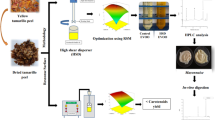
Abstract
The ultrasound assisted extraction (UAE) of total anthocyanins (TA) and total phenolic compounds (TP) from hibiscus calyces was evaluated with the intention of using the extract in future food products. The Box–Behnken Design was used for the extraction evaluating sonication time, solvent concentration and temperature. Final results suggest that 45 min, 65 °C and 25% ethanol can be considered the best region for UAE extraction. The relative area of the peaks identified by UPLC-MS/MS in the extract obtained by the UAE are superior in 2 or even 4 times those obtained by conventional extraction, for all the compounds, suggesting that the UAE method is more effective in the extraction of bioactive compounds. The extract obtained was used to make jelly and submitted to in vitro digestion and colonic fermentation. Loss of TP, TA and antioxidant activity in the jelly with addition of extract was lower when compared to extract loss itself, showing that it is feasible the hibiscus extract application in food products.



Similar content being viewed by others
References
Akhavan S, Mahdi S, Assadpour E (2016) Storage stability of encapsulated barberry’s anthocyanin and its application in jelly formulation. J Food Eng 181:59–66. https://doi.org/10.1016/j.jfoodeng.2016.03.003
Alessandro LGD, Dimitrov K, Vauchel P, Nikov I (2013) Chemical Engineering Research and Design Kinetics of ultrasound assisted extraction of anthocyanins from Aronia melanocarpa (black chokeberry) wastes. Chem Eng Res Des 2:1818–1826. https://doi.org/10.1016/j.cherd.2013.11.020
Azmir J, Zaidul ISM, Rahman MM, Sharif KM, Mohamed A, Sahena F, Jahurul MHA, Ghafoor K, Norulaini NAN, Omar AKM (2013) Techniques for extraction of bioactive compounds from plant materials : a review. J Food Eng 117:426–436. https://doi.org/10.1016/j.jfoodeng.2013.01.014
Baran A, Goud VV, Das C (2017) Extraction of phenolic compounds and anthocyanin from black and purple rice bran (Oryza sativa L.) using ultrasound : a comparative analysis and phytochemical profiling. Ind Crop Prod 95:332–341. https://doi.org/10.1016/j.indcrop.2016.10.041
Belwal T, Dhyani P, Bhatt ID, Rawal RS, Pande V (2016) Optimization extraction conditions for improving phenolic content and antioxidant activity in Berberis asiatica fruits using response surface methodology (RSM). Food Chem 207:115–124. https://doi.org/10.1016/j.foodchem.2016.03.081
Bouayed J, Hoffmann L, Bohn T (2011) Total phenolics, flavonoids, anthocyanins and antioxidant activity following simulated gastro-intestinal digestion and dialysis of apple varieties : bioaccessibility and potential uptake. Food Chem 128:14–21. https://doi.org/10.1016/j.foodchem.2011.02.052
Camelo-Méndez GA, Agama-Acevedo E, Rosell CM, Perea-Flores MJ, Bello-Pérez LA (2018) Starch and antioxidant compound release during in vitro gastrointestinal digestion of gluten-free pasta. Food Chem 263:201–207. https://doi.org/10.1016/j.foodchem.2018.04.075
Cani PD, Delzenne NM (2011) Pharmacology and therapeutics the gut microbiome as therapeutic target. Pharmacol Ther 130:202–212. https://doi.org/10.1016/j.pharmthera.2011.01.012
Celli GB, Ghanem A, Brooks MSL (2015) Optimization of ultrasound-assisted extraction of anthocyanins from haskap berries (Lonicera caerulea L.) using response surface methodology. Ultrason Sonochem 27:449–455. https://doi.org/10.1016/j.ultsonch.2015.06.014
