Abstract
This study investigated the performance of UVC/H2O2 and UVC processes for the degradation and mineralization of ceftriaxone as an antibiotic. The highest ceftriaxone degradation was obtained at a solution pH of 5 and H2O2 concentration of 10 mg/L. The apparent rate constant of ceftriaxone degradation was found to be 0.0302, 0.0165, and 0.0065 min−1 in the UVC/H2O2 process for the initial ceftriaxone concentrations of 5, 10, and 20 mg/L, respectively. Degradation and mineralization efficiencies of ceftriaxone was obtained to be 100% and 58%, respectively, in UVC/H2O2 process at reaction time of 120 min, whereas only 61% and 2.5% of ceftriaxone could be degraded and mineralized by UVC. The synergistic effect of UVC/H2O2 was found to be 35%. The presence of anionic species improved the photolysis efficiency which degraded ceftriaxone from 61 to 83%, while, in the UVC/H2O2 process, ability degradation declined from 100 to 70%. The efficiency of UVC/H2O2 and UVC process was not greatly affected in real tap water. Besides, the reduction patterns in the UVC/H2O2 and UVC processes were better described by pseudo-first- and second-order kinetics model with a reaction rate constant of 0.0165 and 0.0012 min−1, respectively. The rate constant of ceftriaxone degradation in the UVC/H2O2 process and at the presence of radical scavenger was found to be around 3.3 times lower than the one in its absence.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In recent years, there has been an increasing attention to the incidence of pharmaceuticals and related products in the aquatic environment. The prevalent use of pharmaceuticals and their insufficient removal in wastewater treatment plants have increased their entrance into natural waters due to their refractory reaction to conventional biological processes (Munoz et al. 2017; Palominos et al. 2009). Among pharmaceuticals, antibiotics play a significant role in polluting the environment due to their frequent use in both veterinary and human medicine (Elmolla and Chaudhuri 2010; Palominos et al. 2009). Antibiotics have been detected in groundwater, surface water, sediments, and drinking water (Kümmerer 2009). Adverse effects of antibiotics on various organisms such as aquatic organisms, bacterial community, plants, and soil organisms have been found even at very low exposure concentrations (Wen et al. 2011). For instance, the bacteria derived from sewage bioreactors have depicted resistance to several antibiotics (Gulkowska et al. 2008). Therefore, it is essential to treat the effluents containing antibiotics before their discharge into the environment (Wen et al. 2011).
Ceftriaxone is an antibiotic that is effective for the treatment of some bacterial infections. It is a third-generation cephalosporin that prevents bacterial cell wall mucopeptide synthesis (Ratti et al. 2015; Shokri et al. 2016). This antibiotic is confirmed by the federal drug administration (FDA) for intramuscular or intravenous (IV) submissions at the maximum doses of 2–4 g daily for up to 4–6 weeks (Ratti et al. 2015). The antibiotic chemical that is used as a pharmaceutical product reached an estimation of 100,000 to 200,000 tons per year worldwide (Kummerer 2003). Diwan et al. (2009) studied antibiotic concentrations in hospital effluents in India and reported that high concentration of ceftriaxone in two samples was 58.3 and 59.5 μg/L (Diwan et al. 2009). Opris et al. (2013) collected samples from a wastewater treatment plant in Romania in order to detect antibiotics. The ceftriaxone detected in the influent wastewater samples was 334 µg/L, and no target antibiotics were identified in effluent wastewater samples (Opris et al. 2013). Zuccato et al. (2000) detected ceftriaxone in the ng/L range in river or drinking water and river sediments (Zuccato et al. 2000).
In recent years, advanced oxidation processes (AOPs) which produce hydroxyl radicals have been successfully employed as efficient methods for treating organic micro-pollutants in waters (Lester et al. 2010; Maya-Treviño et al. 2014). In these processes, the produced HO· radicals degrade organic contaminants since they are highly reactive and nonselective electrophiles which rapidly react with many pollutants at kinetic constants within the range of 108–1010 M−1 s−1 (Lester et al. 2010; Yao et al. 2013).
