Abstract
The present study includes a systematic analysis of sediment contamination by heavy metals of the River Ghaghara flowing through the Uttar Pradesh and Bihar in Indian Territory. To estimate the geochemical environment of the river, seven heavy metals, namely Co, Cu, Cr, Ni, Cd, Zn, and Pb were examined from the freshly deposited river bed sediment. All the sediment samples were collected on a seasonal basis for the assessment of fluctuation in 2014–2015 and after preparation samples were analyzed using standard procedure. Result showed that heavy metal concentration ranged between 11.37 and 18.42 mg/kg for Co, 2.76 and 11.74 mg/kg for Cu, 61.25 and 87.68 mg/kg for Cr, 15.29 and 25.59 mg/kg for Ni, 0.21 and 0.28 mg/kg for Cd, 13.26 and 17.59 mg/kg for Zn, 10.71 and 14.26 mg/kg for Pb in different season. Metal contamination factor indicates the anthropogenic input in the river sediment was in the range of (0.62–0.97) for Co, (0.04–0.26) for Cu, (0.68–0.97) for Cr, (0.22–0.38) for Ni, (0.70–0.93) for Cd, (0.14–0.19) for Zn, and (0.54–0.71) for Pb. The highest contamination degree of the sediment was noticed as 4.01 at Ayodhya and lowest as 3.16 at Katerniaghat. Geo-accumulation index was noted between (0 and 1) which showed that sediment was uncontaminated to moderately contaminated and may have adverse affects on freshwater ecology of the river. Pollution load index (PLI) was found highest at Chhapra which was 0.45 and lowest at Katerniaghat which was 0.35 and it indicates that the river sediment has a low level of contamination. Significant high correlation was observed between Co, Cu, and Zn, it suggests same source of contamination input is mainly due to human settlement and agriculture activity. Positive correlation between Zn, Co, Cu, Cr, and Ni indicated a natural origin of these elements in the river sediment. Cluster analysis suggests grouping of similar polluted sites. The strong similarity between Co, Zn, Pb, Ni, Cu, and Cd showed relationship of these metals come from the same origin, which is possibly from natural and anthropogenic input which was also confirmed by correlation analysis. Using the various pollution indicators it was found that the river bed sediment is less contaminated by toxic metals during the study but the sediment quality may degrade in the near future due to increasing anthropogenic inputs in the river basin, hence proper management strategies are required to control the direct dumping of wastewater in the river.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Water bodies especially rivers are one of the most important natural resource on earth. They provide habitat for numerous aquatic lives and help in conserving the biodiversity.
The demand of water for industries and agriculture sector in India is continuously growing to meet the demands of 1.2 billion people (Rawat et al. 2017). Rivers are the major source of irrigation, hydropower and recharging the groundwater table, storage of water, purification, shipping, and landscape (Dong 2003). These are the carriers of approximately 37,000 km3 of water (Meybeck 1976) and 13.5 × 109 tons of sediment particles (Milliman and Meade 1983) to the oceans every year. Beside carrying different kinds of waste materials, rivers also carry many particulates, nutrients, and minerals which play a major role in maintaining the productivity of the water bodies. More than 99% of heavy metals entering into a river can be stored in river sediments in various ways (Salomons and Stigliani 1995). Heavy metals can fix in sediment for short periods. Physico-chemical characteristics of water conditions vary season to season. A small amount of these fixed heavy metals will re-enter the overlying water body and uptake by the aquatic biota. Rivers receive sediment from various points and diffused sources which deposited at the bottom of the river and acts as both carriers and potential sources of metal accumulation in aquatic food chain by the process of biomagnifications (Theofanis et al. 2001). Finally, the human health is adversely affected by uptaking of these by the fishes and water. Therefore, it is necessary to investigate the current status of heavy metals in sediments of the river Ghaghara.
Many activities such as land use/land cover change (Singh et al. 2014; Narsimlu et al. 2015; Kumar et al. 2017), sewage sludge, mine waste, industrial waste, waste water (Gautam et al. 2013), atmospheric pollutants, pesticides, and fertilizer applications are the main activities controlling the flow of river and contributors of heavy metals discharged into rivers, lakes, estuaries, and marines (Chen et al. 2004; Adaikpoh et al. 2005; Lepane and Heonsalu 2007; Amin et al. 2014; Singh et al. 2016, 2017). Beside these wastes, rivers also carry large quantities of solid wastes, including thousands of animal carcasses and hundreds of human corpses are dumped into the active channel of the rivers and on its banks every day in recent years. Nowadays, the condition is being worsened due to the discharge of toxic contaminants at large scale from industrial sources. The above-mentioned factors affect the productivity of the river ecosystem and use for irrigation of crops (Bharose et al. 2013) and also affect other life-support systems in various ways. It is very unfortunate for us, besides reducing the sources of pollutants, our interventions in river ecosystem are increasing day by day, which are creating a tremendous pressure on the river systems.
The heavy metals entering into the rivers from various sources are adsorbed onto suspended particulates and form free metal ions and soluble chemical complexes that are available for uptake by aquatic organisms (Salomons and Forstner 1984). Many toxic metals such as arsenic, lead, nickel, cadmium, copper, mercury, zinc, and chromium present in untreated or allegedly treated industrial effluents are carried by rivers in variable amounts (Singare et al. 2011). In previous studies, Indian workers such as Aghor (2007) and Patil (2009) have reported heavy metal accumulation by various components of river ecosystem, i.e., in fishes, oysters, sediments, and in others. In aquatic organisms, there is a greater affinity for gill, liver, and muscles to accumulate Pb, Cd, and Hg, respectively. It is well established that vertebrates and invertebrates are capable of accumulating heavy metals (Cd, Cu, Pb and Zn) from the aquatic environment (Hellou et al. 1992). Pesticides affect crustacean, fishes, and mollusks. Irrigation carried out with the contaminated water may increase the toxicity of the soil and crops growing along both the river banks.
Over the past 3–4 decades, studies on aquatic sediments have increasingly been carried out for assessing the geochemical transport of elements, especially nutrients and trace metals from the terrestrial environment to river bodies and eventually to the oceans. Some natural activities also generate heavy metals and many of them are entering into nearby water systems through erosion every year. The sediment toxicity indicators such as contamination factor, contamination degree, pollution load index, geo-accumulation index, and the enrichment factors used in the present investigation for evaluating the sediment contamination caused by heavy metals have been previously used worldwide (Ideriah et al. 2012; Özkan 2012; Moore et al. 2009). Many other workers such as Borole et al. (1982), Subramanian et al. (1985, 1987), Seralathan (1987), Ramesh et al. (1990), Chakrapani and Subramanian (1990), Singh et al. (1997), Kotoky et al. (1997), Singh (1999), Singh et al. (2013a, b, c) have been working on many other Indian rivers to investigate the geochemical environment of sediments. The present study of river sediments was carried out to assess the concentration of heavy metals of the river sediment.
