude–F3.
According to CWQI, the water can be classified into five types, namely poor, marginal, fair, good and excellent, given in Table 4. If the CWQI ranking stands between poor and marginal, then the water is dangerous and needs to be purified. In the case of good and excellent ranking, the water does not need purification. (Khan et al. 2004). Finally, the fair ranking of CWQI needs to be improved for the water quality by purification. In the study area, 63% of the water samples are categorized as good to excellent in January and June. Figure 6 shows the spatial distribution map and most of the water samples in and around the Manavalakurichi area show marginal to fair condition.
Irrigation water quality
The quality criteria for determining groundwater viability for agricultural purposes include salinity indices, comprising Na%, SAR and PI.
Sodium Absorption ratio (SAR)
The SAR value represents the capability of soils to adsorb Na+ from irrigation water and, therefore, the risk of sodium hazard to soils posed by irrigation with water containing massive amounts of sodium (Davraz and Ozdemir 2014). The estimation of sodium hazard value by the relative concentration of major cations such as Na+, Ca2+ and Mg2+ is expressed in meq/l, and SAR is calculated by Eq. (2) (Hem 1985; Wen et al. 2020)
The calculated values of SAR in the study area range between 0.64 and 16.86 meq/l during January and 1.45 to 9 meq/l during June with averages of 3.3 and 3.6 meq/l. The classification of groundwater samples based on SAR values is given in Table. 5. The correlation plot between SAR and EC in (USSL) salinity diagram (Richards 1954) gives a perfect idea about the salinity hazard in irrigation water (Ravikumar et al. 2010). Utmost, the samples collected in the study area belong to the C3–S1 category deciphering high salinity and low sodium type. Two samples in January and three samples in June indicate high salinity (C3) and medium sodium hazard (S2), which falls under C3–S2 classification (Fig. 7). On soil with restricted drainage, this class cannot be used (Ravikumar et al. 2011). The samples plotted in C1S1 and C2S1 indicate good irrigation quality (Singh et al. 2020).
Sodium percentage
The sodium concentration in irrigation waters is generally denoted as sodium percentage. Na% is measured in meq/l, and it can be calculated using Eq. (3).
High sodium concentration in irrigation water leads to Na+ absorption by clay particles and displacing Mg2+ and Ca2+ that reduces the soil permeability, eventually resulting in poor internal drainage, which indirectly affects plant growth (Ravikumar 2011). Wilcox diagram (Wilcox 1955) for irrigation water classification correlates Na% and EC (Fig. 8). It shows that 47% of samples come under the grade of very good to good, 40% of samples belong to good to permissible, and the remaining 13% of samples the permissible to the doubtful cadre for irrigation purposes in January. During June, 60% of samples fall in very good to good, 30% of samples fall in the good to permissible, 7% of samples fall in permissible to doubtful, and the remaining 3% fall in doubtful to unsuitable for irrigation.
Permeability index
Permeability index (PI) is essential for irrigation water that affects soil permeability, influenced by sodium, calcium, magnesium and bicarbonate contents in the soil (Davraz and Ozdemir 2014). The PI values replicate the groundwater quality for irrigation calculated using Eq. (4) (Doneen 1964; Raghunath 1987) where all the ions were given in milliequivalents per liter.
The PI values greater than 75 indicate the excellent quality of water for irrigation. The PI values ranging between 25 and 75 fall in the insensible water quality, and ranging below 25 is unfit for irrigation (Al-Amry 2008). In January, 47% of samples fall in excellent quality, and 47% of samples under sensible quality and the remaining 6% of samples in the unsuitable category for irrigation. During June, 50% of samples were excellent, and the remaining 50% of samples with sensible quality for irrigation.
Water quality plots and interpretation
Hydrogeochemical facies
The hydrogeochemical facies interpretation is useful to determine the geochemical histories and flow patterns of groundwater (Rajkumar et al. 2019). Piper trilinear plot shows the changes in water quality within an aquifer using the two triangle-shaped fields. Each represents the composition of cations and anions, and both compositions present in the groundwater represented by the diamond-shaped field (Piper 1944). The classification of hydrogeochemical facies for groundwater in January and June plotted by piper trilinear diagram is shown in Fig. 9.
