Abstract
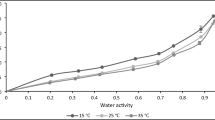
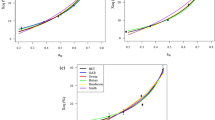
An attempt was made to develop mathematical models to describe the sorption behaviour and determine the thermodynamic properties of sorption of dry-crystallized Palada payasam mix at different water activities (between 0.113 and 0.973) and temperature ranging from 25 to 35 °C. Samples were prepared using both manual and mechanical mixing during the crystallization process and the equilibrium moisture content (EMC) was recorded using the isopiestic technique to compare the sorption behaviour of both samples. The sorption isotherms were found to follow a J-shape—type III plot, with the sorption capacity decreasing with temperature. The isotherms also exhibited a typical temperature inversion of EMC at water activity > 0.70. The sorption data obtained was fitted to 12 mathematical models for sorption and the Guggenheim, Anderson and de Boer (GAB), Peleg and Iglesias and Chirife models were found to describe the data over the entire range of water activity with a good fit. The isosteric heat of sorption and spreading pressure were determined as a function of moisture content. Isosteric heat of the samples was computed and found to decrease from 51.75 to 47.16 kJ/mol for control (manually stirred) and 49.38 to 47.58 kJ/mol for experimental sample (mechanically stirred) for a range of moisture content up to 29% (d.b). The spreading pressures increased with increasing water activity but decreased with increasing temperature. No significant difference was observed between the sorption properties of the dry crystallized samples prepared using manually and mechanically stirring.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study has been incorporated into the research paper.
Code availability
Not applicable (No user defined codes were used for the analysis of data. All the data were analyzed using licensed software’s such as MS Excel, Origin Pro etc.)
Abbreviations
- aw :
-
Water activity
- BET:
-
Brunauer–Emmett–Teller
- EMC:
-
Equilibrium moisture content
- GAB:
-
Guggenheim, Anderson and de Boer
- ICAR-NDRI:
-
Indian Council of Agricultural Research-National Dairy Research Institute
- K2SO4 :
-
Potassium sulphate
- KCl:
-
Potassium chloride
- KI:
-
Potassium iodide
- kJ:
-
Kilo joule
- LiCl:
-
Lithium chloride
- Mg (NO3)2 :
-
Magnesium nitrate
- MgCl2 :
-
Magnesium chloride
- MRCMPU:
-
Malabar Regional Co-operative Milk Producers’ Union Ltd
- NaCl:
-
Sodium chloride
- RH:
-
Relative humidity
- SEM:
-
Scanning electron microscopy
- SNF:
-
Solids not fat
- SRS:
-
Southern regional station
- BDDT:
-
Brunauer–Deming–Deming–Teller
References
Al-Muhtaseb AH, McMinn WAM, Magee TRA (2002) Moisture sorption isotherm characteristics of food products: a review. Food Bioprod Process 80(2):118–128. https://doi.org/10.1205/09603080252938753
Al-Muhtaseb AH, McMinn WA, Magee TRA (2004) Water sorption isotherms of starch powders: part 1: mathematical description of experimental data. J Food Eng 61(3):297–307. https://doi.org/10.1016/S0260-8774(03)00133-X
Arslan N, Togrul H (2005) Modelling of water sorption isotherms of macaroni stored in a chamber under controlled humidity and thermodynamic approach. J Food Eng 69(2):133–145. https://doi.org/10.1016/j.jfoodeng.2004.08.004
Basu S, Shivhar US, Mujumdar AS (2006) Models for sorption isotherms for foods: a review. Dry Technol 24(8):917–930. https://doi.org/10.1080/07373930600775979
Blahovec J, Yanniotis S (2008) GAB generalized equation for sorption phenomena. Food Bioprocess Technol 1(1):82–90. https://doi.org/10.1007/s11947-007-0012-3
Changade SP, Waseem M, Wasnik PG, Narnaware GN, Chapke JS (2012) Storage studies of bottle gourd and pumpkin kheer. J Dairy Foods Home Sci 31(2):23–32
Fasina OO (2006) Thermodynamic properties of sweet potato. J Food Eng 75(2):149–155. https://doi.org/10.1016/j.jfoodeng.2005.04.004
Fazaeli M, Tahmasebi M, Emam Djomeh Z (2012) Characterization of food texture: application of microscopic technology. Curr Microsc Contrib Adv Sci Technol 2:855–871
Hadjikinova M, Menkov N, Hadjikinov D (2003) Sorption characteristics of dietary hard candy. Czech J Food Sci 21(3):97–100. https://doi.org/10.17221/3483-CJFS
Kumar AJ, Singh RRB, Patil GR, Patel AA (2005) Effect of temperature on moisture desorption isotherms of kheer. LWT Food Sci Technol 38(3):303–310. https://doi.org/10.1016/j.lwt.2003.10.009
Liebanes MD, Aragon JM, Palancar MC, Arevalo G, Jimenez D (2006) Equilibrium moisture isotherms of two-phase solid olive oil by-products: adsorption process thermodynamics. Colloids Surf A Physicochem Eng Asp 282:298–306. https://doi.org/10.1016/j.colsurfa.2006.03.025
McHugh ML (2008) Standard error: meaning and interpretation. Biochem Med 18(1):7–13. https://doi.org/10.11613/BM.2008.002
McMinn WAM, Magee TRA (2003) Thermodynamic properties of moisture sorption of potato. J Food Eng 60(2):157–165. https://doi.org/10.1016/S0260-8774(03)00036-0