The objectives of the current study is: (1) to assess heavy metal pollution in sediments, (2) to investigate the seasonal variations and degree of contamination and pollution using pollution indices as contamination factor (CF), contamination degree (CD), pollution load index (PLI) and geo-accumulation index (Igeo), and (3) to identify the similar pollution sites using cluster analysis.
The present study provides the first and useful data for the geochemistry of the river Ghaghara sediments and contamination status.
Materials and methods
Study area
Ghaghara River is a lifeline for the people of the districts of Bahraich, Gonda, Faizabad, Azamgarh, Mau Nath Bhanjan, Gorakhpur, Deoria, Ballia, and Saran in northeastern part of Uttar Pradesh and a small part of western Bihar. It is a major left bank tributary of the river Ganga. The total length of the river is about 1080 km and basin area is about 1,27,950 km2, whereas the average discharge is 2990 m3/s. Out of the total catchment area, India has only 57,647 km2, which is 45% of the total area. In the upper reaches, the river Ghaghara is known as the Manchu and Karnali in Nepal. It originates in the Himalayan glaciers near Lampia-pass at an elevation of 4800 m at latitude 30°38′N and longitude 80°57′E about 60 km southwest of Mansarovar. Out of total length, the upper half stretch of the river runs in Tibet and Nepal and the lower half is in India. The critical locations along the Ghaghara River in India, where major shifting has occurred are Tanda, Ayodhya, Golabazar, Barhaj, and Bansdih. Large scale mining of sand from the river course and variation in flow is the possible reasons for shifting in river Ghaghara. The river enters into the Indian Territory in Bahraich district of Uttar Pradesh at Kotia Ghat near Royal Bardia National Park, Nepal Ganj, where it is known as the river Girwa for about 25 km. The Sarda, most important tributary of the river Ghaghara join the Kaurial River in Rampur. After the junction with the Sarda, the river is known as the Ghaghara.
The soil of the Ghaghara river basin is alluvial and is exceedingly fertile because of the large quantity of soil transported with the water of the flood. The valley of the Ghaghara is wide up to 5–15 km in rainy season, whereas it turns into a narrow channel, i.e. 100–200 m throughout summer. The formation of the many large, middle channel bars, sidebar, natural dike, and flood plains are the precise geomorphic feature of the stream Ghaghara. The large braided bars confined are up to 5–10 km long and 1–5 km wide. The lateral bars are locally known as Diara land, and are used for agronomical activities and settlement. Braided bars are located within the active channel of the river, whereas lateral bars are side bars, denoting the curved braided stream which attached to the river bank (Bluck 1974). A narrow area of water slough can be seen just parallel to the active channel in the downstream stretch of the river (Reineck and Singh 1980); it shows lateral channel shifting filled with muddy sediments. Shifting of the river channel can be seen at many locations within the stretch, it is a specific feature of the river Ghaghara. The geo-morphologic facies and granulometric analyses suggest that the sand and silt are the principle constituents of the sediment of the river Ghaghara. The deposited sediment particles at the bottom of the river are non-cohesive and unconsolidated, in an effort to undergo weathering, transportation, and sliding (Singh and Rastogi 1973). In summer time and winter season, whilst the water discharge could be very low, the sandy facies are uncovered. The exposed areas of sediment particles are non-cohesive due to this dry sand with low degree of compaction slides by way of mass motion to acquire the steadiness. Sliding and elimination of sand from the river bank and bar deposits is an everyday phenomenon during low-discharge length, which leads to the slumping of the upper fine-grained clay unit and resulted in lateral erosion.
Description of sampling sites
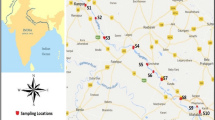
The research work was programmed in two consecutive phases, first a comprehensive survey was conducted to collect the sediment samples and secondly the samples were prepared and analyzed in the laboratory. Five sampling stations were selected for the study purposes. The undertaken river stretch was in Indian Territory from Katerniaghat, Bahraich (UP) to merging point of the River Ganga at Chhapra, Bihar (Fig. 1). To find the positions of the sampling stations using the GPS (GARMIN). The co-ordinates are presented in Table 1.
Katerniaghat is located near the Indo-Nepal border of India in the Bahraich district of Uttar Pradesh and is a sensitive ecosystem spread over an area of approximately 440 km2 with sal (Shorea robusta) and teak (Tectona grandis) forests, lush grasslands, and wetlands along with the ever flowing Ghaghara river. Girwa River is small tributary and is passed through a jungle known as the ‘Katerniaghat Wildlife Sanctuary’. Gharials (Gavialis gangeticus) and Muggar (Crocodile) are seen with basking at the sand bars on every side of the active river channel. Katerniaghat Wildlife sanctuary is an important breeding area of the Ghariyal which builds its nests on the sandbars. Turtle of many species will also be visible basking in the sand. There are several Gangetic Dolphin (Platanista gangetica) visible with jumping within the water of the river Ghaghara. As Census of India (2011), Baharaich had a population of 3,478,275.
Colonelganj is a historic place in Gonda District of Uttar Pradesh. Gonda is about 40 km East of Colonelganj. It is situated on the left bank of the river Saryu, which is about 2 km away from the main city. This river system is again called as Saryu when it enters into Ayodhya city. Presence of alluvial soil is a characteristic feature of this area.
Ayodhya is an ancient city of India. It is situated adjacent to Faizabad city. Ayodhya is located on the right bank of the river Saryu. Diffused source of pollution and some small-scale industries, tanneries, textile, vehicle, and battery-producing units are present in Ayodhya.
Dohrighat is a small town of the district Mau Nath Bhanjan, Uttar Pradesh, spread in a 5 km2 area. The district has fertile plains of the Ganges–Ghaghara Doab (a large land area between the two rivers). The total population of this district is 2,205,170. The district represents geographical characteristics of the Middle Gangetic plain. The soil is ‘Khacchari’ and ‘Khadar’ aforesaid by way of the local people, which is mainly the alluvial soil.
Chhapra is a left bank city of the district Saran and settled near the junction of the Ghaghara and the Ganges River. The district is surrounded by three rivers, namely Ganges, Ghaghara, and Gandak. The district has large fertile plains, but there are few depressions and marshes are also present, which create three broad natural divisions.
Sample collection
The periodic samplings were completed in rainy (August), winter (January), and summer (May) seasons (with three repeats) in the year 2014–2015. The stations were chosen to arbitrarily take over the previously prepared sampling maps (Fig. 1). The sediment samples were collected from 5 to 10 cm depth of the middle of the river or the bank of the running water channel. The sediment samples were collected in impenetrable polythene sacks as 500 gm for each situation with the help of the bed material sampler. All the collected samples were transported to the lab and air dried for 72 h, ground in a total mortar, sieved to <63 µm sediment fraction and homogenized before each weighing. Testing instruments were washed with double-distilled water and dried before use.