Chen M, Zhao Y, Yu S (2015) Optimisation of ultrasonic-assisted extraction of phenolic compounds, antioxidants, and anthocyanins from sugar beet molasses. Food Chem 172:543–550. https://doi.org/10.1016/j.foodchem.2014.09.110
Cid-Ortega S, Guerrero-Beltrán JA (2015) Roselle calyces (Hibiscus sabdariffa), an alternative to the food and beverages industries : a review. J Food Sci Technol 52:6859–6869. https://doi.org/10.1007/s13197-015-1800-9
Da-costa-rocha I, Bonnlaender B, Sievers H, Pischel I, Heinrich M (2014) Hibiscus sabdariffa L.—a phytochemical and pharmacological review. Food Chem 165:424–443. https://doi.org/10.1016/j.foodchem.2014.05.002
Fernandes I, Faria A, Calhau C, Freitas V, Mateus N (2013) Bioavailability of anthocyanins and derivatives. J Funct Foods 7:54–66. https://doi.org/10.1016/j.jff.2013.05.010
Galvan L, Kriaa K, Nikov I, Dimitrov K (2012) Ultrasound assisted extraction of polyphenols from black chokeberry. Sep Purif Technol 93:42–47. https://doi.org/10.1016/j.seppur.2012.03.024
Gonçalves GA, Corrêa RCG, Barros L, Dias MI, Calhelha RC, Correa VG, Bracht A, Peralta RM, Ferreira ICFR (2019) Effects of in vitro gastrointestinal digestion and colonic fermentation on a rosemary (Rosmarinus officinalis L.) extract rich in rosmarinic acid. Food Chem 271:393–400. https://doi.org/10.1016/j.foodchem.2018.07.132
Gullon B, Pintado ME, Fernández-López J, Pérez-Álvarez JA, Viuda-Martos M (2015) In vitro gastrointestinal digestion of pomegranate peel (Punica granatum) flour obtained from co-products: changes in the antioxidant potential and bioactive compounds stability. J Funct Foods 19:617–628. https://doi.org/10.1016/j.jff.2015.09.056
Hanske L, Engst W, Loh G, Sczesny S, Blaut M, Braune A (2018) Contribution of gut bacteria to the metabolism of cyanidin 3-glucoside in human microbiota-associated rats. Br J Nutr 109:1433–1441. https://doi.org/10.1017/S0007114512003376
He B, Zhang L, Yue X, Liang J, Jiang J, Gao X, Yue P (2016) Optimization of ultrasound-assisted extraction of phenolic compounds and anthocyanins from blueberry (Vaccinium ashei) wine pomace. Food Chem 204:70–76. https://doi.org/10.1016/j.foodchem.2016.02.094
Karppinen S, Liukkonen K, Aura A-M, Pirkko F, Kaisa P (2000) In vitro fermentation of polysaccharides of rye, wheat and oat brans and inulin by human faecal bacteria. J Sci Food Agric 80:1469–1476. https://doi.org/10.1002/1097-0010(200008)80:10%3c1469:AID-JSFA675%3e3.0.CO;2-A
Koehnlein EA, Koehnlein ÉM, Corrêa RCG, Nishida VS, Correa VG, Bracht A, Peralta RM (2016) Analysis of a whole diet in terms of phenolic content and antioxidant capacity: effects of a simulated gastrointestinal digestion. Int J Food Sci Nutr 67:614–623. https://doi.org/10.1080/09637486.2016.1186156
Lees DH, Francis FJ (1972) Standardization of pigment analyses in cranberries. HortScience 7:83–84
Liang L, Wu X, Zhao T, Zhao J, Li F, Zou Y, Mao G, Yang L (2012) In vitro bioaccessibility and antioxidant activity of anthocyanins from mulberry (Morus atropurpurea Roxb.) following simulated gastro-intestinal digestion. Food Res Int 46:76–82. https://doi.org/10.1016/j.foodres.2011.11.024
Pedro AC, Granato D, Rosso ND (2016) Extraction of anthocyanins and polyphenols from black rice (Oryza sativa L.) by modeling and assessing their reversibility and stability. Food Chem 191:12–20. https://doi.org/10.1016/j.foodchem.2015.02.045
