Abstract
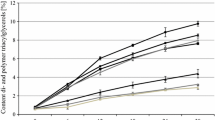
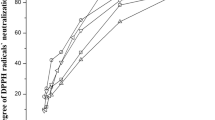
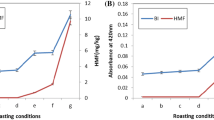
Jatropha curcas seeds are an important source of oil (5–60%), used to obtain biodiesel. The generated residual paste has a high concentration of proteins (50–55%); however, the seeds contain non-nutritional factors that limit their use. The objective of this work was to analyze the effect on the physicochemical properties of the oil obtained from J. curcas seeds subjected to different thermal treatments and to evaluate the biological quality of the proteins contained in the residual cake. The best extraction of oil (95%) was obtained after 10 h from roasted or boiled seeds. In the oil from roasted samples, the acid index increased significantly (p ≤ 0.05) with respect to the untreated sample, whereas the iodine index increased significantly (p ≤ 0.05) in the oil extracted from the boiled samples. With respect to the proximal chemical composition of the flour, roasting and boiling treatments allowed for greater oil extraction (97 and 92%), achieving, in turn, a higher content of proteins (59.56 and 58.5 g/100 g) and fiber (6.67 and 6.67 g/100 g), and lower activity of trypsin inhibitors (45 and 38%) and phytates (63 and 72%), respectively. According to the in vivo biological quality test, conducted on Wistar rats, the thermal treatments applied to the seeds improved digestibility (> 70%) and the protein efficiency index (PER). The thermal treatment allowed extracting more efficiently the oil and improved the quality of the proteins present in the residual paste.


Similar content being viewed by others
References
Abou-Arab AA, Abu-Salem FM (2010) Nutritional quality of Jatropha curcas seeds and effect of some physical and chemical treatments on their antinutritional factors. Afr J Food Sci 4(3):93–103
Anjum F, Anwar F, Jamil A, Iqbal M (2006) Microwave roasting effects on the physico-chemical composition and oxidative stability of sunflower seed oil. J Am Oil Chem Soc 83(9):777–784. https://doi.org/10.1007/s11746-006-5014-1
AOAC (2005) Official methods of analysis. Association of official analytical chemists, 14th edn. EEUU, Arlington
Aregheore EM, Becker K, Makkar HPS (2003) Detoxification of a toxic variety of Jatropha curcas using heat and chemical treatments, and preliminary nutritional evaluation with rats. South Pac J Nat Appl Sci 21(1):51–56
Arguello García E, Martínez Herrera J, Cordova-Téllez L, Sánchez O, Corona Torres T (2016) Textural, chemical and sensorial properties of maize tortillas fortified with nontoxic Jatropha curcas L. flour. CyTA-J Food 15(2):301–306. https://doi.org/10.1080/19476337.2016.1255915
Ayoola PB, Adeyeye A (2010) Effect of heating on the chemical composition and physico—chemical properties of Arachis hypogea (Groundnut) seed flour and oil. Pak J Nutr 9(8):751–754. https://doi.org/10.3923/pjn.2010.751.754
Behera S, Nagarajan S, Rao LJM (2004) Microwave heating and conventional roasting of cumin seeds (Cuminum cyminum L.) and effect on chemical composition of volatiles. Food Chem 87(1):25–29. https://doi.org/10.1016/j.foodchem.2003.10.012
Boudjeko T, Ngomoyogoli JE, Woguia AL, Yanou NN (2013) Partial characterization, antioxidative properties and hypolipidemic effects of oilseed cake of Allanblackia floribunda and Jatropha curcas. BMC Complement Altern Med 13:352. https://doi.org/10.1186/1472-6882-13-352