Preparation of sediment samples
A total digestion method (Allen et al. 1986; modified by Singh et al. 2010) was utilized to determine the seven heavy metals concentrations in which 2 gm of sediment sample was warmed with 20 ml of tri-acid mixture (HNO3, H2SO4 and HClO4) in the proportion of 5:1:1 in a Teflon measuring beaker at 80 °C for 4–5 h. At the point when the sediment totally digested and leaves a transparent solution, the sample was cooled to room temperature and after that it was filtered through Whatman No. 42 filter paper into a pre-cleaned 100-ml volumetric flask. These samples were specifically utilized for the investigation of Co, Cu, Cr, Cd, Ni, Zn, and Pb by atomic absorption spectrophotometer (AAS; iCE 3000 Series, show 3500 AAS, Thermo scientific, UK), fitted with a particular lamp of each metal utilizing proper drift blank. The chemicals utilized as a part of the research were acquired from E. Merck, Mumbai, India, of analytical grade and glasswares were completely cleaned with 10% HNO3, and finally cleaned with water before the utilization. The instrument was calibrated by running self-prepared standard solution of As, and also drift blanks got from Sisco Research Laboratories Pvt. Ltd., India. The standard stock solution was having the concentration of 1000 ppm. This solution was diluted up to the desired concentration to align the instrument. Exactness and precision of examination were kept up by maintaining the repeated investigation of the sample against Standard Reference Material (National Institute of Standard and Technology, SRM1570). The outcomes were found in 72% of the certified value. Quality control measures were conveyed to check the contamination and the reliability of the results obtained. The coefficients of variation of replicating the examination were analyzed in various determinations for testing the exactness of the investigation and variation below 10% were considered as correct.
Pollution index, statistical analysis, and cluster analysis
Index of geo-accumulation (Igeo)
Geo-accumulation index was introduced by Muller (1979). This index is used to assess the anthropogenic impact. This is calculated using the following equation:
where Cn is the measure of the metal concentration in the sediment, Bn is the background concentration of the element (average shale concentration has been given by Turekian and Wedepohl 1961), and 1.5 is the factor compensating background data (correction factor) due to the lithogenic effect (Taylor 1964).
The Igeo factor is not comparable to other indices of metal enrichment due to the nature of the Igeo calculation; it involves a log function and a background multiplication of 1.5. It is composed of seven grades (0–6) indicating various degrees of metal enrichment above the average shale value ranging from unpolluted to very high polluted sediment quality. Class 0 (uncontaminated): Igeo ≤0; Class 1 (uncontaminated to moderately contaminated): 0 < Igeo < 1; Class 2 (moderately contaminated sediment): 1 < Igeo < 2; Class 3 (moderately to strongly contaminated): 2 < Igeo < 3; Class 4 (strongly contaminated): 3 < Igeo < 4; Class 5 (strongly to extremely contaminated): 4 < Igeo < 5; Class 6 (extremely contaminated): 5 < Igeo. Class 6 is an open class and comprises all values of the index higher than class 5.
Metal contamination factor (CF)
Contamination factor was calculated by comparing the mean of trace metal concentration with average shale concentration given by Turekian and Wedepohl (1961), which is used as global standard reference for unpolluted sediment. CF for each metal was determined by the following equation:
Hakanson (1980) classified CF values into four grades, i.e., CF < 1 in class 1 with low CF, 1 ≤ CF < 3 in class 2 with moderate CF, 3 ≤ CF < 6 under class 3 with considerable CF and CF ≥ 6 kept in class 4 with very high CF.
Contamination degree (CD)
CD is the sum of all CF values of a particular sampling site. Ahdy and Khaled (2009) classified CD in terms of four grade ratings of sediments, i.e. CD < 6 follows the class 1 which shows low CD, 6 ≤ CD < 12 follows the class 2 it shows moderate CD, 12 ≤ CD < 24 follows the class 3 it shows considerable CD and CD ≥24 follows the class 4 with very high CD.
Pollution load index (PLI)
Pollution load index for each site was determined by the method proposed by Tomlinson et al. (1980). The PLI for a single site is the nth root of n number multiplying the factors (CF values) together. PLI for each site was determined by the following equation:
where CF is the contamination factor and n is the number of parameters. According to Mohiuddin et al. (2010), PLI = 0 indicates a perfect state of pollution; PLI = 1 points indicate only baseline levels of pollutants present and PLI >1 would indicate progressive deterioration of sites.
Statistical analysis and cluster analysis (CA)
For a data set, the mean is the sum of the total observations divided by the number of observations. It calculates the central position of the data. The standard deviation (SD) is measure of inconstancy, measuring the spread of the data and the relationship of the mean to rest of the data. If the data points are very close to the average, outcomes are genuinely uniform and the SD will be little and vice versa. The SD will be zero, if all the data points show equal values. Correlation is the study of relationship between two data sets. If two sets of data are strongly linked together (high correlation) correlation has a value (−1 to 1), ‘1’ means a perfect positive correlation, ‘0’ means no correlation, and ‘−1’ is a perfect negative correlation. Correlation between two sets of parameters (7 × 7), namely Co, Cu, Cr, Ni, Cd, Zn, and Pb has been studied. SPSS20 software was used to compute Pearson correlation coefficient matrix (at significant level of P < 0.05).
CA groups the objects into the classes on the basis of similarities within a class and dissimilarities between different classes. The results of CA help in interpreting the data and indicate patterns (Singh et al. 2009; Singh et al. 2013a, b; Gupta et al. 2014; Singh et al. 2015). The heavy metals of sediments data sets were used for hierarchical agglomerative CA and were performed on the normalized data set by means of Ward’s linkage method using squared Euclidean distances as a measure of similarity.
Results and discussion
Results are presented in Table 2. Concentration of individual metal at each site is illustrated in Fig. 2(a–g).
Overall Cobalt (Co) concentration was varied from 11.37 mg/kg to 18.42 mg/kg. The highest value of Co was measured at Chhapra and lowest at Katerniaghat. The average value of Co was recorded as 12.64 ± 4.12 mg/kg at Colonelganj, 15.54 ± 6.87 mg/kg at Ayodhya, and 13.81 ± 4.35 mg/kg at Dohrighat. In our past review for the stream Ganga the estimation of Co in sediment was 16.5 mg/kg at Ghazipur and 23.5 mg/kg at Buxar (Singh et al. 2013a). Fixations of Co in the sediment of the River Ghaghara in various sites are introduced in Table 2. Comparative estimates of Co concentration of the waterway of Ghaghara with different rivers of the world have been given in Table 5. According to United State Environment Protection Agency (USEPA) the level of Co should be 50 mg/kg. However, in the present study it was lower than the benchmark established by the USEPA, average shale value (ASV) which is 19 mg/kg (Turekian and Wedepohl 1961) and World River System (WRS) (Martin and Meybeck 1979) (Table 3). Geo-accumulation index (Igeo) was found in the review of class 0 which demonstrates that the sediment of the river is uncontaminated (Table 5). CF was found to be <1 which shows that the sediment is not polluted. Maximum CF value was measured as 0.97 at Chhapra while the minimum value was 0.62 at Katerniaghat (Table 6). The general outcome demonstrates that the stream sediment is not in disturbing condition for Co at present. However, it has little contamination, which may increase in the future and may be alarming to the river ecosystem with rapidly increasing population in the river basin.