During January and June, most water samples fall in the NaCl segment, followed by mixed CaMgCl > CaNaHCO3 > CaHCO3. The dominance of NaCl and CaMgCl in the study area indicates saltwater intrusion to the aquifer and anthropogenic contamination sources like domestic wastewater. However, the presence of CaNaHCO3 and CaHCO3 suggests recharge of freshwater and mineral dissolution (Senthilkumar et al. 2014).
Gibbs plot
Gibbs plot provides a clear idea about the close relationship between aquifer lithology and the chemical composition of groundwater. It is based on the mechanisms that control groundwater chemistry like rock weathering, atmospheric precipitation and evaporation (Gibbs 1970). Two plots proposed by Gibbs are,
-
i)
TDS versus Cl–/ (Cl–+HCO3–)
-
ii)
TDS versus Na+/ (Na+ + Ca2+)
Both the plots drawn for January and June samples are shown in Fig. 10. The Gibbs plots endorse that most of the samples acquired in the study area fall in the rock dominant region. The curve relating the TDS and cation indicates the presence of cations in a few wells, maybe because of evaporation or crystallization processes. The major ion chemistry of groundwater in this sector is primarily controlled by the weathering mechanism of rocks, which has been theorized by the Gibbs diagrams (Senthilkumar and Elango 2013).
The evidence of weathering shown in the plot for (Na+ + K+) vs. cations of both months indicates that nearly half of the samples fall below the equiline, which is shown in Fig. 11. The cations present in the groundwater are derived from the silicate weathering in the geochemical process (Subramani et al. 2010). The scatter diagram of HCO3− + SO42− vs. Ca2+ + Mg2+ indicates that 30% of the samples of both months fall below the equiline shown in Fig. 12. From the result, it is clear that the primary process behind groundwater evolution may be due to silicate weathering (Sheikh et al. 2017).
Cl − /HCO 3 − ratio
Saltwater intrusion can be identified by the ratio of Cl− to HCO3 ions and is studied to find the source of salinity in groundwater (Shin et al. 2020). In our study area, the ratio of Cl− to HCO3− ions ranges from 0.3 to 9.4 in January. During June, it ranges from 0.23 to 8.5, which shows a strong positive correlation with Cl− ion concentration. This relation (ratio of Cl−/HCO3− vs. Cl) proves the occurrence of saltwater intrusion, and Fig. 13 shows that the groundwater is slightly affected by seawater intrusion.
Data validation using the geophysical methods
An integrated approach of using geophysical and hydrogeochemical investigations was utilized (Abou Heleika et al. 2018; Alfaifi et al. 2019). The self-potential (SP) method is an important geophysical technique that responds directly to fluid flow. It involves the passive measurement of electric potential distribution at the surface of the earth with non-polarizable electrodes. The origin of SP has two main components if the groundwater is contaminated. That is, (1) electrokinetic contribution associated with groundwater flow through the permeable soil and (2) electrochemical component associated with oxide reduction phenomena. Many researchers used the SP method to study the hydrological properties and proved it as a suitable way to identify the direction of groundwater flow (Fournier 1989; Titov et al. 2005; Arsène et al. 2018).
Twenty-four measuring points were collected throughout the study area to delineate the self- potential in millivolts. The electrical potentials resulting from underground conditions of fluid pressure, temperature and salt concentration are the primary source for self-potential distribution in the study area (Arsène et al. 2018). SP anomaly increases with increasing ion concentration in the groundwater (Giampaolo et al. 2016). Figure 14 shows the spatial distribution of self-potential, and it ranges from -243 to 330 mV. The area with freshwater shows a clear negative SP signal at the surface, whereas a higher positive SP signal indicates groundwater contamination due to seawater intrusion (Revil et al. 2005). The contour map suggests that contaminated water spread is from Manavalakurichi toward the north and west direction in the study area.
The geophysical resistivity method is a useful technique to differentiate the subsurface resistivity to identify the seawater intrusion. In coastal regions, the seawater intrusion is determined by various researchers using the geophysical resistivity method to distinguish the freshwater and saltwater zones (Senthilkumar et al. 2019; Maiti and Gupta 2020). Especially to reveal the saline water intrusion, the one-dimensional array method in VES is useful (Song et al. 2006). The interpreted VES curves are shown in Fig. 15, which reveals the layer's resistivity and thickness. The resistivity value of aquifer formation decreases with an increase in the ionic concentration of water. Hence, the results of VES-1, VES-2 and VES-3 confirm the presence of saline water intrusion because of low aquifer resistivity with a depth range of 2, 5 and 8 m (Table 6).
