Abstract
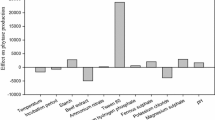
The aim of study was to optimize fermentation parameters for inulinase production from Rhizopus oryzae by a statistical approach and to carry out purification of inulinase. Five isolated fungal strains were screen out inulin degradation by using Lugol’s iodine solution. R. oryzae exhibited maximum zone of clearance around the colony and was used as an inulinase producer. The effect of carbon sources (inulin, glucose, maltose, sucrose, lactose, onion peel, stevia root, wheat bran) as medium component and fermentation parameters (temperature (25–45 °C), initial pH (4–7), time (3–7 days)) on inulinase production was investigated by Plackett–Burman Design. Wheat Bran (WB), temperature, pH, and incubation time were found to be significant for the production of inulinase (P < 0.05). Furthermore, Box–Behnken Design was employed to optimize fermentation conditions. The maximum experimental results for inulinase activity and specific activity were 348.36 EU/mL and 3621.78 EU/mg, respectively. The results were obtained at 5 days of incubation time, 35 °C of incubation temperature, initial pH of 5.5, and 2% (w/v) WB. Also, inulinase was purified by using ammonium sulfate precipitation, gel filtration chromatography with 2.19-fold and its molecular weight was found as 89.12 kDa. The optimal pH and temperature of the purified enzyme were 4.0 and 60 °C, respectively. Furthermore, the purified enzyme showed excellent stability at 60 °C. In conclusion, the present study offers cost-effective method to produce inulinase from Rhizopus oryzae. Also, it can be suggested that the purified inulinase has strong potential for usage in production of fructose syrup and other industrial areas.


Similar content being viewed by others
References
Allais JJ, Kammoun S, Blanc P, Girard C, Baratti JC (1986) Isolation and characterization of bacterial strains with inulinase activity. Appl Environ Microbiol 52:1086–1090
Arnau J, Yaver D, Hjort CM (2020) Strategies and challenges for the development of industrial enzymes using fungal cell factories. In: Nevalainen H (ed) Grand challenges in fungal biotechnology. Springer, Cham, pp 179–210
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72(1–2):248–254
Cordova-Villegas LG, Cordova-Villegas AY, Taylor KE, Biswas N (2019) Response surface methodology for optimization of enzyme-catalyzed azo dye decolorization. J Environ Eng 145(5):04019013. https://doi.org/10.1061/(ASCE)EE.1943-7870.0001513
Danial EN, Ayaz NOA, Alnahdi HSO (2015) Production of inulinase by free and immobilized cells of Penicillium funiculosum p. 36. Braz Arc Biol Technol 58(4):636–642. https://doi.org/10.1590/S1516-8913201500167
Das D, Selvaraj R, Bhat MR (2019a) Optimization of inulinase production by a newly isolated strain Aspergillus flavus var flavus by solid state fermentation of Saccharum arundinaceum. Biocatal Agric Biotechnol 22:101363. https://doi.org/10.1016/j.bcab.2019.101363
Das D, Bhat R, Selvaraj R (2019b) Review of inulinase production using solid-state fermentation. Ann Microbiol 69(3):201–209. https://doi.org/10.1007/s13213-019-1436-5
Dinarvand M, Rezaee M, Foroughi M (2017) Optimizing culture conditions for production of intra and extracellular inulinase and invertase from Aspergillus niger ATCC 20611 by response surface methodology (RSM). Braz J Microbiol 48(3):427–441. https://doi.org/10.1016/j.bjm.2016.10.026
El Aty AAA, Wehaidy HR, Mostafa FA (2014) Optimization of inulinase production from low cost substrates using Plackett–Burman and Taguchi methods. Carbohydr Polym 102:261–268. https://doi.org/10.1016/j.carbpol.2013.11.007
Germec M, Turhan I (2019) Evaluation of carbon sources for the production of inulinase by Aspergillus niger A42 and its characterization. Bioproc Biosyst Eng 42(12):1993–2005. https://doi.org/10.1007/s00449-019-02192-9
Germec M, Gürler HN, Ozcan A, Erkan SB, Karahalil E, Turhan I (2020) Medium optimization and kinetic modeling for the production of Aspergillus niger inulinase. Bioproc Biosyst Eng 43(2):217–232. https://doi.org/10.1007/s00449-019-02219-1
Gill PK, Manhas RK, Singh P (2006) Purification and properties of a heat-stable exoinulinase isoform from Aspergillus fumigatus. Bioresour Technol 97(7):894–902. https://doi.org/10.1016/j.biortech.2005.04.034
Gong F, Zhang T, Chi Z, Sheng J, Li J, Wang X (2008) Purification and characterization of extracellular inulinase from a marine yeast Pichia guilliermondii and inulin hydrolysis by the purified inulinase. Biotechnol Bioproc E 13(5):533–539. https://doi.org/10.1007/s12257-007-0177-7
Ilgın M, Germec M, Turhan I (2019) Inulinase production and mathematical modeling from carob extract by using Aspergillus niger. Biotechnol Prog. https://doi.org/10.1002/btpr.2919
Jain SC, Jain PC, Kango N (2012) Production of inulinase from Kluyveromyces marxianus using Dahlia tuber extract. Braz J Microbiol 43(1):62–69. https://doi.org/10.1590/S1517-83822012000100007
Jeza S, Maseko SB, Lin J (2018) Purification and characterization of exo-inulinase from Paenibacillus sp. d9 strain. Protein J 37(1):70–81. https://doi.org/10.1007/s10930-017-9752-8