Chemat F, Zill-e-Huma, Khan MK (2011) Applications of ultrasound in food technology: processing, preservation and extraction. Ultrason Sonochem 18(4):813–835. https://doi.org/10.1016/j.ultsonch.2010.11.023
Chen Y, Xie MY, Gong XF (2007) Microwave-assisted extraction used for the isolation of total triterpenoid saponins from Ganoderma atrum. J Food Eng 81(1):162–170. https://doi.org/10.1016/j.jfoodeng.2006.10.018
Félix-Bernal JA, Estrada-Angulo A, Angulo-Escalante MA, Castro-Pérez BI, Landeros-López H, López-Soto MA, Barreras A, Zinn RA, Plascencia A (2016) Feeding value of supplemental Jatropha curcas crude oil in finishing diets for feedlot lambs. J Anim Sci 94(9):3875–3882. https://doi.org/10.2527/jas.2016-0598
Gutiérrez LF, Ratt C, Belkacem K (2008) Effects of drying method on the extraction yields and quality of oils from quebec sea buckthorn (Hippophaë rhamnoides L.) seeds and pulp. Food Chem 106(3):896–904. https://doi.org/10.1016/j.foodchem.2007.06.058
Hiai S, Oura H, Nakajima T (1976) Color reaction of some sapogenins and saponins with vainillin and sulfuric acid. Planta Med 29(2):116–122. https://doi.org/10.1055/s-0028-1097639
Hurrell RF, Hurrell RF, Reddy MB, Burri J, Cook JD (2002) Phytate degradation determines the effect of industrial processing and home cooking on iron absorption from cereal-based foods. Br J Nutr 88(2):117–123. https://doi.org/10.1079/BJNBJN2002594
Jarma Arroyo B, Vanegas OY, Pompelli MF, Garrido C, Bezerra NE, Jarma OA (2014) Detoxification of Jatropha curcas L seed meal as possible alternative livestock feed in the Colombian Caribbean. Rev Udca actual Divulg Cient 17(1):171–178
Knothe G (2002) Structure indices in FA chemistry How relevant is the iodine value? J Amer Oil Chem Soc 79(9):847–854. https://doi.org/10.1007/s11746-002-0569-4
León-López L, Márquez-Mota CC, Velázquez-Villegas LA, Gálvez-Mariscal A, Arrieta-Báez D, Dávila-Ortiz G, Tovar AR, Torres N (2015) Jatropha curcas protein concentrate stimulates insulin signaling, lipogenesis, protein synthesis and the PKCα pathway in rat liver. Plant Foods Hum Nutr 70(3):351–356. https://doi.org/10.1007/s11130-015-0502-9
Martínez-Herrera J, Siddhuraju P, Francis G, Dávila-Ortíz G, Becker K (2006) Chemical composition, toxic/antimetabolic constituents, and effects of different treatments on their levels, in four provenances of Jatropha curcas L. from Mexico. Food Chem 96(1):80–89. https://doi.org/10.1016/j.foodchem.2005.01.059
Martínez-Herrera J, Jiménez Martínez C, Martínez Ayala A, Garduño Siciliano L, Mora Escobedo R, Davila Ortiz G, Chamorro CG, Makkar H, Francis G, Becker K (2012) Evaluation of the nutritional quality of nontoxic kernel flour from Jatropha curcas L. in rats. J Food Qual 35(2):152–158. https://doi.org/10.1111/j.1745-4557.2011.00432.x
Martínez-Herrera J, Arguello García E, Sánchez Sánchez O, Valdés-Rodriguez OA (2016) Fortification of hotcakes from edible flour of non-toxic Mexican Jatropha curcas L. Afr J Food Sci 11(4):106–111. https://doi.org/10.5897/AJFS2016.1551
Nasar-Abbas SM, Plummer JA, Siddique KHM, White P, Harris D, Dods K (2008) Cooking quality of faba bean after storage at high temperature and the role of lignins and other phenolics in bean hardening. LWT - Food Sci Tech 41(7):1260–1267. https://doi.org/10.1016/J.LWT.2007.07.017
Nichols D, Sanderson K (2002) The nomenclature, structure, and properties of food lipids. In: Kolakowska A, Sikorski ZE (eds) Chemical, biological, and functional aspects of food lipids, 2nd edn. CRC Press Inc, Boca Ratón