Palou E, Lopez-Malo A, Argaiz A (1997) Effect of temperature on the moisture sorption isotherms of some cookies and corn snacks. J Food Eng 31(1):85–93. https://doi.org/10.1016/S0260-8774(96)00019-2
Pushpadass HA, Emerald FME, Chaturvedi B, Rao KJ (2014) Moisture sorption behaviour and thermodynamic properties of gulabjamun mix. J Food Process Pres 38(6):2192–2200. https://doi.org/10.1111/jfpp.12199
Rahman MS, Sablani SS (2009) Water activity measurement methods of foods. In: Rahman MS (ed) Food properties handbook, 2nd edn. CRC Press, New York, pp 9–32
Rao KJ, Dhas PHA, Emerald FME, Ghosh BC, Balasubramanyam BV, Kulkarni S (2006) Moisture sorption characteristics of chhana podo at 5 °C and 35 °C. J Food Eng 76(3):453–459. https://doi.org/10.1016/j.jfoodeng.2005.04.048
Rizvi SS (2014) Thermodynamic properties of foods in dehydration. In: Rao MA, Rizvi SS, Datta AK, Ahmed J (eds) Engineering properties of foods, 3rd edn. CRC Press, New York, pp 261–348
Ruckold S, Grobecker KH, Isengard HD (2000) Determination of the contents of water and moisture in milk powder. Fresenius J Anal Chem 368(5):522–527. https://doi.org/10.1007/s002160000511
Sahu JK, Das H (2010) Moisture sorption isotherm, properties of sorbed water and heat of sorption of Sandesh: an indian milk product. J Food Process Pres 34(1):152–166. https://doi.org/10.1111/j.1745-4549.2009.00461.x
Samapundo S, Devlieghere F, de Meulenaer B, Atukwase A, Lamboni Y, Debevere JM (2007) Sorption isotherms and isosteric heats of sorption of whole yellow dent corn. J Food Eng 79(1):168–175. https://doi.org/10.1016/j.jfoodeng.2006.01.040
Saravacos GD, Tsiourvas DA, Tsami E (1986) Effect of temperature on the water adsorption isotherms of sultana raisins. J Food Sci 51(2):381–383. https://doi.org/10.1111/j.1365-2621.1986.tb11135.x
Sharma P, Singh RRB, Singh AK, Patel AA, Patil GR (2009) Sorption isotherms and thermodynamics of water sorption of ready-to-use Basundi mix. LWT Food Sci Technol 42(1):441–445. https://doi.org/10.1016/j.lwt.2008.04.010
Thanuja D, Ravindra MR (2014) Thermodynamic analysis of moisture sorption characteristics of cheese-puri mix. J Food Process Pres 38(1):420–429. https://doi.org/10.1111/j.1745-4549.2012.00790.x
Unnikrishnan V, Bhavadasan MK, Nath BS, Vedavathi MK, Balasubramanya NN (2000) Payasam: a sweet delicacy. Indian Dairym 52(10):37–43
Unnikrishnan V, Bhavadasan MK, Vedavati MK, Nath BS (2003) A dry mix for convenient preparation of palada payasam. Indian Dairym 55(7):70–74
Wolf W, Spiess WL, Jung G (1985) Standardization of isotherm measurements (COST project 90 and 90 BIS). In: Simatos D, Multon JL (eds) Properties of water in foods. Martinus Nihoff Publisher, Dordrecht, pp 661–677
Acknowledgement
The manuscript is the original research work done in the Department of Dairy Engineering, SRS of ICAR-National Dairy Research Institute, Bengaluru (India). The first author acknowledges the Institute fellowship and the research facilities received from ICAR-National Dairy Research Institute, Karnal, Haryana (India) for conducting the study. The authors are also grateful to Centre for Nano Science and Engineering of IISC, Bengaluru for providing facilities for characterization of samples. In addition, we gratefully acknowledge and are indebted to the anonymous referees for comments and constructive suggestions provided for improving the manuscript.
Funding
The authors are grateful for the Institute fellowship received from ICAR-National Dairy Research Institute, Karnal, Haryana (India) for conducting the study.
Author information
Authors and Affiliations
Contributions
NJ conceived, carried out the experiments and wrote the manuscript; GPD contributed to analysis and interpretation of the results, MRR supervised the work and edited the manuscript, KJR helped in analysis of results. All authors provided critical feedback and helped shape the research, analysis and manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
With the submission, we hereby confirm that no conflict of interest in submission of paper to journal.
Ethical approval
As corresponding author, I confirm that the manuscript has been read and approved for submission to JFST by all the named authors. Also I confirm that this work is original and has not been published nor under consideration elsewhere. This article does not contain any studies with human or animal subjects.
Consent to participate
I undertake that I may review at least three manuscripts submitted to JFST.
Consent for publication
Not applicable (No third party images or data were used in this paper. All the data or images used in the MS were from the study conducted at the institute. There is no copyright infringement in figures or in any part of manuscript).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jose, N., Ravindra, M.R., Deshmukh, G.P. et al. Sorption and thermodynamic properties of dry crystallized Palada payasam mix prepared using manual and mechanical stirring. J Food Sci Technol 59, 1075–1086 (2022). https://doi.org/10.1007/s13197-021-05111-7
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-021-05111-7