Kamble PP, Kore MV, Patil SA, Jadhav JP, Attar YC (2018) Statistical optimization of process parameters for inulinase production from Tithonia weed by Arthrobacter mysorens strain no.1. J Microbiol Methods 149:55–66. https://doi.org/10.1016/j.mimet.2018.04.019
Kamble PP, Suryawanshi SS, Jadhav JP, Attar YC (2019) Enhanced inulinase production by Fusarium solani JALPK from invasive weed using response surface methodology. J Microbiol Methods 159:99–111. https://doi.org/10.1016/j.mimet.2019.02.021
Karam EA, Kansoh AL, Moharam ME, Hassan ME, Kansoh AL (2018) Immobilization of inulinase produced by Rhizopus oligosporus NRRL 2549 for continuous fructose production. J Mater Environ Sci 9(8):2315–2321
Laemmli DK (1970) Clevage of structual proteins during in assembly of the head of bacteriophage T4. Nature 227:680–683
Li AX, Guo LZ, Fu Q, Lu WD (2011) A simple and rapid plate assay for screening of inulin degrading microorganisms using Lugol’s iodine solution. Afr J Biotechnol 10(46):9518–9521. https://doi.org/10.5897/AJB11.1367
Mohamed SA, Salah HA, Moharam ME, Foda MS, Fahmy AS (2015) Characterization of two thermostable inulinases from Rhizopus oligosporus NRRL 2710. Genet Eng Biotechnol 13(1):65–69. https://doi.org/10.1016/j.jgeb.2014.12.001
Naidoo K, Kumar A, Sharma V, Permaul K, Singh S (2015) Purification and characterization of an endoinulinase from Xanthomonas campestris pv. phaseoli KM 24 Mutant. Food Technol Biotechnol 53(2):146–153. https://doi.org/10.17113/ftb.53.02.15.3902
Ohta K, Suetsugu N, Nakamura T (2002) Purification and properties of an extracellular inulinase from Rhizopus sp. strain TN-96. J Biosci Bioeng 94:78–80. https://doi.org/10.1016/S1389-1723(02)80120-7
Onilude AA, Fadaunsi IF, Garuba EO (2012) Inulinase production by Saccharomyces sp. in solid state fermentation using wheat bran as substrate. Annal Microbiol 62(2):843–848. https://doi.org/10.1007/s13213-011-0325-3
Raja S, Murty VR (2013) Optimization of aqueous two-phase systems for the recovery of soluble proteins from tannery wastewater using response surface methodology. J Eng. https://doi.org/10.1155/2013/217483
Saber W, El-Naggar NE (2009) Optimization of fermentation conditions for the biosynthesis of inulinase by the new source; Aspergillus tamarii and hydrolysis of some inulin containing agrowastes. Biotechnology 8:425–433. https://doi.org/10.3923/biotech.2009.425.433
Saqib AA, Hassan M, Khan NF, Baig S (2010) Thermostability of crude endoglucanase from Aspergillus fumigatus grown under solid state fermentation (SSF) and submerged fermentation (SmF). Process Biochem 45(5):641–646. https://doi.org/10.1016/j.procbio.2009.12.011
Sheng J, Chi Z, Gong F, Li J (2008) Purification and characterization of extracellular inulinase from a marine yeast Cryptococcus aureus G7a and inulin hydrolysis by the purified inulinase. Appl Biochem Biotechnol 144(2):111–121. https://doi.org/10.1007/s12010-007-8025-y
Shet VB, Palan AM, Rao SU, Varun C, Aishwarya U, Raja S, Goveas LC, Rao VC, Ujwal P (2018) Comparison of response surface methodology and artificial neural network to enhance the release of reducing sugars from non-edible seed cake by autoclave assisted HCl hydrolysis. 3 Biotech 8(2):127–135. https://doi.org/10.1007/s13205-018-1163-9
Singh RS, Chauhan K (2017) Inulinase production from a new inulinase producer, Penicillium oxalicum BGPUP-4. Biocatal Agric Biotechnol 9:1–10. https://doi.org/10.1016/j.bcab.2016.10.012
Singh RS, Chauhan K (2018) Production, purification, characterization and applications of fungal inulinases. Curr Biotechnol 7(3):242–260. https://doi.org/10.2174/2211550105666160512142330
Singh R, Singh R (2017) Inulinases. In: Ashok P, Negi S, Soccol CR (eds) Current developments in biotechnology and bioengineering: production, isolation and purification of industrial products. Elsevier, Amsterdam, pp 423–446. https://doi.org/10.1016/B978-0-444-63662-1.00018-X
Singh RS, Chauhan K, Kaur R, Kaur R (2018) Inulinase production in shake-flask fermentations from Mucor circinelloides BGPUP-9. J Appl Biol Biotechnol 6(6):018–025. https://doi.org/10.7324/JABB.2018.60603
Trivedi S, Divecha J, Shah A (2012) Optimization of inulinase production by a newly isolated Aspergillus tubingensis CR16 using low cost substrates. Carbohydr Polym 90(1):483–490. https://doi.org/10.1016/j.carbpol.2012.05.068
Vinoth Kumar V, Premkumar MP, Sathyaselvabala VK, Dineshkirupha S, Nandagopal J, Sivanesan S (2011) Aspergillus niger exo-inulinase purification by three phase partitioning. Eng Life Sci 11(6):607–614. https://doi.org/10.1002/elsc.201000180
Acknowledgements
Aydın Adnan Menderes University Scientific Research Projects (BAP-ADÜ-FEF-15011) provided financial support for the fungal isolates of this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yazici, S.O., Sahin, S., Biyik, H.H. et al. Optimization of fermentation parameters for high-activity inulinase production and purification from Rhizopus oryzae by Plackett–Burman and Box–Behnken. J Food Sci Technol 58, 739–751 (2021). https://doi.org/10.1007/s13197-020-04591-3
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04591-3