The hydroxyl radicals can be generated through the combined application of ultraviolet light and hydrogen peroxide (UV/H2O2), which is one of the most frequently used AOP techniques in water treatment (Vogna et al. 2004b). Compared to chlorination, UV process is advantageous in terms of minimizing the formation of disinfection by-products and has been employed in water disinfection (Pereira et al. 2007). When photolysis is combined with hydrogen peroxide, a strong oxidant, the efficiency of process increased (Pereira et al. 2007). In this process which involves the use of UV/H2O2, hydroxyl radicals are generated by photolysis of H2O2 according to the following equation (Hirsch et al. 1999):
In UV/H2O2 process, some organic chemicals absorb UV light directly, leading to the destruction of the contaminant, and H2O2 addition is necessary to degrade resistant organic species (Kim et al. 2009). UV process is not an appropriate method for removing pharmaceuticals in wastewater treatment system; therefore, addition of H2O2 to the UV process is implemented to elevate the degradation rate (Kim et al. 2009; Lester et al. 2010). Many studies have effectively used the UV/H2O2 process to degrade various organic pollutants (Kim et al. 2009; Mohagheghian et al. 2015; Moussavi et al. 2018; Vogna et al. 2004a). For instance, in a study, the use of UV and UV/H2O2 for degrading carbamazepine showed that they could degrade carbamazepine very effectively, while direct photolysis was not effective for reducing this drug concentration (Vogna et al. 2004a).
Despite the prevalent use of ceftriaxone and the probability of its presence in aqueous environments which lead to serious concerns, there has been a rare attention to the degradation of this antibiotic in the literature. Shokri et al. (2016) investigated the photocatalytic degradation of ceftriaxone using immobilized TiO2 and ZnO nanoparticles. In their study, the UV/TiO2 process resulted in higher removal efficiencies (96.7% COD in 480 min) than that of the UV/ZnO process (75% COD in 480 min).
Thus, the primary objective of this study was to assess the efficacy of UVC radiation and UVC/H2O2 method in degrading and mineralizing ceftriaxone under different experimental conditions in aqueous solutions. In fact, the influences of the solution pH, H2O2 concentration, ceftriaxone concentrations at different reaction times, kinetic of reactions, the presence of different anions, and water impurities on ceftriaxone degradation were investigated.
Materials and methods
Reagents
Ceftriaxone was purchased from Exir Pharmaceutical Co. (Tehran, Iran) and used in the experiments. The properties of ceftriaxone are given in Table 1 (Nirav 2012). The chemicals used in this study (H2O2, H2SO4, and NaOH) were of analytical grade and were obtained from the Merck.
Experimental setup and procedure
The experimental setup was a cylindrical glass reactor with an inner diameter of 40 mm and a height of 200 mm into which a quartz sleeve of 350 mm diameter was longitudinally inserted at the axial center. The photoreactor had a working volume of 100 mL. A 9-W low-pressure mercury UV lamp (Philips, Holland) emitting a maximum wavelength of 254 nm and a light intensity of 2.2 mW/cm2 was used as the radiation source. The reactor walls were wrapped by aluminum foil to prevent release of UV radiation to the ambient. The experiments were conducted in a bench-scale setup, as shown in Fig. 1. The influence of various important parameters including solution pH (5, 7, 9), H2O2 concentration (0, 5, 10, 20, 40, 60, 80, and 100 mg/L), ceftriaxone concentrations (5, 10, 20 mg/L) at different reaction time (5–120 min), kinetic of reactions, and the presence of different anions (SO42−, PO43−, NO3−) with concentration of 3 mM and mixture of anions at tap water were examined to evaluate the efficiency of the UVC/H2O2 and UVC in degradation and mineralization of ceftriaxone in an aqueous solution. The samples were prepared dissolving ceftriaxone in twice-distilled water. In each run, the initial solution pH of 100 mL was adjusted to the specified value (5, 7, 9) via adding dilute 0.1 M HCl or 0.1 M NaOH and was measured using pH meter (Philips PW 9422). Experimental studies were independently repeated twice, and the data are presented in terms of mean ± standard deviation (SD). The space between UV source and solution was 2 cm in all experiments. During experiments, the solution in the reactor was constantly stirred at a rate of 100 rpm. In order to distinguish photolysis efficiency in degrading ceftriaxone, 100 mL of predetermined concentration of ceftriaxone solution was irradiated, and the removal efficiency was acquired after measuring ceftriaxone concentration at the end of radiation.
