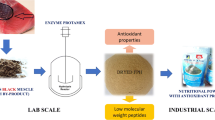
Abstract
High fish production is essential to meet the demand, but inappropriate destination of large volumes of byproduct cause environmental pollution. The cutting step for frozen eviscerated fish using band saw machines produces a type of “fish powder” byproduct with high protein content. The objective of this study was to optimize the process of obtaining protein hydrolysates from the cutting byproducts of Serra Spanish Mackerel (SSM) and to evaluate the final product regarding its technological properties. The optimal conditions for obtaining the protein hydrolysate from the cutting byproducts of SSM using a band saw machine were an enzyme:substrate ratio (w/w) of 5.0% and 240 min of enzymatic hydrolysis. Both treatments (+ DH and − DH) yielded volatile compounds with a characteristic fish aroma, and both can be used for flavoring. The − DH hydrolysate showed better technological performance by stabilizing emulsions and retaining oil, and they could be added to emulsified products, improving their technological and sensory aspects. For the antioxidant capacity, the + DH hydrolysate showed higher efficiency, and it was indicated for use in food products, with the aim of extending the shelf life by stabilizing food lipids and proteins, ensuring the quality of the product during storage.


Similar content being viewed by others
References
Adler-Nissen J (1986) A review of food hydrolysis specific areas. In: Adler-Nissen J (ed) Enzymic hydrolysis of food proteins. Elsevier, Copenhagen, pp 57–109
Association of Official Analysis Chemists International, AOAC (2010) Official methods of analysis of AOAC international, 18th edn. Gaithersburg, MD, USA
Benzie IFF, Strain JJ (1999) Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Method Enzym 299:15–27. https://doi.org/10.1016/S0076-6879(99)99005-5
Brand-Williams W, Cuvelier ME, Berset C (1995) Use of a free radical method to evaluate antioxidant activity. Trends Food Sci Technol 28:25–30. https://doi.org/10.1016/S0023-6438(95)80008-5
Breternitz NR, Fidelis CRV, Silva VM, Eberlin MN, Hubinger MD (2017) Volatile composition and physicochemical characteristics of mussel (Perna perna) protein hydrolysate microencapsulated with maltodextrin and n-OSA modified starch. Food Bioprod Process 105:12–25. https://doi.org/10.1016/j.fbp.2017.05.008
Elavarasan K, Kumar NV, Shamasundar BA (2014) Antioxidant and functional properties of (FPH) from fresh water carp (Catla catla) as influenced by the nature of enzyme. J Food Process Preserv 38:1207–1214. https://doi.org/10.1111/jfpp.12081
Feng L, Peng F, Wang X, Li M, Lei H, Xu H (2018) Identification and characterization of antioxidative peptides derived from simulated in vitro gastrointestinal digestion of walnut meal proteins. Food Res Int 116:518–526. https://doi.org/10.1016/j.foodres.2018.08.068
Folch J, Lees M, Stanley S (1957) A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 226:497–509
Fonkwe LG, Singh RK (1996) Protein Recovery from mechanically deboned Turkey residue by enzymic hydrolysis. Process Biochem 31:605–616. https://doi.org/10.1016/S0032-9592(95)00101-8
Halim NRA, Yusof HM, Sarbon NM (2016) Functional and bioactive properties of fish protein hydolysates and peptides: a comprehensive review. Trends Food Sci Technol 51:24–33. https://doi.org/10.1016/j.tifs.2016.02.007
Halldorsdottir SM, Sveinsdottir H, Gudmundsdottir A, Thorkelsson G, Kristinsson HG (2014) High quality fish protein hydrolysates prepared from by-product material with Fucus vesiculosus extract. J Funct Foods 9:10–17. https://doi.org/10.1016/j.jff.2014.04.009
Brazilian Institute of the Environment and Renewable Natural Resources (2007) Fisheries statistics 2005. Brasília, Brazil
Kittiphattanabawon P, Benjakul S, Visessanguan W, Shahidi S (2012) Gelatin hydrolysate from blacktip shark skin prepared using papaya latex enzyme: antioxidant activity and its potential in model systems. Food Chem 135:1118–1126. https://doi.org/10.1016/j.foodchem.2012.05.080
Laohakunjit N, Selamassakul O, Kerdchoechuen O (2014) Seafood-like flavour obtained from the enzymatic hydrolysis of the protein by-products of seaweed (Gracilaria sp.). Food Chem 158:162–170. https://doi.org/10.1016/j.foodchem.2014.02.101