The level of Copper (Cu) concentration was ranges between 2.76 and 11.74 mg/kg at all the considering stations in the study area, whereas the highest value of Cu was recorded at Chhapra which was 11.74 ± 4.55 mg/kg and it was followed by Ayodhya, Dohrighat, Katerniaghat, and Colonelganj. The measured estimations of Cu concentration at various destinations of the waterway of Ghaghara are displayed in the Table 2. According to United State Environment Protection Agency (USEPA), maximum permissible value for Cu in the river sediments is 31.6 mg/kg, average shale value (ASV) is 45 mg/kg and TRV (Toxicity Reference Value) is 16 mg/kg. The observed value for Cu was found below the permissible limit set up by USEPA (Table 3). CF of Cu was measured most noteworthy at Chhapra which was 0.26 and the least value was recorded as 0.04 at Ayodhya. Contamination Factor is <1 fall in the class 1 (Table 6). Igeo value was found in the grade of class 0 which indicates that the sediment of the river is uncontaminated (Table 5). The findings indicate that the river sediment is slightly contaminated and may be alarming in future for the river biota. In similar studies for the sediment of the river Ganga it has been found that the Cu was 49 mg/kg at Ghazipur and 58 mg/kg at Buxar (Singh et al. 2013a, b, c). Relative estimations of Cu concentration of the stream Ghaghara with different rivers of the World has been given in Table 5. It is imperative to note here that Cu is very dangerous to a large portion of the fishes, invertebrates, and aquatic plants than some other trace metals except mercury. Aquatic plants absorb three times more Cu than plants on dry lands (IISc 2001). The higher value of Cu substance can harm the plant roots by destroying the cell membrane structure, restrain root development and formation of many short auxiliary roots.
Cr concentration in the sediment of the River Ghaghara was recorded as 61.25 ± 15.25 mg/kg at Katerniaghat, 82.74 ± 20.49 mg/kg at Colonelganj, 87.68 ± 22.47 mg/kg at Ayodhya, 69.71 ± 29.40 mg/kg at Dohrighat, and 61.25 ± 15.25 mg/kg at Chhapra. Detail of the Cr concentrations in sediment of the river Ghaghara at different sites is presented in the Table 2. The overall Cr concentration ranged between 10.65 and 111.5 mg/kg for all the considering sites which exceeded the standard values set by USEPA, TRV, and IRS, while it was below the ASV and WRS (Table 3). The exceeded estimation of Cr can cause lethality to some aquatic species in the river system (USEPA 1999). Contamination factor demonstrates that the higher concentration of Cr was found in Ayodhya which is near to 1 and lies in class 1 (Table 6). The above result shows that the sediment of the stream has a low level of contamination. Contamination degree indicates that the sediment was more contaminated at Ayodhya in comparison to other sites. It may be harmful to the aquatic life. Igeo value for Cr was found between 0 and 1 and comes under the class 1, this demonstrates the sediment is uncontaminated to moderately contaminated. In our previous studies, it was found that the average value of the Cr for the river Ganga was 148 mg/kg at Ghazipur, 163 mg/kg at Buxar, and 161 mg/kg at Ballia in sediments of size 20 µm (Singh et al. 2013a, b, c). Similar estimations of Cr concentration of the stream Ghaghara with different rivers of the World have been given in Table 5. Chromium may go into the aquatic body through the small-scale tanneries, mining sites, natural origins, etc. (Howarth et al. 2005).
Chromium is available in the Earth’s crust layer with a concentration of 100 mg/kg. The only ore of chromium is chromite (FeO·Cr2O4) of commercial importance. Out of six oxidation conditions of Cr the trivalent (III) and hexavalent (VI) structures are thought to be of biological significance. In aquatic environment, Cr+6 will be present in soluble form and eventually converted to Cr+3, by reduction with hydrogen sulfide, iron sulfide, ammonium, and nitrate. Cr+6 and Cr+3 have been found accumulated in many aquatic species, especially in fishes such as Cyprinus carpio, Oreochromis niloticus, Aorichthys aor, Labeo calbasu, Cirrhinus mrigala, and Clarias batrachus. The hexavalent shape exists as a segment of an unpredictable anion that shifts with pH and may be changed over as chromate (CrO4 2−), hydrochromate (HCrO4 −), or dichromate (Cr2O7 2−). Cr6+ frame is highly dissolvable in water and consequently portable in the aquatic condition. Precipitated Cr+3 hydroxides are persisting in the sediments under aerobic conditions; under low pH and anoxic conditions, while Cr+3 hydroxides may be solubilized and remain as ionic Cr+3 unless oxidizes to Cr+6 through mixing and aeration (Ecological Analysts Inc. 1981). Acute toxicity of Cr to invertebrates is highly diversified depending upon the species (Moore and Ramamoorthy 1984). It was seen by the specialists of Indian Institute of Sciences, Bangalore (IISc 2001) that the harmful impact of Cr on plants demonstrates that the roots remain smaller, and the leaves seem to be reduced and display reddish brown staining with little necrotic blotches. The sources of emission of Cr in the surface waters are from municipal effluents, laundry chemicals, paints, leather, road runoff due to tire wear, corrosion of bushings, brake wires, radiators, etc. The abnormal state of Cr in wastewater effluent shows contamination from textile and tanneries (Pachpande and Ingle 2004) and when these effluents enter into the river, contaminate both the water and sediment of the river.