Analytical methods
After the appropriate irradiation time, samples were analyzed to determine the TOC and residual concentration of ceftriaxone and H2O2. Ceftriaxone concentration was measured using an Agilent 1260 infinity HPLC equipped with a ZORBAX Eclipse Plus C18 column (4.6 * 100 mm, 3.5 µm) with a diode array detector at 242 nm. The mobile phase involved a phosphate buffer (20 mM)/acetonitrile (90:10) with an injection flow rate of 1 mL/min. The percentage of ceftriaxone degradation was calculated as:
where C0 and Ct denote the ceftriaxone concentrations before and after process, respectively.
In order to specify the amount of mineralization, samples were taken out at certain time intervals and the organic content was determined by using a TOC analyzer (Shimadzu 5000). The TOC removal percentage was calculated based on the following equation:
where TOC0 and TOC1 (mg/L) represent the initial and final TOC of the solution, respectively.
A specific amount of H2O2 (mg/L) was added to reactor before the treatment, and no further hydrogen peroxide was added during the degradation. At the end of process, the residual concentration of hydrogen peroxide was determined through a spectrophotometer at 410 nm using titanium(IV) oxysulfate in accordance with the DIN 38402H15 method (Giannakis et al. 2017). To determine the apparent reaction rate constant (kapp) values for the degradation of ceftriaxone, a pseudo-first-order kinetics model was used. The following equation shows the pseudo-first-order reaction kinetics:
where k1 is the apparent rate constant, and C0 and Ct are the ceftriaxone concentration at the beginning and after time (t) of the reaction.
Results and discussions
Effect of pH
The effect of pH value was studied by adding incremental amounts of either diluted H2SO4 or NaOH to the ceftriaxone solution in the presence of UVC/H2O2. Surprisingly, the degradation rate was observed to be lower in the alkaline media as compared to the one in the acidic media (Fig. 2). The photolytic ceftriaxone degradation appeared to be at its best level in acidic pH and lowest in neutral pH. For example, after 120-min irradiation, the removal percentages of ceftriaxone were observed to be 100, 77, and 85% in pH levels of 5, 7, and 9, respectively. The standard deviation was below 3% for all experiments.
The reduction in ceftriaxone degradation as a result of the increase in solution pH can be explained through the decomposition of H2O2 into H+, hydroperoxy anion (HO2−) in alkaline pH [Eq. (5)], and reaction HO2− to H2O2 [Eq. (6)], which cause the amount of H2O2 accessible for ·OH generation to reduce (Lin et al. 2016; Moussavi et al. 2018).
Thus, higher solution pH led to an increase in H2O2 dissociation and therefore a decrease in the amount of hydroxyl free radical production. The total of these events reduces the process efficiency in alkaline media.
The mechanisms contributing to ceftriaxone degradation in UVC/H2O2 process are (1) indirect reactions of ceftriaxone molecules to hydroxyl radicals and (2) absorption of UV radiation by ceftriaxone molecules and thus direct photolysis [Eqs. (7) and (8)].
In alkaline media, hydroperoxy anion (HO2−) is formed, might react with the ·OH [Eq. (9)], and thus reduces the role of the main reaction [Eq. (7)] (Lin et al. 2016; Moussavi et al. 2018).
