Abstract
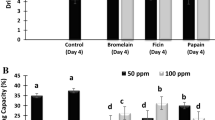
In this study, the semitendinosus of horse meat was used as the raw material. The study assessed the variation of the tenderness of horse meat during postmortem aging through the injection of papain, bromelain and fungal protease. The cooking loss of the horse meat became worse during postmortem aging. Low concentration of protease improved water retention properties of horse meat. Papain, bromelain and fungal protease had significant influence on MFI and shear force. MFI increased obviously but the shear force decreased significantly with the addition of more protease (p <0.05). During postmortem aging, many small molecules popeptide appeared in treatment group. Myosin light chain 2, 20 KDa, 32 KDa and 75 KDa bands appeared at first, however later they disappeared in the group with high concentration of protease treatment in addition to the disappearance of Desmin and Troponin I. The muscle fiber, perimysium and endomysium were degraded because of papain, bromelain and fungal protease treatment. More muscle fiber fragments appeared during postmortem aging.Thus, the tenderness and eating quality of horse meat were improved by adding three kinds of protease.



Similar content being viewed by others
References
Bagley JL (2007) Consumer and shear force evaluation of steaks from the M. Texas A&M University, Serratus ventralis
Culler R, Smith G, Cross H (1978) Relationship of myofibril fragmentation index to certain chemical, physical and sensory characteristics of bovine longissimus muscle. J Food Sci 43:1177–1180
Davey CL, Gilbert K (1969) Studies in meat tenderness. 7. Changes in the fine structure of meat during aging. J Food Sci 34:69–74
Doneva M, Miteva D, Dyankova S (2015) Efficiency of plant proteases bromelain and papain on turkey meat tenderness. Biotechnol in Anim Husbandry 31:407–413
Goll DE, Otsuka Y, Nagainis PA, Shannon JD, Sathe SK, Muguruma M (1983) Role of muscle proteinases in maintenance of muscle integrity and mass. J Food Biochem 7:137–177
Gornall AG, Bardawill CJ, David MM (1949) Determination of serum Proteins by means off the biuret reaction [J]. J Biological Chem 177:751–766
Ho C-Y, Stromer MH, Robson RM (1996) Effect of electrical stimulation on postmortem titin, nebulin, desmin, and troponin-T degradation and ultrastructural changes in bovine longissimus muscle. J Anim Sci 74:1563–1575
Ho C, Stromer M, Robson R (1994) Identification of the 30 kDa polypeptide in post mortem skeletal muscle as a degradation product of troponin-T. Biochimie 76:369–375
Hu Y, Lv D, Yang H, Ge C, Zhu R (2010) Effect of calcium chloride and papain on the mutton tenderness. Mod Food Sci Technol 26:389–391
Huff-Lonergan E, Parrish FC Jr, Robson RM (1995) Effects of postmortem aging time, animal age, and sex on degradation of titin and nebulin in bovine longissimus muscle. J Anim Sci 73:1064–1073
Koohmaraie M (1996) Biochemical factors regulating the toughening and tenderization processes of meat. Meat Sci 43s1:193–201
Koohmaraie M, Seidemann S, Schollmeyer J, Dutson T, Crouse J (1987) Effect of post-mortem storage on Ca++-dependent proteases, their inhibitor and myofibril fragmentation. Meat Sci 19:187–196
Koohmaraie M, Shackelford S (1991) Effect of calcium chloride infusion on the tenderness of lambs fed a β-adrenergic agonist. J Anim Sci 69:2463–2471
Labeit S, Kolmerer B (1995) Titins giant proteins in charge of muscle ultrastructure and elasticity. Science 270:293–296
Lei C, Meng Y, Xi H (2009) Effect of calcium chloride, phosphate, papain influence on MFI and shear force of beef. Meat Industry 2:23–25
Lonergan EH, Zhang W, Lonergan SM (2010) Biochemistry of postmortem muscle—Lessons on mechanisms of meat tenderization. Meat Sci 86:184–195
Ma XX, Shuang Q, Li XL (2012) Effects of papain on tenderness of two-humped camel meat (J). Food Ind 11:29–37
Macbride MA, Parrish F Jr (1977) The 30,000-dalton component of tender bovine longissimus muscle. J Food Sci 42:1627–1629
Mckeith FK, Brewer MS, Bruggen KA (1994) Effects of enzyme applications on sensory, chemical and processing characteristics of beef steaks and roasts. J Muscle Foods 5:149–164
Ming J, Zeng KF, Li H (2009) Study on effects of papain treatment on tenderness of beef (J). J Food Sci 30:210–214
Ngapo T, Babare I, Reynolds J, Mawson R (1999) A preliminary investigation of the effects of frozen storage on samples of pork. Meat Sci 53:169–177
O'Halloran G, Troy D, Buckley D (1997) The relationship between early post-mortem pH and the tenderisation of beef muscles. Meat Sci 45:239–251
Pieszka M, Bederska-Łojewska Dorota, Pieszka M (2016) Effect of dietary supplementation with dried fruit and vegetable pomaces on production parameters and meat quality in fattening pigs. Meat sci 126:1–10
Rajagopal K, Oommen GT, Kuttinarayanan P, George S, SyamMohan K (2015) Effect of post rigor marination with calcium chloride on the tenderness, colour and palatability traits of buffalo meat. Nutr Food Sci 45:20–38
Robson RM, Huiatt TW, Parrish Jr FC (1991). Biochemical and structural properties of titin, nebulin and intermediate filaments in muscle. In: Proceedings Recip Meat Conf. pp. 20
Schenkova N, Šikulová M, Jelenikova J, Pipek P, Houška M, Marek M (2007) Influence of high isostatic pressure and papain treatment on the quality of beef meat. High Press Res 27:163–168
Shackelford SD, Wheeler TL, Meade MK, Reagan JO, Byrnes BL, Koohmaraie M (2001) Consumer impressions of tender select beef. J Anim Sci 79:2605–2614
Sullivan GA, Calkins C (2010) Application of exogenous enzymes to beef muscle of high and low-connective tissue. Meat Sci 85:730–734
Taylor RG, Geesink GH, Thompson VF, Koohmaraie M, Goll DE (1995) Is Z-disk degradation responsible for postmortem tenderization? J Anim Sci 73:1351–1367
Wheeler T, Koohmaraie M (1999) The extent of proteolysis is independent of sarcomere length in lamb longissimus and psoas major. J Anim Sci 77:2444–2451
Whipple G, Koohmaraie M, Dikeman M, Crouse J (1990) Predicting beef-longissimus tenderness from various biochemical and histological muscle traits. J Anim Sci 68:4193–4199
White A, O’Sullivan A, Troy D, O’Neill E (2006) Effects of electrical stimulation, chilling temperature and hot-boning on the tenderness of bovine muscles. Meat Sci 73:196–203
Funding
The research was financially supported by the National Natural Science Foundation of China (No. 31660440).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cheng, Y., Jiang, X., Xue, Y. et al. Effect of three different proteases on horsemeat tenderness during postmortem aging. J Food Sci Technol 58, 2528–2537 (2021). https://doi.org/10.1007/s13197-020-04759-x
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04759-x