Abstract
Drinking water quality from two major treatment plants in Ghana; Kpong and Weija Plants, and distribution networks in the Accra-Tema Metropolis were monitored monthly for a year at fifteen different locations. The study determined the relationship between chlorine residual, other physico-chemical qualities of the treated water, and, bacteria regrowth. Results indicated that the treated water at the Kpong and Weija Treatment Plants conformed to WHO guidelines for potable water. However, the water quality deteriorated bacteriologically, from the plants to the delivery points with high numbers of indicator and opportunistic pathogens. This could be due to inadequate disinfection residual, biofilms or accidental point source contamination by broken pipes, installation and repair works. The mean turbidity ranged from 1.6 to 2.4 NTU; pH varied from 6.8 to 7.4; conductivity fluctuated from 71.1 to 293 μS/cm. Chlorine residual ranged from 0.13 to 1.35 mg/l. High residual chlorine was observed at the treatment plants, which decreased further from the plants. Results showed that additional chlorination does not take place at the booster stations. Chlorine showed inverse relationship with microbial counts. Total coliform bacteria ranged from 0 to 248 cfu/100 ml, and faecal coliform values varied from 0 to 128 cfu/100 ml. Other microorganisms observed in the treated water included Aeromonas spp., Clostridium spp. and Pseudomonas spp. Boiling water in the household before consumption will reduce water-related health risks.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Access to safe drinking water is a fundamental human need and, therefore, a basic right. Contaminated water jeopardizes both the physical and social health of all people and it is an affront to human dignity (Craun et al. 2002). The purpose of a water supply distribution system is to deliver safe potable water which is also adequate in quantity and acceptable in terms of taste, odour, and appearance.
A significant proportion of the world’s population uses potable water for drinking, cooking, personal and home hygiene (WHO 2004). However, there has been a growing concern among the general public with respect to the safety and aesthetic qualities of potable water supplies. In extreme cases, customers may avoid aesthetically unacceptable but otherwise safe drinking water in favour of more pleasant but potentially unsafe source (WHO 2011). Potable water released into the distribution system becomes altered during its passage through pipes, open reservoirs, standpipes and storage tanks.
Bacteria may enter the distribution system through failure to disinfect water or maintenance of adequate disinfection residual, low pipeline water pressure, intermittent service, excessive network leakages, corrosion of parts, and inadequate sewage disposal (Lee and Schwab 2005).
Treatment of raw water will result in a decrease in microbial load, with many distribution systems later experiencing an increase in bacterial numbers with distance away from the point of treatment. The deterioration in water quality occurs either because of regrowth of microorganisms in biofilms, which are formed on interior surfaces of water pipes, or because of back siphonage of contaminated water. Biological activity in biofilms is controlled by nutrient content of water, temperature and residual chlorine (Gatel et al. 2000).
The regrowth of bacteria within any water distribution system is of concern to water authorities for a variety of reasons. High numbers of environmental coliforms undermine the validity of routine sampling programs and often lower annual water quality results into the non-compliance categories. Many microorganisms found in distribution systems also have the capacity to behave as opportunistic pathogens. Aeromonas have been detected in many treated drinking water supplies mainly as a result of regrowth in distribution systems (WHO 2011).
Water distribution systems play a pivotal role in preserving and providing quality water to the public. Little is known about the movement of contaminants, particulates, and disinfectants within the distributions system (Ndambuki 2006). Chlorine has been widely used as disinfectant due to its low cost and effectiveness in many countries. It is added to drinking water to reduce or eliminate pathogenic microorganisms and frequency of occurrence at the consumer’s tap. Addition of chlorine in different water treatment plants is a common practice, but it is not sufficient to ensure the safety of water. Regular testing is essential to ensure that adequate free residual chlorine is still present in the treated water. The maintenance of chlorine residue is needed at all points in distribution system supplied with chlorine as a disinfectant (Munavalli and Mohan Kumar 2003).
Chlorine residuals of drinking water have long been recognized as an excellent indicator for studying water quality in the distribution network (Lienyao et al. 2004). In the absence of a disinfectant residual, microorganisms in the distribution network will be recovered at high levels. Keeping residual chlorine at a certain level in tap water is effective not only in improving sanitary conditions but also in suppressing the regrowth of microorganisms and preventing the formation of biofilms on the internal surface of distribution pipelines (Kitazawa 2006).