On the other hand, significant dissociation of H2O2 occurs at pH > 11. Thus, the role of H2O2 dissociation is not meaningful, and the reduction in ceftriaxone degradation at alkaline media can be due to the diminution of ·OH in the redox potential with the increase in pH level (Moussavi et al. 2018). AlHamedi et al. (2009) investigated the degradation of rhodamine B via UV/H2O2 process and observed the maximum degradation of rhodamine B at acidic media (pH = 3). Similarly, Shokri et al. (2016) reported that UV/TiO2 process reached its maximum degradation (92.8%) of ceftriaxone at pH 5.0 after 120-min irradiation.
In the present study, in order to better understand the mechanism of ceftriaxone removal in the UVC/H2O2 process, the removal efficiency of ceftriaxone in the presence of one radical scavenge (tert-butyl alcohol with a reaction rate constant of 6.0 × 108 M−1 s−1) was examined. The results showed that (Fig. 2), within 120 min, a complete degradation of ceftriaxone occurred in the UVC/H2O2 process, while, in the presence of 0.5 g tert-butyl alcohol, the degradation efficiency of ceftriaxone decreased to 83.6% (the result is not shown). According to Fig. 3, under the same experimental conditions, the pseudo-first-order rate constant of ceftriaxone degradation in the UVC/H2O2 process in presence of tert-butyl alcohol is around 3.3 times lower than that of the condition where it was absent. Accordingly, the presence of powerful hydroxyl radical scavenger reduces the reaction rate constant (Pignatello et al. 1999). This confirms that hydroxyl radical [Eq. (7)] plays a major role in ceftriaxone degradation in the UVC/H2O2 process.
Effect of H2O2 concentration
The effect of H2O2 concentration on 10 mg/L ceftriaxone degradation was investigated at pH = 5 and time reaction of 120 min. Figure 4 demonstrates the efficiency of UVC, UVC/H2O2, and H2O2 and synergistic effect of UVC/H2O2 at various H2O2 concentrations of 0–100 mg/L.
As Fig. 4 illustrates, in the absence of H2O2, the degradation efficiency is only 61% (photolysis process). This is due to the fact that hydroxyl radicals were produced only through photolysis (Eq. (10)) and photoionization of water molecules and in the absence of H2O2 (Eq. (11)).
Moreover, it can be concluded that the increase in H2O2 from 5 to 10 mg/L enhances the degradation efficiency from 77 to 100% and then remains constant up to 20 mg/L H2O2 concentration. This phenomenon can be explained by the higher production of hydroxyl radicals due to the presence of H2O2 in the UVC reactor through reactions [Eqs. (1 and 12)] (Liao et al. 2016). Accordingly, 10 mg/L H2O2 can be considered as an optimum addition loading for efficient degradation.
Additional increases in H2O2 concentration above 20 mg/L decreases the degradation efficiency from 100 to 90%. This is sensible considering the fact that, in the case of over plus H2O2, ·OH radicals efficiently react with H2O2 and produce hydroperoxyl radicals (\({\text{HO}}_{2}^{ \cdot }\)) [Eq. (13)] and hydroxyl free radicals generated at great dosage react with \({\text{HO}}_{2}^{ \cdot }\) [Eq. (14)] or dimerize to H2O2 [Eq. (15)] (Daneshvar et al. 2007, 2008).
In this study to calculate the synergistic effect of H2O2 adding to the UVC process, the following equation is used:
Removal efficiency of ceftriaxone at constant time (120 min) in UVC is 61%, whereas, in H2O2 and UVC/H2O2 of 10 mg/L dosage, it is 4 and 100%, respectively. Thus, the synergistic effect of UVC/H2O2 was found to be 35% [Eq. (16)]. This shows that the hydroxyl radicals generated from interactions between H2O2 and UVC in the UVC/H2O2 process [Eqs. (1 and 12)] were the main species extremely contributed to ceftriaxone degradation (Moussavi et al. 2018). Peng et al. (2017) reported that the degradation of ibuprofen in UVC/H2O2 process increased when the H2O2 concentration increased from 0.1 to 0.6 mM. In their study, the optimum concentration of H2O2 was obtained to be 0.54 mM.