The resistivity pseudo-cross sections were constructed along five profiles based on the resistivity values (Fig. 16.). The pseudo-cross sections are prepared using IPI2win software, and it gives a simple two-dimensional image that provides an idea about the subsurface based on resistivity. The resistivity cross sections enable us to decipher subsurface resistivity distribution in both horizontal and vertical directions. For the generation of pseudo-cross section, a maximum number of VES locations have been considered along with the selected profiles. The pseudo-section profile is prepared by joining the VES stations VES 4, VES 3, VES 2, VES 1 and VES 5 along the west to east direction. On comparison of resistivity pseudo-cross-section map with Public Works Department (PWD) bore hole data and the geological information of the study area, depth-wise subsurface layers in the form of sand, sandy clay and clayey sand are identified.
In the coastal region of the study area, the aquifer material consists of fine to coarse grain sand, sandy clay, clayey sand and small patches of clay occurring as lenses. These are identified using resistivity pseudo-cross section and available borehole lithology map of the study area. According to Perumal (2008), the clay that exists as thin lenses or small patches is mostly deposited in fluvial and shallow marine environments in the study area. Geologically, we see the presence of clay in a shallow depth, which has the characteristics to control the seawater intrusion. Thereby, VES aquifer resistivity of the coastal line of the study area whose value is low but not less than 5 is indicated as moderately affected by coastal salinity, whereas the other areas of western and southern parts (VES-4, VES-5 and VES-6) are noted as high resistivity values indicating the presence of fresh groundwater for measured depth.
Conclusion
In the current study, the hydrogeochemical analysis reveals that the groundwater in the Mandaikkadu area is acidic. The concentrations of TDS and EC were observed to be high due to saltwater intrusion in the coastal region of Manavalakurichi. From this study, in and around the area of Manavalakurichi, looking at the WQI value, it is clear that the water needs to be purified to improve the quality before use. Major cations and anions in the study are Na+ and Cl−. Therefore, the greater number of the samples comes under Na-Cl and mixed CaMgCl type in piper plot, which is mainly due to saltwater intrusion. Cl−/HCO3− ratio indicates that most of the samples show low to moderate seawater intrusion in the study area. No major differences in physico-chemical parameters of groundwater quality during January and June were noticed. It may be due to the prevailing hydrogeological condition in the study area. The geophysical self-potential method reveals a high positive value in Manavalakurichi, indicating saltwater intrusion in a particular location. Moreover, the resistivity studies confirm the presence of contaminated water due to saline water intrusion with a higher ionic concentration near the coastal region. From this result, marine water influencing the chemistry of groundwater can be proved. We recommend the use of water from the northwest area, where the water quality is best suitable for drinking. There is a need to properly monitor surrounding wells and Valliyar river backwater salinity to prevent the increase in saltwater contamination in the Manavalakurichi region.
Data availability
The data were collected and analyzed primarily by us.
References
Abou Heleika M, Ismail E, Ahmed M (2018) Delineation of contamination zone using geophysical and hydrogeochemical methods around the El Moheet drain in the El Minia district upper Egypt. Arab J Geosci. https://doi.org/10.1007/s12517-018-3927-2
Adimalla N (2019) Controlling factors and mechanism of groundwater quality variation in semiarid region of South India: an approach of water quality index (WQI) and health risk assessment (HRA). Environ Geochem Health 42(6):1725–1752. https://doi.org/10.1007/s10653-019-00374-8
Adimalla N, Li P, Venkatayogi S (2018) Hydrogeochemical evaluation of groundwater quality for drinking and irrigation purposes and integrated interpretation with water quality index studies. Environ Process 5:363–383. https://doi.org/10.1007/s40710-018-0297-4
Al-Amry AS (2008) Hydrogeochemistry and groundwater quality assessment in an arid region: a case study from Al Salameh Area, Shabwah, Yemen. The 3rd International Conference on Water Resources and Arid Environments and the 1st Arab WaterForum.
