Abstract
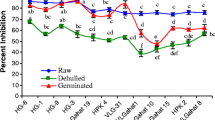
Kodo (Paspalum scrobiculatum) and little (Panicum sumatrense) millet grains were utilized to minimize their antinutrient content (phytate and tannin) and maximize their antioxidant activity (DPPH) by studying the effect of ultrasonication time, germination time and temperature using central composite rotatable design. Results revealed the optimum conditions for producing ultrasonicated and germinated kodo and little millet flour of the highest antioxidant activity and lowest antinutrient content (phytate and tannin) by using 30 min for ultrasonication, 72 h for germination at 40 °C. Further, a second order model was developed to describe and predict the effect of process variables on antioxidant activity and antinutrient contents. Extended experiments were carried out under the optimized conditions to validate the developed model. The antioxidant activity obtained was 88.46% RSA and 89.06% RSA for kodo and little millet grain flours, respectively whereas antinutrient content for phytate was 0.165 mol/kg and 0.199 mol/kg and for tannin 2.88 mol/kg and 9.51 mol/kg, for kodo and little millet grain flours, respectively. This study provides useful information about the potential utilization of ultrasonicated and germinated kodo and little millet grain flours for the development of functional foods.


Similar content being viewed by others
Data availability
Not applicable.
Code availability
Not applicable.
References
Azeke MA, Egielewa SJ, Eigbogbo MU, Ihimire IG (2011) Effect of germination on the phytase activity, phytate and total phosphorus contents of rice (Oryza sativa), maize (Zea mays), millet (Panicum miliaceum), sorghum (Sorghum bicolor) and wheat (Triticum aestivum). J Food Sci Technol 48(6):724–729. https://doi.org/10.1007/s13197-010-0186-y
Badau MH, Nkama I, Jideani IA (2005) Phytic acid content and hydrochloric acid extractability of minerals in pearl millet as affected by germination time and cultivar. Food Chem 92(3):425–435. https://doi.org/10.1016/j.foodchem.2004.08.006
Chang MJ, Collins JL, Bailey JW, Coffey DL (1994) Cowpeas tannins related to cultivar, maturity, dehulling and heating. J Food Sci 59(5):1034–1036. https://doi.org/10.1111/j.1365-2621.1994.tb08183.x
Chauhan ES (2018) Effects of processing (germination and popping) on the nutritional and anti-nutritional properties of finger millet (Eleusine coracana). Curr Res Nutr Food Sci 6(2):566–572. https://doi.org/10.12944/CRNFSJ.6.2.30
Dave S, Yadav BK, Tarafdar JC (2008) Phytate phosphorus and mineral changes during soaking, boiling and germination of legumes and pearl millet. J Food Sci Technol 45(4):344
Dubey A, Tripathy PP (2023) Ultrasound-mediated hydration of finger millet: effects on antinutrients, techno-functional and bioactive properties, with evaluation of Ann-PSO and Rsm optimization methods. Techno-functional and bioactive properties, with evaluation of Ann-PSO and rsm optimization methods.
Fu Y, Zhang F, Liu Z, Zhao Q, Xue Y, Shen Q (2021) Improvement of diabetes-induced metabolic syndrome by millet prolamin is associated with changes in serum metabolomics. Food Biosci 44:101434. https://doi.org/10.1016/j.fbio.2021.101434
Fu Y, Chen B, Liu Z, Wang H, Zhang F, Zhao Q, Zhu Y, Xue Y, Shen Q (2023) Effects of different foxtail millet addition amounts on the cognitive ability of mice. Food Biosci 51:102286. https://doi.org/10.1016/j.fbio.2022.102286
Grgić T, Pavišić Z, Maltar-Strmečki N, Voučko B, ČukeljMustač N, Ćurić D, Le-Bail A, Novotni D (2023) Ultrasound-assisted modification of enzymatic and antioxidant activities, functional and rheological properties of oat and barley bran. Food Bioproc Tech 28:1–14
Hassan S, Imran M, Ahmad MH, Khan MI, Xu C, Khan MK, Muhammad N (2020) Phytochemical characterization of ultrasound-processed sorghum sprouts for the use in functional foods. Int J Food Properties 23(1):853–863
Iftikhar M, Zhang H, Iftikhar A, Raza A, Begum N, Tahamina A, Syed H, Khan M, Wang J (2020) Study on optimization of ultrasonic assisted extraction of phenolic compounds from rye bran. LWT-Food Sci Technol 134:110243. https://doi.org/10.1016/j.lwt.2020.110243
Khandelwal S, Udipi SA, Ghugre P (2010) Polyphenols and tannins in Indian pulses: Effect of soaking, germination and pressure cooking. Food Res Int 43(2):526–530. https://doi.org/10.1016/j.foodres.2009.09.036
Krogdahl Å, Bakke AM (2015) Antinutrients. Dietary Nutr Addit Fish Health 29:211–235