The Nickel (Ni) was ranged between 15.29 and 25.59 mg/kg for all the considering stations. The results showed that the estimation of Ni concentration in the sediment of the River Ghaghara was 15.29 ± 5.59 mg/kg at Katerniaghat, 20.16 ± 4.25 mg/kg at Colonelganj, 25.59 ± 10.48 mg/kg at Ayodhya, 16.41 ± 8.72 mg/kg at Dohrighat, 19.19 ± 7.81 mg/kg, and 18.19 ± 9.83 mg/kg at Chhapra. Level of Ni in river sediment is 22.7 mg/kg and TRV is 16 mg/kg (USEPA 1999, Table 5). However, the concentration of Ni was found either close or slightly above the permissible limit at most of the sites. Contamination factor was followed the class 1. It was highest at Ayodhya and followed by Colonelganj, Chhapra, Dohrighat, and Katerniaghat. This indicates the low level of the contamination of the river sediment. Igeo value follows the class zero. It indicates that river sediment of these sites is very close to the background value which may increase in future due to increasing anthropogenic addition. In similar studies for the sediment of the river Ganga it was found that the value of Ni was 42 mg/kg at Ghazipur, 59 mg/kg at Buxar, and 52 mg/kg at Ballia (Singh et al. 2013a). Ni concentration in sediments of the river Ghaghara is compared with other rivers of the World and is presented in Table 5. Ni concentration was 27.7 mg/kg for the river Cauvery (Raju et al. 2012), 67.1 mg/kg for the river Euphrates (Salah et al. 2012), 41.9 mg/kg for the river Yangtze (Wang et al. 2011), and 68 mg/kg was the World average (Turekian and Wedepohl 1961). Nickel can be accumulated in aquatic biota, but its magnification in the food chain is not confirmed. CF factor indicates that the Ni contamination follows the class 1 with low contamination level. It is maximum at Ayodhya. Ni in the aquatic body forms complexes with varying soluble organic and inorganic materials. It adsorbs directly on clay particles and has the capability to co-precipitate with hydroxides of iron and manganese. It is soluble at higher pH values and gets precipitated at pH 10. It is bio-accumulated through aquatic organisms such as phytoplanktons, seaweeds, and algae.
The average concentration of cadmium (Cd) in the sediment of the River Ghaghara was recorded as 0.21 ± 0.08 mg/kg at Katerniaghat, 0.23 ± 0.11 mg/kg at Colonelganj, 0.28 ± 0.10 mg/kg at Ayodhya, 0.25 ± 0.10 mg/kg at Dohrighat, and 0.24 ± 0.06 mg/kg at Chhapra. The Cd value in the present study was found to be less than the permissible value set up by USEPA and TRV (Table 5). However, it exceeded the estimation of the ASV. This can cause toxicity to some aquatic species in the vicinity (Turekian and Wedepohl 1961, Hakanson, 1980). CF index follows the class 1 which shows low level of contamination. As indicated by the metal contamination factor (CF) the river stretch near the Ayodhya is more polluted in comparison to the other undertaken sites. Igeo value followed the class zero, it demonstrates that sediment is near the background value. In our previous study of the river Ganga sediment quality (Singh et al. 2013a) using the same method, Cd concentration was 0.72 mg/kg at Ghazipur and 0.62 mg/kg at Buxar, which was slightly lower than the present study. Comparative estimates of Cd concentration of the stream Ghaghara with different rivers of the World have been listed in Table 5. Cd is a by-product of Zn and Pb mining and smelting, and more mobile in the aquatic environments than most of the other metals. It is also bio-accumulative and persistent in the environment. Cd is similar in toxicity to Pb and Cr whereas it is less toxic than Cu for the plants, while it is equally toxic to invertebrates and fishes (Moore and Ramamoorthy 1984). Cadmium is added to the surface waters through different sources such as paints, colors, glass finish, galvanized pipes, and so forth. Cd deposited on road surfaces from the studded tires. It is currently used for the production of nickel–cadmium batteries. Cadmium is more mobile in the waterway body than other metals. It is also bio-accumulative and persistent in the river body.
The average value of Zinc (Zn) concentration in the sediment of the River Ghaghara was recorded as 13.26 ± 6.75 mg/kg at Katerniaghat, 17.59 ± 4.59 mg/kg at Colonelganj, 15.11 ± 6.27 mg/kg at Ayodhya, 14.29 ± 4.74 mg/kg at Dohrighat, and 18.11 ± 6.67 mg/kg at Chhapra. The observed value of Zn was found below the permissible limit proposed by USEPA, ASV, TRV, and the WRS; however, it exceeded the limit of IRS (Table 5) which may cause adverse effect on aquatic biota. The metal contamination factor (CF) follows the class 1 grade which demonstrates that the residue of the stream has low-level contamination of zinc. CF for Zn was found to be higher at Chhapra and Colonelganj whereas it was comparatively lower (Table 4). This demonstrates the waterway sediment was marginally contaminated by Zn in the study area. In past reviews, the average estimate of Zn was recorded as 97 mg/kg at Ghazipur urban station, 109 mg/kg at Buxar, and 104 mg/kg at Ballia for the stream Ganga by Singh et al. (2013a). Relative estimations of Zn concentration of the stream Ghaghara with different rivers of the World have been given in Table 5. The main sources of Zn are smelting, fertilizers, and pesticides used in agriculture, soil erosion due to rainfall, fossil fuel, and land construction activities (Higgins et al. 2007; Xiang et al. 2002; Chen et al. 2004). Zinc is found on the earth’s crust in an average concentration of 80 mg/kg. It is often associated with the ores of other metals such as Cu, Pb, and Cd. In the aquatic body, it will predominantly combine with suspended materials before finally accumulating in the sediment. A solvent state (sulfate or chloride) is much more prone to move through the earth than a complex bound state as in natural matter or present in insoluble precipitate. Zinc is an essential metal having enzymatic and regulatory roles in biological systems. High concentration of Zn may bring about rot, chlorosis, and repress the development of plants, while gastrointestinal misery, loose bowels, pancreatic harm, and sickliness, in both people and animals.
Lead (Pb) fixation was recorded between 10.54 and 22.06 mg/kg among each of the sites. The average Pb concentration in the sediment of the River Ghaghara was recorded as 13.79 ± 4.32 mg/kg at Katerniaghat, 12.85 ± 4.04 mg/kg at Colonelganj, 14.26 ± 5.12 mg/kg at Ayodhya, 10.83 ± 5.23 mg/kg at Dohrighat, and 10.71 ± 6.21 mg/kg at Chhapra. The observed estimation of Pb in the present review was found below the permissible limit set up by USEPA, ASV, and TRV. CF indicates low level of Pb contamination in sediment of the river. The value of Pb was recorded highest at Ayodhya in the study area. This is presumably because of the presence of small-scale battery fabricating units at this site. Results indicate that the river sediment had not surpassed the danger level for Pb in the review territory. In our past reviews, (Singh et al. 2013a), the average estimate of Pb was recorded as 19 mg/kg at Ghazipur, at Buxar it was recorded as 19 mg/kg, while at Ballia the average estimate of Pb was recorded as 21 mg/kg. Comparative values of Pb concentration of the river Ghaghara with other rivers of the World have been given in Table 5. The overall Pb concentration is near the standard qualities set up by various ecological agencies; however, at present it is safe to the aquatic life, but rather it might be possibly destructive later on with expanding population in the stream basin.