Alfaifi H, Kahal A, Albassam A, Ibrahim E, Abdelrahman K, Zaidi F, Alhumidan S (2019) Integrated geophysical and hydrochemical investigations for seawater intrusion: a case study in southwestern Saudi Arabia. Arab J Geosci. https://doi.org/10.1007/s12517-019-4540-8
Alfarrah N, Walraevens K (2018) Groundwater overexploitation and seawater intrusion in coastal areas of arid and semi-arid regions. Water. https://doi.org/10.3390/w10020143
Ameen HA (2019) Spring water quality assessment using water quality index in villages of Barwari Bala Duhok, Kurdistan Region, Iraq. Appl Water Sci. https://doi.org/10.1007/s13201-019-1080-z
Arnous MO, Aboulela HA, Green DR (2011) Geo-environmental hazards assessment of the north western Gulf of Suez. Egypt J Coast Conserv 15:37–50. https://doi.org/10.1007/s11852-010-0118-z
Arsène M, Wassouo Elvis BW, Daniel G et al (2018) Hydrogeophysical investigation for groundwater resources from electrical resistivity tomography and self-potential data in the Méiganga Area, Adamawa, Cameroon. Int J Geophys 2018:1–14
Ayman AA, Mohamed HA (2011) Hydrochemical evolution and variation of groundwater and its environmental impact at Sohag. Egypt Arab J Geosci 4:339–352. https://doi.org/10.1007/s12517-009-0055-z
Bhunia GS (2020) An approach to demarcate groundwater recharge potential zone using geospatial technology. Appl Water Sci. https://doi.org/10.1007/s13201-020-01231-1
BIS (2000) Drinking water specification. Bureau of Indian Standard; New Delhi IS10500.
Bilgin A (2018) Evaluation of surface water quality by using Canadian council of ministers of the environment water quality index (CCME WQI) method and discriminant analysis method: a case study Coruh River Basin. Environ Monit Assess. https://doi.org/10.1007/s10661-018-6927-5
Bouwer H (1978) Groundwater hydrology. McGraw-Hill, New York
Burrough PA, McDonell RA (1998) Principles of geographical information systems. Oxford University Press, Oxford, p 333
Chidambaram S, Sarathidasan J, Srinivasamoorthy K, Thivya C, Thilagavathi R, Prasanna MV, Singaraja C, Nepolian M (2018) Assessment of hydrogeochemical status of groundwater in a coastal region of southeast coast of India. Appl Water Sci. https://doi.org/10.1007/s13201-018-0649-2
Davraz A, Ozdemir A (2014) Groundwater quality assessment, and its suitability in Celtikci plain (Burdur/Turkey). Environ Earth Sci 72:1167–1190. https://doi.org/10.1007/s12665-013-3036-1
Doneen LD (1964) Notes on water quality in agriculture. Published as a water science and engineering paper 4001, Department of Water Science and Engineering, University of California.
FAO (2003) The irrigation challenge: increasing irrigation contribution to food security through higher water productivity from canal irrigation systems. IPTRID Issue Paper 4, IPTRID Secretariat, Food and Agricultural Organization of the United Nations, Rome.
Fournier C (1989) Spontaneous potentials and resistivity surveys applied to hydrogeology in a volcanic area: case history of the Chaıˆne des Puys (Puy-de-Doˆme, France). Geophys Prospect 37:647–668
Ghalib HB (2017) Groundwater chemistry evaluation for drinking and irrigation utilities in east Wasit province, Central Iraq. Appl Water Sci 7:3447–3467. https://doi.org/10.1007/s13201-017-0575-8
Giampaolo V, Calabrese D, Rizzo E (2016) Transport processes in porous media by self-potential method. Appl Environ Soil Sci 2016:1–12. https://doi.org/10.1155/2016/3951486
Gibbs RJ (1970) Mechanisms controlling world water chemistry. Science 17:1088–1090
Gnanachandrasamy G, Ramkumar T, Venkatrammanan S, Vasudevan S, Chung SY, Bagyaraj M (2015) Accessing groundwater quality in lower part of Nagapattinam district, Southern India: using hydrogeochemistry and GIS interpolation techniques. Appl Water Sci 5:39–55. https://doi.org/10.1007/s13201-014-0172-z
Golchin I, Azhdary Moghaddam M (2016) Hydro-geochemical characteristics and groundwater quality assessment in Iranshahr plain aquifer Iran. Environ Earth Sci. https://doi.org/10.1007/s12665-015-5077-0
GSI (2005) Geology and mineral map of Kanyakumari district compiled by Sundaram M, R. Renganathan and Vasudevan, D. Geological Survey of India, Kolkata
Gupta N, Pandey P, Hussain J (2017) Effect of physicochemical and biological parameters on the quality of river water of Narmada, Madhya Pradesh, India. Water Sci 31:11–23. https://doi.org/10.1016/j.wsj.2017.03.002