Kinetics of UV/H2O2 and UV processes
The influence of ceftriaxone concentration (5–20 mg/L) on its decay kinetic during UV/H2O2 process at pH = 5 and 10 mg/L H2O2 is presented in Fig. 5. It can be seen that the removal efficiencies of ceftriaxone decreased in accordance with the increasing in initial concentrations. For example, the increase in the initial concentration from 5 to 20 mg/L led to a decrease in the degradation efficiency from 81 to 43% at a 60 min irradiation time. This can be explained considering that both the contaminant and H2O2 absorb UV radiation in the range emitted by the lamp (Aleboyeh et al. 2005). In addition, a greater amount of ceftriaxone molecules were in the reaction medium due to the increase in ceftriaxone concentration for a specified amount hydroxyl free radicals. Besides, an increased competition might be accomplished between ceftriaxone molecules and the degradation by-products with hydroxyl radicals oxidizing molecules (Sharma et al. 2015). These processes resulted in a reduction in ceftriaxone degradation efficiency due to an increase in initial concentration.
Table 2 demonstrates the calculated values of Kapp and liner correlation (R2) in case of pseudo-first-order kinetic for UV/H2O2 process. According to table, in the UVC/H2O2 process, the apparent rate constant of ceftriaxone degradation is 0.0302, 0.0165, and 0.0065 min−1 for the initial ceftriaxone concentrations of 5, 10, and 20 mg/L, respectively. The decrease in kapp due to the increase in ceftriaxone concentration can be attributed to the progressive acceleration of competitive reactions between ·OH radicals and the lately formed oxidant by-products of ceftriaxone during the process (El-Desoky et al. 2010). Li et al. (2018) used nano-graphite anode for electrochemical degradation of ceftriaxone sodium (10 mg/L) and found that the apparent rate constant is 0.01473 min−1. Hence, we can postulate that the treatment time required in UVC/H2O2 process is 1.12 times lower than the one needed in electrochemical degradation of ceftriaxone using nano-graphite anode. In order to obtain the kinetic model in UV process, the experimental data were fitted with zero-, first-, and second-order equations. The calculated values of reaction rate constant (k) and the linear correlation coefficients (R2) for UV process are presented in Table 3 and indicate that the second-order reaction model exactly matched the experimental data with a determination coefficient (R2) greater than 0.98 for all concentrations.
Mineralization of ceftriaxone by UVC/H2O2 and UVC
The rate of ceftriaxone mineralization (based on TOC reduction) as a function of electrolysis time in the UVC/H2O2 and UVC processes is depicted in Fig. 6. The UVC/H2O2 process efficiency was obviously higher than that of UVC process. As shown in Fig. 6, after 120 min of treatment, 58% of TOC was removed in UVC/H2O2 process, while the UVC process mineralization increased from 2.5% at the reaction time of 5 min to 28% at the reaction time of 120 min. As compared to its degradation, the lower rate of ceftriaxone mineralization can be relevant to the formation of organic by-products which are simpler than the ceftriaxone in terms of structure during the degradation process (Moussavi et al. 2018).
Effect of anions on degradation of ceftriaxone in UVC/H2O2 process
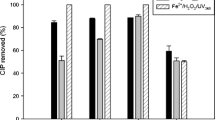
The efficiency of UVC/H2O2 and UV processes in the presence of several main water anions including phosphate, sulfate, and nitrate as well as in the tap water is depicted in Fig. 7a, b, respectively. The experiments carried out in the presence of several main water anions depicted a decrease in the removal efficiency of ceftriaxone in UVC/H2O2 process. As shown in Fig. 7a, removal efficiency of ceftriaxone through UVC/H2O2 in the absence of anions was 100%, while, in the presence of sulfate, phosphate, and nitrate removal efficiency decreased to 85, 77, and 70%, respectively. This is due to the fact that the anions react with ·OH radicals [Eqs. (17–19)], absorb the UV radiation and thus limit the amount of ·OH radical accessible for the ceftriaxone oxidation (Jafari et al. 2016; Moussavi et al. 2018; Sharma et al. 2016).