Lassoued I, Mora L, Barkia A, Aristoy M, Nasri M, Toldrá F (2015) Bioactive peptides identified in thornback ray skin's gelatin hydrolysates by proteases from Bacillus subtilis and Bacillus amyloliquefaciens. J Proteomics 128:8–17. https://doi.org/10.1016/j.jprot.2015.06.016
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Martínez-Alvarez O, Chamorro S, Brenes A (2015) Protein hydrolysates from animal processing by-products as a source of bioactive molecules with interest in animal feeding: a review. Food Res Int 73:204–212. https://doi.org/10.1016/j.foodres.2015.04.005
Morales-Medina R, Tamm F, Guadix AM, Guadix EM, Drusch S (2016) Functional and antioxidant properties of hydrolysates of sardine (S. pilchardus) and horse mackerel (T. mediterraneus) for the microencapsulation of fish oil by spray-drying. Food Chem 194:1208–1216. https://doi.org/10.1016/j.foodchem.2015.08.122
Noman A, Xu A, AL-Bukhaiti WQ, Abed SM, Ali AH, Ramadhana AH, Xia W (2018) Influence of enzymatic hydrolysis conditions on the degree of hydrolysis and functional properties of protein hydrolysate obtained from Chinese sturgeon (Acipenser sinensis) by using papain enzyme. Process Biochem 67:19–28. https://doi.org/10.1016/j.procbio.2018.01.009
Peinado I, Koutsidis G, Ames J (2016) Production of seafood flavor formulations from enzymatic hydrolysates of fish by-products. Food Sci Technol 66:444–452. https://doi.org/10.1016/j.lwt.2015.09.025
Petenuci ME, Rocha INA, Sousa SC, Schneider VVA, Costa LAMA, Visentainer JV (2016) Seasonal variations in lipid content, fatty acid composition and nutritional profiles of five freshwater fish from the Amazon Basin. J Am Oil Chem' Soc 93:1373–1381. https://doi.org/10.1007/s11746-016-2884-8
Putra SNKM, Ishak NH, Sarbon NM (2018) Preparation and characterization of physicochemical properties of golden apple snail (Pomacea canaliculata) protein hydrolysate as affected by different proteases. Biocatal Agric Biotechnol 13:123–128. https://doi.org/10.1016/j.bcab.2017.12.002
Quadros DA, Bolini HMA (2015) Biometric characterization, proximate composition, and fillet yield and waste of Serra Spanish mackarel (Scomberomorus brasiliensis). Bol Inst de Pesca 41:877–888
Queiroz ALM, Araújo ARR, Pacheco MTB, Madruga MS (2017) Potential use of goat by-products to obtain protein hydrolysates. Food Biosci 20:19–27. https://doi.org/10.1016/j.fbio.2017.07.009
Razali AN, Sarbon NM, Amin AM (2015) Antioxidant activity and functional properties of fractionated cobia skin gelatin hydrolysate at different molecular weight. Int Food Res J 22:651–660
Re R, Pellegrini N, Proteggente A, Pannala A, Yang M, Rice-Evans C (1999) Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radical Biol Med 26:1231–1237. https://doi.org/10.1016/S0891-5849(98)00315-3
Robert M, Zatylny-Gaudina C, Fournierd V, Corree E, Corguillée GL, Bernayc B, Henry J (2015) Molecular characterization of peptide fractions of a Tilapia (Oreochromis niloticus) by-product hydrolysate and in vitro evaluation of antibacterial activity. Process Biochem 50:487–492. https://doi.org/10.1016/j.procbio.2014.12.022
Sai-Ut S, Benjakul S, Sumpavapol P, Kishimura H (2014) Effect of drying methods on odorous compounds and antioxidative activity of gelatin hydrolysate produced by protease from B. amyloliquefaciens H11. Dry Technol 32:1552–1559. https://doi.org/10.1080/07373937.2014.904338
Sarteshnizi RA, Sahari MA, Gavlighi HA, Regenstein JM, Nikoo M (2019) Antioxidant activity of Sind sardine hydrolysates with pistachio green hull (PGH) extracts. Food Biosc 27:37–45. https://doi.org/10.1016/j.fbio.2018.11.007
Sila A, Bougatef A (2016) Antioxidant peptides from marine by-products: isolation, identification and application in food systems. A review. J Funct Foods 21:10–26. https://doi.org/10.1016/j.jff.2015.11.007
Silva JFX, Ribeiro K, Silva JF, Cahú TB, Bezerra RS (2014) Utilization of tilapia processing waste for the production offish protein hydrolysate. Ani Feed Sci Technol 196:96–106. https://doi.org/10.1016/j.anifeedsci.2014.06.010
Silva TE (2018) The communities 'fishing ethnochinism radicionals from the Ponta do Tubarão state sustainable development reserve (Brazil)—Brazil. State University of Rio Grande do Norte
Statsoft, Inc (2004) Statistica (data analysis software system), version 7.0. INC, OK, EUA.