Lead is one of the most established metals known to man and the greater part of its compounds is noxious in nature. It is found on the earth’s crust in an average concentration of 0.1 mg/kg. Pb binds with sulfide ores of Zn, Cu and Pb which is obtained as a by-product during the processing of these ores and is discharged in the surface water through paints, solders, pipes, building material, gasoline, etc. Pb is a metal toxicant and it is gradually being phased out of the materials that human beings regularly use. Combustion of oil and gasoline accounts for >50% of all anthropogenic emissions, and thus forms a major component of the global cycle of Pb. Higher levels of Pb pose eventual threat to fisheries resources, whereas, in plants, it initially results in enhanced growth, but from a concentration of 5 mg/kg onwards, this is counteracted by severe growth retardation, discoloration, and morphological abnormalities.
Geo-accumulation index (Igeo)
Igeo index demonstrated that the majority of the undertaken metals have index value below zero, whereas Cr has the Igeo value between 0 and 1, and falls in class 1 (Table 5). This indicates that the river sediment is uncontaminated at most of the sites by most of the considering metals; however, the sediment is slightly contaminated by Cr at Ayodhya and Colonelganj which may contribute to sediment toxicity in the river ecosystem of the river. Several negative Igeo values were noted for all considering metals at many sites and showed that the Ghaghara river bed sediment is uncontaminated for most of the trace metals in the study area. Previous study carried out by Singh et al. (2013a) on the Ganga river noted several negative Igeo values for Co, Ni, Cu, and Pb at various sites. Index of geo-accumulation permits assessment of the level of sediment contamination with respect to global standards. The geo-accumulation index was introduced by Muller (1979). The calculated Igeo class value was found between 0 or 1, the Igeo class values showed uncontaminated to moderately contaminated river sediments.
Metal contamination factor (CF) and contamination degree (CD)
Assessment of the anthropogenic effect on bed sediment quality has been ascertained by standardized contamination factor (CF) for metal concentration. CF is a mathematically computed record, contingent upon a direct extent between the concentration of the metals in the sample taken from the review area and earth crust (Hakanson 1980; Pekey et al. 2004). CF for Co was seen to be most extreme at Chhapra and it was followed by Ayodhya, Dohrighat, Colonelganj, and Katerniaghat, respectively (Table 6). Contamination factor for Cu was highest among all the sites at Chhapra which was 0.26 and it was followed by Dohrighat, Katerniaghat, Colonelganj, and Ayodhya. However, the class 1 of CF grade was followed. Metals such as Cr, Ni, Pb, and Cd also followed the CF class 1 and it was the highest at Ayodhya, while Zn was the highest at Chhapra and Colonelganj. The overall study indicates that the CF followed class 1 and river sediment has a low level of contamination. Contamination degree was found highest at Ayodhya and lowest at Colonelganj (Table 6). CD also suggested that sediment of the river falls in class 1, which indicates that sediment has a low level of contamination.
Pollution load index (PLI)
The PLI is used to assess the overall toxicity and quality status of the samples and furthermore it is a consequence of the contribution of the few metals (Tomlinson et al. 1980). The PLI of the sediment of the stream is presented in Table 3. PLI values of sediments of all the studied sites ranged between 0.35 and 0.45. The lowest PLI value was recorded at Katerniaghat which has low anthropogenic activity at upstream, but increasing the population load in the downstream stretch of the river, gradually increasing the anthropogenic input, results in the highest PLI value, which was recorded at Chhapra. The overall pollution load is highest at Chhapra and lowest at Katerniaghat. PLI can give some comprehension to the general population about the nature of river bed sediment.
Correlation coefficient and cluster analysis
Heavy metals available in the sediment of the stream Ghaghara indicate the different level of correlation with each other at a significant level of P < 0.05 (Table 7). Co demonstrates a high level of positive relationship with Cu and a moderate degree with Zn though it is adversely associated with Cd and Pb. The correlation matrix demonstrates that Cu has a low level of relationship with Ni, Cd, and Zn though it was adversely associated with Cr and Pb. It was noticed that Cr was emphatically corresponded with Ni and low degree with Zn and Pb while it was negatively correlated with Cd. The Cd is negatively associated with Co, Cr, Z, Pb, and Ni except Cu. Zn shows a moderate degree of positive correlation with Co whereas it has a low degree of positive correlation with Cu, Cr, and Ni. Zn is negatively correlated with Cd. Lead shows low-degree positive correlation with Cr and Ni. But it is negatively correlated with Co, Cu, Cd, and Zn. Good correlation between Co, Cu, and Zn with each other could be suggested from common anthropogenic sources, and that they can be affected by possible additions. While positive relationship between Zn, Co, Cu, Cr, and Ni demonstrated a characteristic natural origin of these components in the waterway sediment. As per Suresh et al. (2011), if the relationship coefficient between the considering metals is higher, metals have a common source of origin with mutual dependence and similar behavior during transport. Weak correlation among other metals recommends that the substance of these metals are not controlled by any single element, but rather it is controlled by a combination of geochemical support and associations.
The cluster analysis was performed using standard method as Squared Euclidean Distance and Ward’s linkage method (Singh et al. 2015). Based on Fig. 3 two clusters (groups) with different hydrogeochemical characteristic can be distinguished. The first cluster includes locations of Ayodhya_winter and all the Katerniaghat_summer, Colonelganj_summer, Ayodhya_summer, Dohrighat_summer, and Chhapra_summer. The second cluster has rest sampling sites of different season. This grouping gives evidence that some sites have similar sources of pollution from point or nonpoint sources. These sites are polluted by industries and agriculture activities.
Conclusion
Results suggest the regular pattern of heavy metal distribution. Indices namely Igeo, CF, and CD were used to assess the metal enrichment in the sediment which demonstrates that the river sediment has a low level of contamination all through the review territory. Among all of the studied sites Ayodhya has the highest metal contamination degrees (CD) but still beyond the first grade limit. CD shows all the studied sites fall into the non-contaminated category. However, PLI showed that the level of overall sediment pollution is highest at Chhapra. Because of the absence of large-scale industries and availability of some small-scale units in the study zone, all sources of the contaminations are not anthropogenic, but rather the normal weathering and disintegration is likewise to add to the current accumulation. The presence of natural and anthropogenic sources of heavy metals input was confirmed by the correlation. The contents of some heavy metals are higher than the average shale value and toxicological reference values which may cause additional adverse health risks to the aquatic ecosystem associated with the river. The experimental findings demonstrate that the river bed sediment is not contaminated except some points; it might alter with the expanding population load in the Middle Ganga Plain.