Pierpoint WS (2004) The extraction of enzymes from plant tissues rich in phenolic compounds. Methods Mol Biol 244:65–74
Pineda-Vadillo C, Nau F, Guerin-Dubiard C, Jardin J, Lechevalier V, Sanz-Buenhombre M, Guadarrama A, Tóth T, Csavajda E, Hingyi H, Karakaya S, Sibakov J, Capozzi F, Bordoni A, Dupont D (2017) The food matrix affects the anthocyanin profile of fortified egg and dairy matrices during processing and in vitro digestion. Food Chem 214:486–496. https://doi.org/10.1016/j.foodchem.2016.07.049
Pulido R, Bravo L, Saura-Calixto F (2000) Antioxidant activity of dietary polyphenols as determined by a modified ferric reducing/antioxidant power assay. J Agric Food Chem 48:3396–3402. https://doi.org/10.1021/jf9913458
Rafaella Y, Santos R, Nogueira JP, Narain N (2017) Comparison and optimization of conventional and ultrasound assisted extraction for bioactive compounds and antioxidant activity from agro-industrial acerola (Malpighia emarginata DC) residue. LWT Food Sci Technol 85:158–169. https://doi.org/10.1016/j.lwt.2017.07.020
Sancho RAS, Pavan V, Pastore GM (2015) Effect of in vitro digestion on bioactive compounds and antioxidant activity of common bean seed coats. Food Res Int 76(74–78):10. https://doi.org/10.1016/j.foodres.2014.11.042
Saura-Calixto F, Gon I (2007) Intake and bioaccessibility of total polyphenols in a whole diet. Food Chem 101:492–501. https://doi.org/10.1016/j.foodchem.2006.02.006
Sharayei P, Azarpazhooh E, Zomorodi S, Ramaswamy HS (2019) Ultrasound assisted extraction of bioactive compounds from pomegranate (Punica granatum L.) peel. LWT Food Sci Technol 101:342–350. https://doi.org/10.1016/j.lwt.2018.11.031
Shirahigue LD, Contreras-Castillo CJ, Selani MM, Nadai AP, Mourão GB, Gallo CR (2011) Winery grape-residue extract: effects on quality and sensory attributes of cooked chicken meat. Food Sci Biotechnol 20:1257–1264. https://doi.org/10.1007/s10068-011-0173-8
Sinela A, Rawat N, Mertz C, Mertz C, Achir N, Fulcrand H, Dornier M (2017) Anthocyanins degradation during storage of Hibiscus sabdariffa extract and evolution of its degradation products. Food Chem 214:234–241. https://doi.org/10.1016/j.foodchem.2016.07.071
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Tagliazucchi D, Verzelloni E, Bertolini D, Conte A (2010) In vitro bio-accessibility and antioxidant activity of grape polyphenols. Food Chem 120:599–606. https://doi.org/10.1016/j.foodchem.2009.10.030
Thaipong K, Boonprakob U, Crosby K, Cisneros-Zevalhos L, Byrne DH (2006) Comparison of ABTS, DPPH, FRAP, and ORAC assays for estimating antioxidant activity from guava fruit extracts. J Food Compos Anal 19:669–675. https://doi.org/10.1016/j.jfca.2006.01.003
Wijngaard H, Hossain MB, Rai DK, Brunton N (2012) Techniques to extract bioactive compounds from food by-products of plant origin. Food Res Int 46:505–513. https://doi.org/10.1016/j.foodres.2011.09.027
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Paraíso, C.M., dos Santos, S.S., Correa, V.G. et al. Ultrasound assisted extraction of hibiscus (Hibiscus sabdariffa L.) bioactive compounds for application as potential functional ingredient. J Food Sci Technol 56, 4667–4677 (2019). https://doi.org/10.1007/s13197-019-03919-y
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-019-03919-y