Özdemir M, Açkurt F, Yildiz M, Biringen G, Gürcan T, Löker M (2001) Effect of roasting on some nutrients of hazelnuts (Corylus avellena L.). Food Chem 73(2):185–190. https://doi.org/10.1016/S0308-8146(00)00260-0
Popov S, Abdel-Fattah T, Kumar S (2016) Hydrothermal treatment for enhancing oil extraction and hydrochar production from oilseeds. Renew Energ 85:844–853. https://doi.org/10.1016/J.RENENE.2015.07.048
Rakshit KD, Darukeshwara J, Rathina Raj K, Narasimhamurthy K, Saibaba P, Bhagya S (2008) Toxicity studies of detoxified Jatropha meal (Jatropha curcas) in rats. Food Chem Toxicol 46(12):3621–3625. https://doi.org/10.1016/J.FCT.2008.09.010
Ramos LB, Sánchez RJ, De Figueiredo AK, Nolasco SM, Fernández MB (2017) Optimization of microwave pretreatment variables for canola oil extraction. J Food Process Eng 40(3):e12431. https://doi.org/10.1111/jfpe.12431
Shi J, Arunasalam K, Yeung D, Kakuda Y, Mittal G, Jiang Y (2004) Saponins from edible legumes: chemistry, processing and health benefits. J Med Food 7(1):67–78. https://doi.org/10.1089/109662004322984734
Singleton VL, Orthofe R, and Lamuela-Raventós RM (1999). Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin–Ciocalteu reagent. In: Lester P (ed) Methods in enzymology, vol 299. Academic Press, pp 152–178
Sirisomboon P, Kitchaiya P (2009) Physical properties of Jatropha curcas L. kernels after heat treatments. Biosyst Eng 102(2):244–250. https://doi.org/10.1016/J.BIOSYSTEMSENG.2008.11.003
Smith C, van Megen W, Twaalfhoven L, Hitchcock C (1980) The determination of trypsin inhibitor levels in foodstuffs. J Sci Food Agric 31(4):341–350. https://doi.org/10.1002/jsfa.2740310403
Tan CP, Che Man YB, Jinap S, Yusoff MSA (2001) Effects of microwave heating on changes in chemical and thermal properties of vegetable oils. J Am Oil Chem Soc 78(12):1227–1232. https://doi.org/10.1007/s11745-001-0418-5
Thi HT, Le BA, Le HNT, Okitsu K, Imamura K, Takenaka N, Luu BV, Maeda Y (2018) Screening of fatty acids, saccharides, and phytochemicals in Jatropha curcas seed kernel as their trimethylsilyl derivatives using gas chromatography/mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 1102–1103:66–73. https://doi.org/10.1016/j.jchromb.2018.10.009
Ulusoy BO, Karabulut I, Semra T (2004) Performance of some edible oils during heating in a steam pressure cooker. J Food Lipids 11(3):234–241. https://doi.org/10.1111/j.1745-4522.2004.01135.x
Vaintraub IA, Lapteva NA (1988) Colorimetric determination of phytate in unpurified extracts of seeds and the products of their processing. Anal Biochem 175(1):227–230. https://doi.org/10.1016/0003-2697(88)90382-X
Xiao J, Zhang H, Niu L (2015) Effect of detoxification on conformational and functional properties of Jatropha curcas proteins. Int J Food Prop 18(7):1524–1534. https://doi.org/10.1080/10942912.2011.615432
Zapata-Bustamante S, Tamayo Tenorio A, Rojano AB (2014) Effect of roasting on the secondry metabolites and antioxidant activity of Colombian cocoa clones. Rev Fac Nac Agron 68(1):7497–7507. https://doi.org/10.15446/rfnam.v68n1.47836
Acknowledgements
We are grateful for the financial support of the Instituto Politécnico Nacional through SIP project.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sánchez Chino, X.M., Corzo Ríos, L.J., Martínez Herrera, J. et al. Effect of thermal treatment on the extraction efficiency, physicochemical quality of Jatropha curcas oil, and biological quality of its proteins. J Food Sci Technol 56, 1567–1574 (2019). https://doi.org/10.1007/s13197-019-03666-0
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-019-03666-0