References
Adaikpoh E, Nwaijei G, Ogala J (2005) Heavy metal concentration in coal and sediment from River kulu in Enugu, coal city of Nigeria. J App Sci Env Mgt 9:5–8
Aghor (2007) ‘Chemicals make Thane creek the worst polluted water body’. Daily DNA. August 14, Mumbai, India. Available: http://www.dnaindia.com/mumbai/report_chemicals make thane creek the-worst polluted water body 1115439. Accessed 03 Feb 2016
Ahdy HHH, Khaled A (2009) Heavy metals contamination in sediments of the western part of the Egyptian Mediterranean sea. Aust J Basic Appl Sci 3:3330–3336
Allen SE, Grimshaw HM, Rowland AP (1986) Chemical analysis. In: Moore PD, Chapman SB (eds) Methods in plant ecology. Blackwell Scientific Publication, Oxford, London, pp 285–344
Amin A, Fazal S, Mujtaba A, Singh SK (2014) Effects of land transformation on water quality of Dal lake, Srinagar, India. J Indian Soc Remote Sens 42:119–128
Bharose R, Singh SK, Srivastava PK (2013) Heavy metals pollution in soil-water-vegetation continuum irrigated with ground water and untreated sewage 1. Bull Environ Sci Res. 2:1–8
Bluck BJ (1974) Structure and directional properties of some valley sand deposits of southern Iceland. Sedimentology 21:533–554
Borole DV, Sarin MM, Somayajulu BLK (1982) Composition of Narmada and Tapti Estuarine particles and adjacent Arabian sea sediments. Ind J Mar Sci 11:51–62
Centre for Ecological Sciences, IISc environmental hand-book—documentation on monitoring and evaluating environmental impacts, compendium of environmental standards, vol. 3, Indian Institute of Science, Bangalore (2001). Available http://bis.ces.iisc.ernet.in/energy/HC270799/HDL/ENV/START.HTM
Chakrapani GJ, Subramanian V (1990) Preliminary studies on the geochemistry of the Mahanadi river basin, India. Chem Geol 70:247–266
Chen Z, Saito Y, Kanai Y, Wei T, Li L, Yao H (2004) Low concentration of heavy metals in the Yangtze estuary sediments, China: a diluting setting. J Estuar Coast Shelf Sci 60:91–100
Dong ZR (2003) Diversity of river morphology and diversity of bio-communities. J Hydraul Eng 48(11):1e6 (in Chinese)
Ecological Analysts Inc (1981) The sources, chemistry, fate, and effects of chromium in aquatic environments. Avail. From American Petroleum Institute, 2101 L St., NW, Washington, DC 20037, p 207
Gautam SK, Sharma D, Tripathi JK, Singh SK, Ahirwar S (2013) A study of the effectiveness of sewage treatment plants in Delhi region. Appl Water Sci 3:57–65
Gupta LN, Avtar R, Kumar P, Gupta GS, Verma RL, Sahu N, Sil S, Jayaraman A, Roychowdhury K, Mutisya E, Singh SK (2014) A multivariate approach for water quality assessment of river Mandakini in Chitrakoot, India. J Water Resour Hydraul Eng 3:22–29
Hakanson L (1980) An ecological risk index for aquatic pollution control, a sedimentological approach. Water Res 14(8):975–1001
Hellou J, Warren WG, Payne JF, Belkhode S, Lobel P (1992) Heavy metals and other elements in three tissues of cod, Gadus morhua from the Northwest Atlantic. Mar Poll Bull 24:452–458
Higgins SA, Jaffe BE, Fuller CC (2007) Reconstructing sediment age profiles from historical bathymetry change in San Pablo bay, California. Estuar Coast Shelf Sci 73:165–174
Howarth R, Evans G, Croudace IW, Cundy AB (2005) Sources and timing of anthropogenic pollution in the Ensenada de San Simon (inner Ria de Vigo), Galicia, NW Spain: an application of mixture-modeling and non linear optimization to recent sedimentation. J Sci Total Environ 340:149–176
Ideriah TJK, David-Omiema S, Ogbonna DN (2012) Distribution of heavy metals in water and sediment along Abonnema Shoreline, Nigeria. Res Environ 2(1):33–40
Jain CK (2004) Metal fractionation study on bed sediments of River Yamuna, India. Water Res 38(3):569–578
Kotoky P, Baruah J, Baruah NK, Sarma JN (1997) Geoenvironmental studies of the river Jhanji, Assam. J Hum Ecol Special Issue No 6:55–67
Kumar N, Singh SK, Srivastava PK, Narsimlu B (2017) SWAT model calibration and uncertainty analysis for streamflow prediction of the tons river basin, India, using sequential uncertainty fitting (SUFI-2) algorithm. Model Earth Syst Environ 3:1–13
Lepane VV, Heonsalu A (2007) Sedimentary record of heavy metals in Lake Rouge Liinjarv, southern Estonia. J Estonian Earth Sci 56(4):221–232
Marathe R, Marathe Y, Sawant C, Shrivastava V (2011) Detection of trace metals in surface sediment of Tapti River: a case study. Arch Appl Sci Res 3(2):472–476
Martin J, Meybeck M (1979) Elemental mass-balance of material carried by major world rivers. Mar Chem 7(3):178–206. doi:10.1016/0304-4203(79)90039-2
Meybeck M (1976) Total annual dissolved transport by world major rivers. Hydrol Sci Bull 30:265–289
Milliman JD, Meade RH (1983) World wide delivery of river sediments to the Ocean. J Geol 91:1–21
Mohiuddin KM, Zakir HM, Otomo K, Sharmin S, Shikazono N (2010) Geochemical distribution of trace metal pollutants in water and sediments of downstream of an urban river. Int J Environ Sci Technol 7(1):17–28
Moore JW, Ramamoorthy S (1984) Heavy metals in natural waters: applied monitoring and impact assessment. Springer, New York, pp 28–246
Moore F, Forghani G, Qilshlaqi A (2009) Assesment of heavy metal contamination in water and surface sediments of the Maharlu saline Lake, SW, Iran. Iran J Sci Technol Trans A 33:43–55
Muller G (1979) Schwermetalle in den sedimenten des Rheins-Veranderungen seit 1971. Umschau 79(24):778–783
Narsimlu B, Gosain AK, Chahar BR, Singh SK, Srivastava PK (2015) SWAT model calibration and uncertainty analysis for streamflow prediction in the Kunwari River Basin, India, using sequential uncertainty fitting. Environ Process 2:79–95
Özkan EY (2012) A new assessment of heavy metal contaminations in an eutrophicated bay (Inner Izmir Bay, Turkey). Turk J Fish Aquat Sci 12:135–147
Pachpande BG, Ingle ST (2004) Recovery of the chromium by chemical precipitation from tannery effluent. Orient J Chem 20(1):117–123
Patil D (2009) A lot’s fishy about our creek and lake fish’. Daily-Times of India. March 22, 2009. Mumbai, India. Available: http://timesofindia.indiatimes.com/city/thane/A-lots- fishy-about-our-creek-and-lake-fish/articleshow/4298566.cms
Raju KV, Somashekar R, Prakash K (2012) Heavy metal status of sediment in river Cauvery, Karnataka. Environ Monit Assess 184(1):361–373. doi:10.1007/s10661-011-1973-2
Ramesh R, Subramanian V, Van Grieken R (1990) Heavy metal distribution in sediments of Krishna river basin, India. Environ Geol Water Sci 15:303–324