The oxidation power of these anion radicals formed in these reactions (Eqs. 17–19) is much lower than that of hydroxyl radicals. On the other, these anion radicals may react with H2O2 and consume it (Moussavi et al. 2018). Therefore, the presence of anions caused a decrease in the removal efficiency of ceftriaxone in UVC/H2O2 process. Moussavi et al. (2018) reported the greatest decrease in removal efficiency of BPA in the order of nitrate > sulfate > chloride > bicarbonate. As Fig. 7a shows, the highest amount of reduction effect was related to nitrate (30% reduction effect). This observation can be due to the absorption of UV by the nitrate which prevents the hydroxyl radicals generation reactions (Moussavi et al. 2014). The decrease in ceftriaxone degradation rate in the presence of sulfate and phosphate can be related to ·OH scavenging by these anions (Arany et al. 2014). In addition, Fig. 7a shows that the degradation efficiency in real tap water is 7% less than the one in distilled water solution. Therefore, the efficiency of UV/H2O2 process in tap water did not greatly reduce. This can be due to the low concentration of these anions in the water (Moussavi et al. 2018).
As shown in Fig. 7b, removal efficiency of ceftriaxone through UV process in the absence of anions was 61%, while, in the presence of sulfate, phosphate, and nitrate the removal efficiency enhanced to 83%, 80, and 78%, respectively. UV light converts of anions into anionic radicals whose oxidation power is higher than that of UV only irradiation (Sharma et al. 2016).
Conclusion
The efficiency of UVC and UVC/H2O2 to ceftriaxone removal from aqueous solution was investigated. Optimum H2O2 dosage was equal to 10 mg/L which caused 35% synergistic effect on the photolysis process. The presence of·OH in UV/H2O2 process by reducing the reaction rate constant in the presence of tert-butanol as radical scavengers was verified. In the same conditions experiments, the pseudo-first-order rate constant of ceftriaxone degradation in the UVC/H2O2 process at the presence of tert-butyl alcohol was around 3.3 times lower of its absence. Mineralization efficiency of ceftriaxone by UVC/H2O2 and UVC after 120 min of treatment was equal to 28 and 58.1%, respectively. The efficiency of UV/H2O2 process in real tap water did not greatly reduce. The results of this study showed that the UVC/H2O2 can be used as an appropriate way to remove pharmaceuticals from contaminated waters.
References
Aleboyeh A, Moussa Y, Aleboyeh H (2005) The effect of operational parameters on UV/H2O2 decolourisation of Acid Blue 74. Dyes Pigm 66(2):129–134
Alhamedi FH, Rauf M, Ashraf SS (2009) Degradation studies of Rhodamine B in the presence of UV/H2O2. Desalination 239(1–3):159–166
Arany E, Lang J, Somogyvari D, Lang O, Alapi T, Ilisz I, Gajda-Schrantz K, Dombi A, Kohidai L, Hernadi K (2014) Vacuum ultraviolet photolysis of diclofenac and the effects of its treated aqueous solutions on the proliferation and migratory responses of Tetrahymena pyriformis. Sci Total Environ 468–469:996–1006
Daneshvar N, Behnajady M, Asghar YZ (2007) Photooxidative degradation of 4-nitrophenol (4-NP) in UV/H2O2 process: influence of operational parameters and reaction mechanism. J Hazard Mater 139(2):275–279
Daneshvar N, Behnajady M, Mohammadi MKA, Dorraji MS (2008) UV/H2O2 treatment of Rhodamine B in aqueous solution: influence of operational parameters and kinetic modeling. Desalination 230(1–3):16–26