He X, Wu J, He S (2018) Hydrochemical characteristics and quality evaluation of groundwater in terms of health risks in Luohe aquifer in Wuqi County of the Chinese Loess Plateau, northwest China. Human Ecol Risk Assess: An Int J 25:32–51. https://doi.org/10.1080/10807039.2018.1531693
Hem JD (1985) Study and interpretation of the chemical characteristics of natural water USGS. Water Supply paper 2254:117–120
Hem JD (1991) Study and interpretation of the chemical characteristics of natural waters. Book 2254, 3rd edn. Scientific Publishers, Jodhpur
Hurley T, Sadiq R, Mazumder A (2012) Adaptation and evaluation of the Canadian council of ministers of the environment water quality index (CCME WQI) for use as an effective tool to characterize drinking source water quality. Water Res 46:3544–3552
IWS (2000). Thanni model for Kodaiyar river basin (Draft Report), Technical secretariat for river basin boards, Institute of Water Studies, Chennai
Jakeman AJ, Barreteau O, Hunt RJ, Rinaudo J, Ross A (2016) Integrated groundwater management: concepts, approaches and challenges. Springer, New York
John-Mark D (2006) Application and tests of the canadian water quality index for assessing changes in water quality in lakes and rivers of central North America. Lake and Reserv Manag 22(4):308–320. https://doi.org/10.1080/07438140609354365
Khan AA, Paterson R, Khan H (2004) Modification and application of the Canadian council of ministers of the environment water quality index (CCME WQI) for the communication of drinking water quality data in newfoundland and labrador. Water Qual Res J Canada. 39:285–293
Krishnakumar S, Mohan VR, Dajkumar SJ, Jeevanandam M (2009) Assessment of groundwater quality and hydrogeochemistry of Manimuktha River basin, Tamil Nadu, India. Environ Monit Assess 159:341–351
Kumar SK, Logeshkumaran A, Magesh NS, Godson PS, Chandrasekar N (2015) Hydro- geochemistry and application of water quality index (WQI) for groundwater quality assessment, Anna Nagar, part of Chennai City, Tamil Nadu. India Appl Water Sci 5:335–343. https://doi.org/10.1007/s13201-014-0196-4
Kumari M, Rai SC (2020) Hydrogeochemical evaluation of groundwater quality for drinking and irrigation purposes using water quality index in semi arid region of India. J Geol Soc India 95:159–168. https://doi.org/10.1007/s12594-020-1405-4
Li P, He S, He X, Tian R (2018) Seasonal hydrochemical characterization and groundwater quality delineation based on matter element extension analysis in a Paper Wastewater Irrigation Area, Northwest China. Expos Health 10:241–258. https://doi.org/10.1007/s12403-17-0258-6
Li H, Smith C, Wang L, Li Z, Xiong C, Zhang R (2019) Combining spatial analysis and a drinking water quality index to evaluate monitoring data. Int J Environ Res Public Health 16:357. https://doi.org/10.3390/ijerph16030357
Lumb A, Halliwell D, Sharma T (2006) Application of CCME water quality index to monitor water quality: a case study of the Mackenzie River Basin, Canada. Environ Monit Assess 113:411–429. https://doi.org/10.1007/s10661-005-9092-6
Mahanta N, Mishra I, Hatui A, Mahanta PS, Sahoo HK, Goswami S (2019) Geochemical appraisal of groundwater qualities and its uses in and around Maneswar Block of Sambalpur District Odisha, India. Environ Earth Sci. https://doi.org/10.1007/s12665-019-8719-9
Maiti S, Gupta G (2020) Integrated geoelectrical and hydrochemical investigation of shallow aquifers in Konkan Coastal Area, Maharashtra, India: advanced artificial neural networks based simulation approach. Springer Geophys. https://doi.org/10.1007/978-3-030-28909-6_3
Mostafa MG, Uddin SM, Haque AB (2017) Assessment of hydro-geochemistry and groundwater quality of Rajshahi City in Bangladesh. Appl Water Sci 7:4663–4671. https://doi.org/10.1007/s13201-017-0629-y
Nakhaei M, Dadgar MA, Amiri V (2016) Geochemical processes analysis and evaluation of groundwater quality in Hamadan Province. Western Iran Arab J Geosci 9:384. https://doi.org/10.1007/s12517-016-2409-7
Naudet T, Revil A, Bottero JY (2003) Relationship between self-potential (SP) signals and redox conditions in contaminated groundwater. Geophys Res Lett 30:21–2091
Perumal SB (2008) Groundwater quality assessment and regional flow modelling of kanyakumari colachel coastal aquifer, Tamilnadu, India. Ph.D thesis, Anna university Chennai.