Kumar Y, Singhal S, Tarafdar A, Pharande A, Ganesan M, Badgujar PC (2020) Ultrasound assisted extraction of selected edible macroalgae: effect on antioxidant activity and quantitative assessment of polyphenols by liquid chromatography with tandem mass spectrometry (LC-MS/MS). Algal Res 52:102114. https://doi.org/10.1016/j.algal.2020.102114
Liang S, Liang K (2019) Millet grain as a candidate antioxidant food resource: a review. Int J Food Prop 22(1):1652–1661
Mbithi-Mwikya S, Van Camp J, Yiru Y, Huyghebaert A (2000) Nutrient and antinutrient changes in finger millet (Eleusine coracan) during sprouting. LWT-Food Sci Technol 33(1):9–14. https://doi.org/10.1006/fstl.1999.0605
Pushparaj FS, Urooj A (2014) Antioxidant activity in two pearl millet (Pennisetum typhoideum) cultivars as influenced by processing. Antioxidants 3(1):55–66. https://doi.org/10.3390/antiox3010055
Savelkoul FHMG, Van der Poel AFB, Tamminga S (1992) The presence and inactivation of trypsin inhibitors, tannins, lectins and amylase inhibitors in legume seeds during germination. A Review Plant Foods Hum Nutr 42(1):71–85. https://doi.org/10.1007/BF02196074
Sharma B, Gujral HS (2020) Modifying the dough mixing behavior, protein & starch digestibility and antinutritional profile of minor millets by sprouting. Int J Biol Macromol 153:962–970. https://doi.org/10.1016/j.ijbiomac.2019.10.225
Sharma S, Saxena DC, Riar CS (2016) Analysing the effect of germination on phenolics, dietary fibres, minerals and γ-amino butyric acid contents of barnyard millet (Echinochloa frumentaceae). Food Biosci 13:60–68. https://doi.org/10.1016/j.fbio.2015.12.007
Sharma S, Saxena DC, Riar CS (2017) Using combined optimization, GC–MS and analytical technique to analyze the germination effect on phenolics, dietary fibers, minerals and GABA contents of Kodo millet (Paspalum scrobiculatum). Food Chem 233:20–28. https://doi.org/10.1016/j.foodchem.2017.04.099
Sharma S, Jan R, Riar CS (2021) Analyzing the effect of germination on the pasting, rheological, morphological and in-vitro antioxidant characteristics of kodo millet flour and extracts. Food Chem 361:130073. https://doi.org/10.1016/j.foodchem.2021.130073
Shimelis EA, Rakshit SK (2007) Effect of processing on antinutrients and in vitro protein digestibility of kidney bean (Phaseolus vulgaris L.) varieties grown in East Africa. Food Chem 103(1):161–172. https://doi.org/10.1016/j.foodchem.2006.08.005
Silva DAROD, Jorge LMDM, Jorge RMM (2019) Estudo e modelagem da cinética de hidratação de grãos de sorgo. Rev Cienc Agron 50(1):44–53
Suma P, Urooj A (2014) Influence of germination on bioaccessible iron and calcium in pearl millet (Pennisetum typhoideum). J Food Sci Technol 51(5):976–981. https://doi.org/10.1007/s13197-011-0585-8
Yadav S, Mishra S, Pradhan RC (2021) Ultrasound-assisted hydration of finger millet (Eleusine Coracana) and its effects on starch isolates and antinutrients. Ultrason Sonochem 73:105542. https://doi.org/10.1016/j.ultsonch.2021.105542
Yousaf L, Hou D, Liaqat H, Shen Q (2021) Millet: a review of its nutritional and functional changes during processing. Food Res Int 142:110197. https://doi.org/10.1016/j.foodres.2021.110197
Zheng B, Yuan Y, Xiang J, Jin W, Johnson JB, Li Z, Luo D (2022) Green extraction of phenolic compounds from foxtail millet bran by ultrasonic-assisted deep eutectic solvent extraction: optimization, comparison and bioactivities. Lwt-Food Sci Technol 154:112740
Acknowledgements
First author is grateful to the Vice Chancellor, Late Prof. Manjeet Aggarwal and Dr. Komal Chauhan, National Institute of Food Technology Entrepreneurship and Management (NIFTEM-K) for providing necessary facilities to carry out the research work.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
SD conceived, carried out the experiments and wrote the manuscript; AS supervised the work, revised, and edited the manuscript; YK revised and edited the manuscript; TM revised the experiments; AT revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The author confirm that they have no conflicts of interest with respect to the work described in this manuscript.
Ethical approval
Not applicable.
Consent to participate
All the authors declare there in no conflict of interest in publishing this manuscript.
Consent for publication
All the authors provide the consent to participate.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dey, S., Saxena, A., Kumar, Y. et al. Optimizing the effect of ultrasonication and germination on antinutrients and antioxidants of kodo (Paspalum scrobiculatum) and little (Panicum sumatrense) millets. J Food Sci Technol 60, 2990–3001 (2023). https://doi.org/10.1007/s13197-023-05837-6
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-023-05837-6