Diwan V, Tamhankar AJ, Aggarwal M, Sen S, Khandal RK, Lundborg CS (2009) Detection of antibiotics in hospital effluents in India. Curr Sci 97:1752–1755
El-Desoky HS, Ghoneim MM, Zidan NM (2010) Decolorization and degradation of Ponceau S azo-dye in aqueous solutions by the electrochemical advanced Fenton oxidation. Desalination 264(1):143–150
Elmolla ES, Chaudhuri M (2010) Comparison of different advanced oxidation processes for treatment of antibiotic aqueous solution. Desalination 256(1):43–47
Giannakis S, Hendaoui I, Jovic M, Grandjean D, De Alencastro LF, Girault H, Pulgarin C (2017) Solar photo-Fenton and UV/H2O2 processes against the antidepressant Venlafaxine in urban wastewaters and human urine. Intermediates formation and biodegradability assessment. Chem Eng J 308:492–504
Gulkowska A, Leung HW, So MK, Taniyasu S, Yamashita N, Yeung LW, Richardson BJ, Lei A, Giesy JP, Lam PK (2008) Removal of antibiotics from wastewater by sewage treatment facilities in Hong Kong and Shenzhen, China. Water Res 42(1):395–403
Hirsch R, Ternes T, Haberer K, Kratz K-L (1999) Occurrence of antibiotics in the aquatic environment. Sci Total Environ 225(1):109–118
Jafari SJ, Moussavi G, Hossaini H (2016) Degradation and mineralization of diazinon pesticide in UVC and UVC/TiO2 process. Desalin Water Treat 57(8):3782–3790
Kim I, Yamashita N, Tanaka H (2009) Performance of UV and UV/H2O2 processes for the removal of pharmaceuticals detected in secondary effluent of a sewage treatment plant in Japan. J Hazard Mater 166(2):1134–1140
Kummerer K (2003) Significance of antibiotics in the environment. J Antimicrob Chemother 52(1):5–7
Kümmerer K (2009) Antibiotics in the aquatic environment–a review–part I. Chemosphere 75(4):417–434
Lester Y, Avisar D, Mamane H (2010) Photodegradation of the antibiotic sulphamethoxazole in water with UV/H2O2 advanced oxidation process. Environ Technol 31(2):175–183
Li D, Guo X, Song H, Sun T, Wan J (2018) Preparation of RuO2-TiO2/Nano-graphite composite anode for electrochemical degradation of ceftriaxone sodium. J Hazard Mater 351:250–259
Liao Q-N, Ji F, Li J-C, Zhan X, Hu Z-H (2016) Decomposition and mineralization of sulfaquinoxaline sodium during UV/H2O2 oxidation processes. Chem Eng J 284:494–502
Lin C-C, Lin H-Y, Hsu L-J (2016) Degradation of ofloxacin using UV/H2O2 process in a large photoreactor. Sep Purif Technol 168:57–61
Maya-Treviño M, Guzmán-Mar J, Hinojosa-Reyes L, Ramos-Delgado N, Maldonado MI, Hernández-Ramírez A (2014) Activity of the ZnO–Fe2O3 catalyst on the degradation of Dicamba and 2, 4-D herbicides using simulated solar light. Ceram Int 40(6):8701–8708
Mohagheghian A, Karimi S-A, Yang J-K, Shirzad-Siboni M (2015) Photocatalytic degradation of a textile dye by illuminated tungsten oxide nanopowder. J Adv Oxid Technol 18(1):61–68
Moussavi G, Hossaini H, Jafari SJ, Farokhi M (2014) Comparing the efficacy of UVC, UVC/ZnO and VUV processes for oxidation of organophosphate pesticides in water. J Photochem Photobiol A 290:86–93
Moussavi G, Pourakbar M, Shekoohiyan S, Satari M (2018) The photochemical decomposition and detoxification of bisphenol A in the VUV/H2O2 process: degradation, mineralization, and cytotoxicity assessment. Chem Eng J 331:755–764
Munoz M, Mora FJ, De Pedro ZM, Alvarez-Torrellas S, Casas JA, Rodriguez JJ (2017) Application of CWPO to the treatment of pharmaceutical emerging pollutants in different water matrices with a ferromagnetic catalyst. J Hazard Mater 331:45–54