In Ghana there is no regular independent monitoring programme to assess the water quality at the treatment plants and the distribution system. Earlier studies by Karikari and Ampofo (2006) showed contamination by faecal coliforms in drinking water from Accra-Tema Metropolis.
A range of physico-chemical parameters together with coliforms and non-coliform bacteria may be involved in regrowth providing possible health risk. The objective of this study is to determine the relationship between bacteria regrowth, chlorine residual and physico-chemical parameters in water supply distribution network to ensure good water quality in Accra-Tema to reduce possible health risks.
Materials and methods
Study area
Accra, the capital of Ghana, is located along the Atlantic Ocean and covers an area of 241 km2 (Abraham et al. 2007). In 2000 it had an estimated population of 1.66 million with annual growth rate of about 3.4 %. Beyond the municipal boundary of the Accra Metropolitan Area (AMA) is the adjoining district of Tema with a faster growth rate of 9.2 %. Total water supplied to Accra is 11.5 million m3 per month (inclusive of 30 % leakage losses) by the Ghana Water Company Limited (GWCL). Water supply coverage to the City is said to be 75 % but this does not imply a house connection. Only 45 % of the population has a household or at best a yard connection and this category includes the urban rich (Van Rooijen et al. 2008).
Since June 2006 urban water supplies in Ghana, including that of Accra, are under a 5-year management contract arrangement between GWCL as the Grantor and Aqua Vitens Rand Limited (AVRL) as the Operator. GWCL owns the physical assets and is also responsible for asset development and performance monitoring of the Operator whilst the day to day operations (Abstraction of raw water, production, distribution, maintenance and customer services) of water systems and personnel management in a service area are catered for by AVRL.
Potable water supply to Accra originates from Weija headworks located 17 km and Kpong headworks 110 km away from Accra. The Weija and Kpong headworks, respectively, take their source water from Densu and Volta Rivers and also supply an annual volume of 64 and 73 million m3 of potable water, respectively. The piped supply network covers only 36 % of the AMA municipal area.
Sampling sites
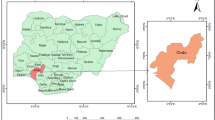
To assess the current status of water quality in Accra-Tema distribution system, water samples were collected from 15 different locations and monitored monthly for a period of 1 year from February 2008 to January 2009 (Fig. 1). These include Weija treatment plant which supply water to the west of Accra and Kpong treatment plant, supplying water to the east of Accra and Tema. At both treatment works, grab samples were collected in the day time from the laboratories taps at each visit. The distribution line is a combination of fibre glass, steel pipe, asbestos cement, high density polyethylene (HDPE) and polyvinyl chloride (PVC) pipes. The sampling sites were chosen based on the Public Utilities Regulatory Commission (PURC) of Ghana’s designated stations for independent assessment of water quality in Accra-Tema Metropolis and are mentioned in Table 1. Each particular station was sampled about the same time during the entire sampling period. GPS of each station was taken and the coordinates superimposed on the topographic field sheet of Survey of Ghana with a scale of 1:50,000 to generate the map of the study area (Fig. 1).
Chemical analysis
In the field, pH was measured by portable pH meter, Cyberscan pH 300 series. Analysis of chlorine residual was carried out using Palintest Chlorine DPD Reagent. For the detection of free chlorine, a sachet of DPD No. 1 was added to a 10 ml sample and development of a red colour was directly read at transmittance-display photometer (520 nm) and free chlorine determined using calibration chart.
Water samples for other physico-chemical analyses in the laboratory were collected directly into clean 1 l plastic bottles. Water samples for metal analyses were collected into 100 ml plastic bottles and nitric acid (HNO3) added to the samples to bring the pH down to 2 for the metals to remain in solution. All samples were stored in an ice chest and transported to the CSIR Water Research Institute’s laboratory in Accra for analyses.
The physico-chemical parameters were determined according to procedures outlined in the Standard Methods for the Examination of Water and Wastewater (APHA et al. 1998). Conductivity was measured with Cybersan 510 conductivity meter; turbidity with a HACH 2100 P Turbidimeter. Iron (Fe) and manganese (Mn) were measured by atomic absorption spectrophotometer.