Rawat KS, Bala A, Singh SK, Pal RK (2017) Quantification of wheat crop evapotranspiration and mapping: a case study from Bhiwani District of Haryana, India. Agric Water Manag 187:200–209
Reineck HE, Singh IB (1980) Depositional sedimentary environments. Springer, Berlin
Saha P, Hossain M (2010) “Assessment of heavy metal concentration and sediment quality in the Buriganga River, Bangladesh.” International Proceedings of Chemi-cal, Biological and Environmental Engineering, Singa-pore city, 26–28 Feb, pp VI-384–VI-387
Salomons W, Forstner U (1984) Metals in the hydrocycle’. Springer, New York
Salah E, Zaidan T, Al-Rawi A (2012) Assessment of heavy metals pollution in the sediments of Euphrates River, Iraq. J Water Res Prot 4:1009–1023
Salomons W, Stigliani WM (1995) Biogeodynamics of pollutants in soils and sediments: risk assessment of delayed and non-linear responses. Springer, New York, pp 331–343
Seralathan P (1987) Trace element geochemistry of modern deltaic sediments of the Cauvery river, east coast of India. Ind J Mar Sci 16:235–239
Singare PU, Lokhande RS, Jagtap AG (2011) Water pollution by discharge effluents from Gove Industrial Area of Maharashtra, India: dispersion of heavy metals and their Toxic effects. Int J Global Environ Issues 11(01):28–36
Singh AK (1999) Elemental composition of the Damodar river sediments—a tributary of the Lower Ganga, India. J Geol Soc India 53:219–231
Singh IB, Rastogi SP (1973) Tectonic framework of Gangetic alluvium, with special reference to Ganga River in Uttar Pradesh. Curr Sci 42:305–307
Singh M, Ansari AA, Muller G, Singh IB (1997) Heavy metals in freshly deposited sediments of the Gomti river (a tributary of the Ganga river): effects of human activities. Environ Geol 29:246–252
Singh S, Singh C, Kumar K, Gupta R, Mukherjee S (2009) Spatial-temporal monitoring of groundwater using multivariate statistical techniques in Bareilly district of Uttar Pradesh, India. J Hydrol Hydromech 57:45–54
Singh A, Sharma RK, Agrawal M, Marshall FM (2010) Health risk assessment of heavy metals via dietary intake of food stuffs from the wast water irrigated site of a dry tropical area of India. Food Chem Toxicol 48:611–619
Singh H, Yadav S, Singh BK, Dubey B, Tripathi K, Srivastava V, Shukla DN (2013a) Assessment of geochemical environment from study of River sediments in the middle stretch of River Ganga at Ghazipur, Buxar and Ballia area’. Proc Natl Acad Sci India Sect B Biol Sci 83(3):371–384. doi:10.1007/s40011-012-0134-x
Singh D, Gautam PK, Gautam IK, Singh SK (2013b) Assessment of Ganga River water quality using multivariate statistical techniques. In: Dwivedi PK, Singh SN (eds) Technol Innov through Mod Eng Sci. Savera Publishing House, New Delhi, India, pp 36–38
Singh SK, Srivastava PK, Pandey AC, Gautam SK (2013c) Integrated assessment of groundwater influenced by a confluence river system: concurrence with remote sensing and geochemical modelling. Water Resour Manag 27:4291–4313
Singh SK, Srivastava K, Gupta M, Thakur K, Mukherjee S (2014) Appraisal of land use/land cover of mangrove forest ecosystem using support vector machine. Environ Earth Sci 71:2245–2255
Singh SK, Srivastava PK, Singh D, Han D, Gautam SK, Pandey AC (2015) Modeling groundwater quality over a humid subtropical region using numerical indices, earth observation datasets, and X-ray diffraction technique: a case study of Allahabad district, India. Environ Geochem Health 37:157–180
Singh SK, Singh Prafull, Gautam SK (2016) Appraisal of urban lake water quality through numerical index, multivariate statistics and earth observation data sets. Int J Environ Sci Technol 13:445–456
Singh H, Singh D, Singh SK, Shukla DN (2017) Assessment of river water quality and ecological diversity through multivariate statistical techniques, and earth observation dataset of rivers Ghaghara and Gandak, India. Int J River Basin Manag, pp 1–14. doi:10.1080/15715124.2017.1300159
Siqueira GW (2003). Study of heavy metals levels and other elements in surface sediments at the Santos Estuarine System and Shelf Coastal of the Amazon. PhD thesis, São Paulo University, Instituto Oceanográfico, Brazil, p 386 (in Portuguese)
Subramanian V, Van’t Dack L, Grieken V (1985) Chemical composition of river sediments from the Indian subcontinent. Chem Geol 48:271–279
Subramanian V, Sitaswad R, Abbas N, Jha PK (1987) Environmental geology of the Ganga River basin. J Geol Soc India 30:335–355
Suresh G, Ramasamy V, Meenakshisundaram V, Venkatachalapathy R, Ponnu-samy V (2011) Influence of mineralogical and heavy metal composition on natural radionuclide contents in the river sediments. Appl Radiat Isot 69:1466–1474
Taylor SR (1964) Abundance of chemical element in the chemical element in the continental crust-A new table. Geochem Cosmochim Acta 28:1273–1285
Theofanis ZU, Astrid S, Lidia G, Calmano WG (2001) Contaminants in sediments: remobilisation and demobilization. Sci Total Environ 266:195–202
Tomlinson DC, Wilson JG, Harris CR, Jeffrey DW (1980) Problems in assessment of heavy metals in the estuaries and the formation of pollution index. Helgol Mar Res 33:566–575
Turekian KK, Wedepohl DH (1961) Distribution of the element in some major units of the earth’s crust. Bull Geol Soc Am 72:175–192
USEPA (1999) National recommended water quality criteria-correction-United State Environmental Protection Agency EPA 822-Z-99-001, (http://www.epa.gov./ostwater/pci/revcom), pp 25
Wang Y, Yang Z, Shen Z, Tang Z, Niu J, Gao F (2011) Assessment of heavy metals in sediments from a typical catchment of the Yangtze River, China. Environ Monit Assess 172(1–4):407–417
Xiang L, Lu XX, Higgitt DL, Wang SM (2002) Recent lake Sedimentation in the Middle and Lower Yangtze basin inferred from Cs-137 and Pb-210 measurements. J Asian Earth Sci 21:77
Acknowledgements
The authors wish to thank the University Grants Commission (UGC), New Delhi for financial assistance, Prof. Sheo Mohan Prasad, Department of Botany, University of Allahabad, Allahabad, India for help in heavy metal analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Singh, H., Pandey, R., Singh, S.K. et al. Assessment of heavy metal contamination in the sediment of the River Ghaghara, a major tributary of the River Ganga in Northern India. Appl Water Sci 7, 4133–4149 (2017). https://doi.org/10.1007/s13201-017-0572-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13201-017-0572-y