Nirav BP (2012) Development and validation of stability indicating method for simultaneous estimation of ceftriaxone and sulbactam injection using RP-UPLC method. IOSR J Pharm 2:29–37
Opris O, Soran M-L, Coman V, Copaciu F, Ristoiu D (2013) Determination of some frequently used antibiotics in waste waters using solid phase extraction followed by high performance liquid chromatography with diode array and mass spectrometry detection. Cent Eur J Chem 11(8):1343–1351
Palominos RA, Mondaca MA, Giraldo A, Peñuela G, Pérez-Moya M, Mansilla HD (2009) Photocatalytic oxidation of the antibiotic tetracycline on TiO2 and ZnO suspensions. Catal Today 144(1):100–105
Peng M, Li H, Kang X, Du E, Li D (2017) Photo-degradation ibuprofen by UV/H2O2 process: response surface analysis and degradation mechanism. Water Sci Technol 75(12):2935–2951
Pereira VJ, Weinberg HS, Linden KG, Singer PC (2007) UV degradation kinetics and modeling of pharmaceutical compounds in laboratory grade and surface water via direct and indirect photolysis at 254 nm. Environ Sci Technol 41(5):1682–1688
Pignatello JJ, Liu D, Huston P (1999) Evidence for an additional oxidant in the photoassisted Fenton reaction. Environ Sci Technol 33(11):1832–1839
Ratti E, Berry JD, Greenblatt DJ, Loci L, Ellrodt AS, Shefner JM, Cudkowicz ME (2015) Preclinical rodent toxicity studies for long term use of ceftriaxone. Toxicol Rep 2:1396–1403
Sharma J, Mishra I, Kumar V (2015) Degradation and mineralization of Bisphenol A (BPA) in aqueous solution using advanced oxidation processes: UV/H2O2 and UV/S2O8 2− oxidation systems. J Environ Manag 156:266–275
Sharma AK, Tiwari RK, Gaur MS (2016) Nanophotocatalytic UV degradation system for organophosphorus pesticides in water samples and analysis by Kubista model. Arab J Chem 9:1755–1764
Shokri M, Isapour G, Shamsvand S, Kavousi B (2016) Photocatalytic degradation of ceftriaxone in aqueous solutions by immobilized TiO2 and ZnO nanoparticles: investigating operational parameters. J Mater Environ Sci 7(8):2843–2851
Vogna D, Marotta R, Andreozzi R, Napolitano A, D’ischia M (2004a) Kinetic and chemical assessment of the UV/H2O2 treatment of antiepileptic drug carbamazepine. Chemosphere 54(4):497–505
Vogna D, Marotta R, Napolitano A, Andreozzi R, D’ischia M (2004b) Advanced oxidation of the pharmaceutical drug diclofenac with UV/H2O2 and ozone. Water Res 38(2):414–422
Wen Q, Kong F, Zheng H, Yin J, Cao D, Ren Y, Wang G (2011) Simultaneous processes of electricity generation and ceftriaxone sodium degradation in an air-cathode single chamber microbial fuel cell. J Power Sources 196(5):2567–2572
Yao H, Sun P, Minakata D, Crittenden JC, Huang C-H (2013) Kinetics and modeling of degradation of ionophore antibiotics by UV and UV/H2O2. Environ Sci Technol 47(9):4581–4589
Zuccato E, Calamari D, Natangelo M, Fanelli R (2000) Presence of therapeutic drugs in the environment. The Lancet 355(9217):1789–1790
Acknowledgements
We appreciate the Urmia University of Medical Science for providing financial and instrumental support to conduct this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have replied that they have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Khorsandi, H., Teymori, M., Aghapour, A.A. et al. Photodegradation of ceftriaxone in aqueous solution by using UVC and UVC/H2O2 oxidation processes. Appl Water Sci 9, 81 (2019). https://doi.org/10.1007/s13201-019-0964-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13201-019-0964-2