Bacteriological analyses
In order to assess the types of bacteria the consumer actually ingests, the taps were sanitized before sample collection. Water samples were collected aseptically in sterile 0.5-l glass bottles from the tap. Each bottle contained 2 ml of 10 % sterile sodium thiosulphate to neutralize any residual chlorine. Samples were stored in an ice chest for transport to the laboratory for immediate processing. All bacteriological analyses were carried out according to the Standard Method for the Examination of Water and Wastewater (APHA et al. 1998). Aeromonas hydrophila,Pseudomonas aeruginosa, and total and faecal coliforms were enumerated by the membrane filtration method (0.45 mm, Gelman Science, Ann Arbor, MI, USA) using the Aeromonas agar, Pseudomonas agar base (Oxoid, Hampshire, Eng) and ECC agar (Chromagar, Paris, France), respectively.
Statistical analysis
Pearson’s correlation (two-tailed) analysis of the results was carried out to determine if there was any relationship between variables. Relationships were considered for significance at P < 0.05. Average values for each parameter and station were used. SPSS version 13 for Windows Package (2004) was employed.
Results and discussion
Physical characteristics
The water samples were examined for different physical constituents. Mean pH value observed in the water samples varied from 6.8 to 7.4 as shown in Tables 2 and 3. Most water treatment processes, particularly clarification and disinfection, require pH control to optimize the efficacy of the process. The World Health Organisation (WHO) proposes a treatment pH value of <8.0 for effective disinfection with chlorine. All the values obtained were within the WHO (2011) limit of 6.5–8.5 for potability. These results are also in accordance with the earlier study conducted by Karikari and Ampofo (2006) in which the pH values varied from 6.5 to 7.8
Mean turbidity ranged from 1.6 to 2.4 NTU. Turbidity consists of suspended particles in water and may be caused by a number of materials, organic or inorganic. The microbiological quality of drinking water can be affected by turbidity. Turbidity provides adsorption sites for biological organisms (some of them may be pathogens) and interferes with disinfection. Turbidities above 5 NTU may be discernible to consumers. The turbidity values observed in this study may be acceptable.
Conductivity value varied from 71.1 to 124 and 276 to 293 μS/cm for Kpong and Weija sources, respectively. The measurement of conductivity provides an indication of the total dissolved solids in the water. Conductivity is higher for water that has more dissolved ionic species. Waters from Weija source have higher conductivities, implying more dissolved substances.
Chlorine residual
The use of chlorine is essential for ensuring that water is bacteriologically safe to drink unless other reliable means of disinfection (e.g. boiling) can be used. Chlorine residuals of drinking water have long been recognized as an excellent indicator for studying water quality in the distribution network (Lienyao et al. 2004). The mean free chlorine value for this study varied from the lowest value of 0.13 to 1.4 mg/l. The treatment plants apply a chlorine dosage of 2 mg/l for disinfection. The recommended chlorine residual for water that is centrally treated at the point of delivery should fall within 0.2–0.5 mg/l (WHO 2011). The target for free chlorine residuals set by Ghana Water Company in the distribution network is 0.5 mg/l. In this study, free chlorine residuals were obtained at all the sampling sites, with most of them below the set target of 0.5 mg/l. The maintenance of chlorine residue is needed at all points in distribution system to prevent microbial growth (Munavalli and Mohan Kumar 2003). If no residual disinfectant is present, pathogens survive in drinking water distribution pipelines and are delivered to consumers at the tap.
It was observed that the free chlorine levels at the booster stations and beyond were not very different from other stations which suggest that chlorine is not frequently applied at the booster stations. Though, residual chlorine was detected in the waters at various stations, the presence of bacteria was also encountered.
Iron and manganese
Iron and manganese impart colour to water and render it undesirable for drinking purposes. Iron is essential element in the human diet. The concentrations usually found in drinking water are not harmful but iron can impart a bitter taste when present above 1 mg/l.
Even at lower concentrations it may cause ‘dirty’ water problems with consumers rejecting the water on the grounds of appearance. It can also cause brown stains on laundry and plumbing fixtures. Iron levels in this study ranged from 0.050 to 0.149 mg/l at S13. The WHO (2011) guideline value is 0.3 mg/l to avoid discolouration and staining.