Stookey LL (1970) Ferrozine-a new spectrophotometric reagent for iron. Anal Chem 42:779–781
Sukkhown P, Jangchud K, Lorjaroenphon Y, Pirak T (2018) Flavored-functional protein hydrolysates from enzymatic hydrolysis of dried squid by-products: Effect of drying method. Food Hydrocoll 76:103–112. https://doi.org/10.1016/j.foodhyd.2017.01.026
Taheri A, Anvar SA, Ahari H, Fogliano V (2013) Comparison the functional properties of protein hydrolysates from poultry by-products and rainbow trout (Onchorhynchus mykiss) viscera. Iran J Fish Sci 12:154–169
Tocher DR (2015) Omega-3 long-chain polyunsaturated fatty acids and aquaculture in perspective. Aquaculture 449:94–107. https://doi.org/10.1016/j.aquaculture.2015.01.010
Todd RH, Allen DK, Alting L (1994) Manufacturing processes reference guide, Industrial Press Inc., ISBN 0–8311–3049–0. 486p.
Vannabun A, Ketnawa S, Phongthai S, Benjakul S, Rawdkuen S (2014) Characterization of acid and alkaline proteases from viscera of farmed giant catfish. Food Biosci 6:9–16. https://doi.org/10.1016/j.fbio.2014.01.001
Villamil O, Váquiro H, Solanilla JF (2017) Fish viscera protein hydrolysates: production, potential applications and functional and bioactive properties. Food Chem 224:160–171. https://doi.org/10.1016/j.aaspro.2016.02.167
Witono Y, Taruna I, Windrati WS, Azkiyah L, Norma T (2016) ‘Wader’ (rasbora jacobsoni) protein hydrolysates: production, biochemical, and functional properties. Agric Agric Sci Proced 9:482–492. https://doi.org/10.1016/j.aaspro.2016.02.167
Zhan P, Tian H, Zhang X, Wang L (2013) Contribution to aroma characteristics of mutton process flavor from the enzymatic hydrolysate of sheep bone protein assessed by descriptive sensory analysis and gas chromatography olfactometry. J Chromatogr B 921–922:1–8. https://doi.org/10.1016/j.jchromb.2012.12.026
Zhang Y, Olsen K, Grossi A, Otte J (2013) Effect of pretreatment on enzymatic hydrolysis of bovine collagen and formation of ACE-inhibitory peptides. Food Chem 141:2343–2354. https://doi.org/10.1016/j.foodchem.2013.05.058
Acknowledgements
We thank the fish processing company for ceding the by-product used in the present study and thank the CNPQ for the grant of a research grant during the development of this work.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lima, D.A.S., Santos, M.M.F., Duvale, R.L.F. et al. Technological properties of protein hydrolysate from the cutting byproduct of serra spanish mackerel (Scomberomorus brasiliensis). J Food Sci Technol 58, 2952–2962 (2021). https://doi.org/10.1007/s13197-020-04797-5
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04797-5