Perumal SB, Thamarai P, Elango L (2010) Hydrogeological studies along the coastal area of Kanyakumari to colachel after Tsunami, South Tamil Nadu. IJEP 30:793–813
Piper AM (1944) A graphic procedure in the geochemical interpretation of water analysis. Trans Am Geophys Union 25:914–923
Raghunath HM (1987) Groundwater, 2nd edn. Wiley, New Delhi, pp 344–369
Rajkumar S, Srinivas Y, Nair NC, Arunbose S (2019) Groundwater quality and vertical electrical sounding data of the Valliyar River Basin, South West Coast of Tamil Nadu. India Data in Brief 24:103919. https://doi.org/10.1016/j.dib.2019.103919
Ravikumar P, Venkatesharaju K, Prakash KL, Somashekar RK (2010) Geochemistry of groundwater and groundwater prospects evaluation, Anekal Taluk, Bangalore Urban District, Karnataka, India. Environ Monit Assess. https://doi.org/10.1007/s10661-010-1721-z
Ravikumar P, Somashekar R, Angami M (2011) Hydrochemistry and evaluation of groundwater suitability for irrigation and drinking purposes in the Markandeya River basin Belgaum District, Karnataka State, India. Environ Monit Assess 173:459–487. https://doi.org/10.1007/s10661-010-1399-2
Revil L, Cary Q, Fan A, Finizola F, Trolard, (2005) Self-potential signals associated with preferential ground water flow pathways in a buried paleo-channel. Geophys Res Lett 32:7. https://doi.org/10.1029/2004GL022124
Richards LA (1954) Diagnosis and improvement of saline alkali soils: agriculture, handbook. US Department of Agriculture, Washington DC, vol 160, p 60
Saraswat C, Kumar P, Bhalani P (2018) Sustainability Assessment of the Groundwater Quality in the Western India to Achieve Urban Water Security. Preprints 2018050199. https://doi.org/10.20944/preprints201805.0199
Sawyer GN, McCarthy DL (1967) Chemistry of sanitary engineers, 2nd edn. Mc Graw Hill, New York, p 518
Senthilkumar M, Elango L (2013) Geochemical processes controlling the groundwater quality in lower Palar river basin, southern India. J Earth Syst Sci 122:419–432
Senthilkumar S, Balasubramanian N, Gowtham B, Lawrence JF (2014) Geochemical signatures of groundwater in the coastal aquifers of Thiruvallur district, south India. Appl Water Sci 7:263–274. https://doi.org/10.1007/s13201-014-0242-2
Senthilkumar SK, Vinodh G, Johnson Babu B, Gowtham AV (2019) Integrated seawater intrusion study of coastal region of Thiruvallur district, Tamil Nadu South India. Appl Water Sci. https://doi.org/10.1007/s13201-019-1005-x
Sexenal N, Mishra SN (2011) J Chem Pharm Res 3:162–167
Shaikh H, Gaikwad H, Kadam A, Umrikar B (2020) Hydrogeochemical characterization of groundwater from semiarid region of western India for drinking and agricultural purposes with special reference to water quality index and potential health risks assessment. Appl Water Sci. https://doi.org/10.1007/s13201-020-01287-z
Sheikh MA, Azad C, Mukherjee S, Rina K (2017) An assessment of groundwater salinization in Haryana state in India using hydrochemical tools in association with GIS. Environ Earth Sci. https://doi.org/10.1007/s12665-017-6789-0
Shin K, Koh D, Jung H, Lee J (2020) The Hydrogeochemical characteristics of groundwater subjected to seawater intrusion in the archipelago. Korea Water 12(6):1542. https://doi.org/10.3390/w12061542
Singh DSH, Lawrence JF (2007) Groundwater quality assessment of shallow aquifer using geographical information system in part of Chennai city Tamilnadu. J. Geol. Soc. India 69:1067–1076
Singh KK, Tewari G, Kumar S (2020) Evaluation of groundwater quality for suitability of irrigation purposes: a case study in the Udham Singh Nagar, Uttarakhand. J Chem 2020:1–15. https://doi.org/10.1155/2020/6924026
Song S-H, Lee J-Y, Park N (2006) Use of vertical electrical soundings to delineate seawater intrusion in a coastal area of Byunsan, Korea. Environ Geol 52:1207–1219