Manganese is aesthetically undesirable in drinking water, even in small quantities, as it coats the interior of the distribution mains with a black slime, giving rise to consumer complaints. The WHO has set a guideline (2011) value for manganese of 0.4 mg/l on health grounds but recommends a much lower level of 0.1 mg/l on the basis of staining laundry and sanitary ware. Manganese concentrations observed in the distribution network varied between 0.010 and 0.049 mg/l. These values are satisfactory.
Microbiological characteristics
Presence of coliform bacteria, total viable count, Aeromonas spp., Clostridium spp. and Pseudomonas spp. were used as indication of possible presence of pathogens in the treated water in the distribution systems. Coliform bacteria were detected in all the 15 sampling points. Aeromonas spp. was not detected at the two water treatment plants and the first stations receiving water from the treatment works.
Water from S1, generally, had lower values of bacterial contaminants. Levels of faecal coliform (FC) bacteria detected in the water samples from S1 ranged from 0 to 15 cfu/100 ml. The highest bacterial counts were detected in the samples from S4 with faecal coliform bacteria ranging from 0 to 128 cfu/100 ml.
Total coliform (TC) counts for treated water from the Weija Treatment Works, S10 ranged from 0 to 56 cfu/100 ml, with the highest being recorded at the S12. Faecal coliform counts ranged from 0 to 7.0 cfu/100 ml. Even though disinfection is an effective barrier to many pathogens (especially bacteria) during drinking water treatment, in this study coliform bacteria were still detected in the distribution lines. According to WHO (2011), chemical disinfection of a drinking supply that is faecally contaminated will reduce the overall risk of disease but may not necessarily render the supply safe. For example, chlorine disinfection of drinking water has limitations against the protozoan pathogens, in particular cryptosporidium and some viruses. Disinfection efficacy may also be unsatisfactory against pathogens within flocs or particles, which protect them from the action of disinfection.
Infectious diseases are transmitted through human and animal excreta; faecal contamination of water sources will result in the causative organisms being present in the water. The use of such water for drinking or for preparing food, washing or bathing, may result in infection (Craun et al. 2002).
Water used by elderly or the very young, patients with burns or extensive wounds, those undergoing immunosuppressive therapy, or those with acquired immunodeficiency syndrome (AIDS) for drinking or bathing, if it contains large numbers of these organisms (Pseudomonas aeruginosa, Aeromonas, and certain slow-growing’’ Mycobacteria), can cause various infections of the skin and the mucous membranes of the eye, ear, nose, and throat (WHO 2011). In this study, Aeromonas spp., Clostridium spp. and Pseudomonas spp. were detected in varied concentrations in water from the distribution systems of the two Treatment Works. Aeromonas spp. values ranged from 0 to 13 cfu/100 ml, with the S12 registering the highest value. Counts of Clostridium spp. ranged from 0 to 558 cfu/100 ml, and counts of Pseudomonas spp. ranged from 0 to 744 cfu/100 ml.
One of the factors that tend to increase the load of microorganisms is interrupted water supply which allows pathogens to grow in the distribution lines. Other causes are attributed to unskilled persons tampering with lines to connect water into homes or to repair broken lines. Underground leakages have also been cited as contributory factor. In old settlements, because of poor maintenance culture, most of the lines which need replacement are still in use.
In many unplanned settlements in the metropolis, it was observed that distribution lines were competing with open drains and waste materials for space. Pipeline leak events provide potential for entry of open drains into the treated water; and permits faecal indicators and microbial pathogens present in the soil and water exterior to enter the distribution system (LeChevallier et al. 2003). However, if the levels of chlorine in treated water pumped from head works are maintained in the distribution lines, it would markedly prevent bacteria regrowth and entry.
A wide variety of opportunistic pathogens such as Pseudomonas can be incorporated into the piped potable water, colonizing the surfaces of pipelines with high numbers of biofilm-forming bacteria (Codony et al. 2005). Aeromonas spp. which was not detected at the two water works was found in the distribution systems. Aeromonas have been detected in many treated drinking water supplies mainly as a result of regrowth in distribution systems (WHO 2011). The presence of aeromonas spp. in drinking water supplies is generally considered a nuisance. Suggested control measures to limit growth of the bacteria in distribution system include treatment to optimize organic carbon removal, restriction of the residence time of water in distribution systems and maintenance of disinfectant residuals (WHO 2011). Most of these microorganisms may not cause illness in healthy persons, but they can cause nuisance through generation of tastes, odours or discoloration of drinking water supplies. The high counts of heterotrophic bacteria in some of the sampling areas are reflections of the bacteria “regrowth” that occurred in some parts of the distribution systems. The elevated heterotrophic bacteria counts were found in areas where there was intermittent flow of water supply.
Correlation analyses
Some parameters from Kpong distribution networks did not show any strong significant correlation with any other. However, some of the physico-chemical parameters were found to be significantly correlated with each other. pH significantly related positively with turbidity (r = 0.86) and negatively correlated with conductivity (r = −0.88) (Table 5).
TC and FC were significantly correlated (P < 0.01). Both TC and FC had positive significant correlation with Pseudomonas. This suggests that increase in coliforms causes a corresponding increase in Pseudomonas. Weak inverse correlation was observed between free chlorine and TC and free chlorine with FC. As free chlorine concentration decreases, microbial count increases. This finding is in accordance with a study conducted by Farooq et al. (2008). The presence of coliforms in the water samples collected indicates contamination. One of the sources of coliform occurrence in drinking water has been attributed to biofilms on submerged surfaces of distribution systems (Flemming et al. 2002). Beate et al. (2003) have demonstrated that under certain conditions biofilms can develop on rubber-coated valves in drinking water distribution systems. These biofilms may harbour coliform bacteria where they persist and survive remedial actions such as chlorination. In this study, some levels of coliform bacteria were detected in the water collected. This could be due to inadequate disinfection residual, biofilms or accidental point source contamination by broken pipes, installation and repair works.
There were positive relationships observed between turbidity with TC and FC, indicating that increase in turbidity would result in increase of coliform count. A strong relationship between MPN and turbidity was observed by Farooq et al. (2008). According to WHO (2011), high levels of turbidity can protect microorganisms from the effects of disinfection, stimulate the growth of bacteria and give rise to a significant chlorine demand. Water with high turbidity from organic sources may give rise to substantial chlorine demand. This could result in reductions in the free chlorine residual in the distribution systems as protection against possible recontamination (Farooq et al 2008).
From Table 4, free chlorine showed a negative relationship with the other microorganisms. This implies that as free chlorine decreases other variables such as Aeromonas, Clostridium, Pseudomonas increase in the drinking water.
Similar relationships obtained for parameters in Kpong distribution system were also observed for Weija distribution networks (correlation data not shown). There was a significantly positive correlation between pH and turbidity (r = 0.79) and negative correlation with conductivity (r = −0.87). TC and FC were positively correlated. Both TC and TC were observed to have a high positive relationship with Pseudomonas, Clostridium, Aeromonas and THB. This suggests that increase in coliforms would cause a corresponding increase of these microorganisms.
Conclusions
The study has provided information about the water quality status in the distribution network of Accra-Tema Metropolis. Parameters such as pH, turbidity, conductivity, Fe and Mn were found to be within their WHO guideline limits. Weija source of water recorded higher conductivity values than Kpong source, suggesting more dissolved substances. Residual chlorine was detected at all sites. However, there was microbial pollution indicating bacterial regrowth.
From the residual chlorine data, it was observed that rechlorination does not regularly take place in the booster stations. The water leaving the treatment plants meet bacteriological standards; however, coliform bacteria were detected in the distribution lines suggesting that the water is contaminated in the distribution networks. This could be attributed to adhering of bacteria onto biofilms or accidental point source contamination by broken pipes, installation and repair works. Boiling water in the household before drinking may be necessary to avoid water-related health risks.
High total heterotrophic bacteria counts were detected at most sites which can pose health risk. Interrupted water supply and unskilled persons tampering with distribution lines to connect water into homes or to repair broken lines contributed to increased load of microorganisms allowing pathogens to grow in the distribution lines.
Recommendations
To prevent the delivery of potentially unsafe water, the following are recommended:
-
As a matter of urgency, rechlorination should be done frequently at the booster stations by GWCL to check any bacterial regrowth.
-
Regular monitoring of residual chlorine in the distribution system should be carried out by GWCL to ensure that residual chlorine of 0.2–0.5 mg/l is available at the consumer end.
-
Frequent assessment of water quality should be carried out at different points in the distribution system by an independent body.