Srinivas Y, Oliver DH, Raj AS, Chandrasekar N (2014) Quality assessment and hydrogeochemical characteristics of groundwater in Agastheeswaramtaluk, Kanyakumari district, Tamil Nadu. India Chin J Geochem 33:221–235. https://doi.org/10.1007/s11631-014-0681-3
Srinivas Y, Aghil TB, Oliver DH, Nair NC, Chandrasekar N (2015) Hydrochemical characteristics and quality assessment of groundwater along the Manavalakurichi coast Tamil Nadu, India. Appl Water Sci. https://doi.org/10.1007/s13201-015-0325-8
Subramani T, Rajmohan N, Elango L (2010) Groundwater geochemistry and identification of hydrogeochemical processes in a hard rock region Southern India. Environ Monitor Assess 162:123–137
Subramani T, Damodarasamy SR, Elango L (2005) Groundwater quality and its suitability for drinking and agricultural use in Chithar River Basin, Tamil Nadu, India. Environ Geol 47:1099–1110. https://doi.org/10.1007/s00254-005-1243-0
Thamarai P (2014) Groundwater quality assessment and regional flow modelling of kanyakumari colachel coastal aquifer Tamilnadu, India
Titov K, Revil A, Konosavsky P, Straface S, Troisi S (2005) Numerical modelling of self-potential signals associated with a pumping test experiment. Geophys J Int 162:641–650
Tiwari AK, De Maio Ghione R, Lavy MM (2017) Evaluation of hydrogeochemical processes and groundwater quality for suitability of drinking and irrigation purposes: a case study in the Aosta Valley region Italy. Arab J Geosci. https://doi.org/10.1007/s12517-017-3031-z
Venkatramanan S, Viswanathan PM, Chung SY (2019) GIS and geostatistical techniques for groundwater science. Elsevier, Amsterdam
Verma P, Singh PK, Sinha RR, Tiwari AK (2019) Assessment of groundwater quality status by using water quality index (WQI) and geographic information system (GIS) approaches: A case study of the Bokaro district, India. Appl Water Sci 10(1) https://doi.org/10.1007/s13201-019-1088-4
Wen Y, Qiu J, Cheng S, Xu C, Gao X (2020) Hydrochemical evolution mechanisms of shallow groundwater and its quality assessment in the estuarine coastal zone: a case study of Qidong China. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph17103382
WHO (2004) Guidelines for drinking-water quality, world health organization Geneva: p. 515 3rdedRecommendations. vol. 1.
WHO (2011) Guidelines for Drinking-water Quality. Fourth ed. World Health Organization.
Wilcox LV (1948) The quality water for irrigation use. US Dept Agric Bull 2003:40
Wilcox LV (1955) Classification and use of irrigation waters. USDA; Circular 969, Washington.
Zhang Q, Xu P, Qian H (2019) Assessment of groundwater quality and human health risk (HHR) evaluation of nitrate in the central-western Guanzhong basin, China. Int J Environ Res Public Health 16:4246. https://doi.org/10.3390/ijerph16214246
Funding
This research did not receive any specific grand from funding in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have participated in (a) conception and design or analysis and interpretation of the data; (b) drafting the article or revising it critically for important intellectual content; and (c) approval of the final version. This manuscript has not been submitted to nor is under review at, another journal or other publishing venue. The authors have no affiliation with any organization with a direct or indirect financial interest in the subject matter discussed in the manuscript. On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Stanly, R., Yasala, S., Oliver, D.H. et al. Hydrochemical appraisal of groundwater quality for drinking and irrigation: a case study in parts of southwest coast of Tamil Nadu, India. Appl Water Sci 11, 53 (2021). https://doi.org/10.1007/s13201-021-01381-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-021-01381-w