-
Connections to GWCL lines should not be tampered by unskilled persons. Accredited professionals only should be allowed to connect water into homes or to repair broken lines to reduce introduction of contaminants in the distribution lines.
-
The public must be encouraged to inform the appropriate GWCL office of any leakages in the pipelines in their locality. GWCL must provide toll free lines to facilitate easy communication.
-
The Government and the management of GWCL must ensure that there is a constant flow of water in the pipeline to reduce bacterial contaminants into the distribution system.
References
Abraham EM, Rooijen DV, Spalthoff D (2007) Planning urban water - dependent livelihood opportunities for the poor in Accra. Ghana, Switch First Scientific Meeting
APHA, AWWA, WEF (1998) Standard methods for the examination of water and wastewater, 20th edn. American Public Health Association/American/Water Works Association/Water Environment Federation, Washington DC
Beate K, Bernd L, Gabriela S, Hans-Curt F, Jost W (2003) Contamination of drinking water by coliforms from biofilms grown on rubber-coated valves. Int J Hg Environ Health 206:563–573
Codony F, Morato J, Mas J (2005) Role of discontinuous chlorination on microbial production by drinking water biofilms. Water Res 39:1896–1906
Craun GF, Nwachuku N, Calderon RL, Craun MF (2002) Outbreaks in drinking-water systems, 1991–1998. J Environ Health 65:16–25
Farooq S, Hashmi I, Qazi IA, Qaiser S, Rasheed S (2008) Monitoring of coliforms and chlorine residual in water distribution network of Rawalpindi, Pakistan. Environ Monit Assess 140:339–347
Flemming HC, Percival S, Walker JT (2002) Contamination potential of biofilms in drinking water distribution systems. Wat Sci Technol Wat Supply 2:271–280
Gatel D, Servais P, Block JC, Bonne P, Cavard J (2000) Microbiological water quality management in the Paris suburbs distribution system. J Water Supply Res Technol AQUA, pp 231–241
Karikari AY, Ampofo JA (2006) Independent assessment of drinking water quality in Accra-Tema Metropolis. Consultancy report, WRI, Accra. WRI/CAR No.215 pp 1–15
Kitazawa H (2006) Keeping residual chlorine and decreasing unpleasant odour caused by disinfection tap water. Water supply 6:193–199
LeChevallier MW, Gullick RW, Karim MR, Friedman M, Funk JE (2003) The potential for health risks from intrusion of contaminant into the distribution system from pressure transients. J. Water Health 1:1–14
Lee EL, Schwab KJ (2005) Deficiencies in drinking water distribution systems in developing countries. J. Water Health 3:109–127
Lienyao L, Chungsying L, Shyang-Lai K (2004) Spacial diversity of chlorine residual in a drinking water distribution system. J Environ Eng 30:1263–1268
Munavalli GR, Mohan Kumar MS (2003) Water quality parameter estimation in steady state distribution system. J Water Resour Plan Manag 129:124–134
Ndambuki JM (2006) Water quality variation within a distribution system: a case study of Eldoret municipality, Kenya. In: Proceeding (515) environmentally sound technology in water resources management
Rooijen DV, Spalthoff D, Raschid-Sally L (2008) Domestic water supply in Accra: how physical and social constraints to planning have greater consequences for the poor. 33rd WEDC International Conference, Accra, Ghana pp 262–267
WHO (2004) Guidelines for drinking-water quality, vol.1, 3rd edn. Recommendations. World Health Organization, Geneva, Switzerland
WHO (2011) Guidelines for drinking- water quality, 4th edn. World Health Organisation, Geneva, pp 1–541
Acknowledgments
We gratefully acknowledge the financial support of the Government of Ghana to carry out this work. The authors appreciate the support of staff from CSIR Water Research Institute, especially Mohammed Bello, Sampson Abu and Michael Afram. Facilities provided by CSIR Water Research institute are hereby acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under license to BioMed Central Ltd.Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Karikari, A.Y., Ampofo, J.A. Chlorine treatment effectiveness and physico-chemical and bacteriological characteristics of treated water supplies in distribution networks of Accra-Tema Metropolis, Ghana. Appl Water Sci 3, 535–543 (2013). https://doi.org/10.1007/s13201-013-0101-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13201-013-0101-6