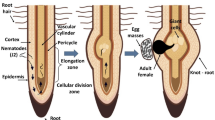
Abstract
Root-knot nematodes (RKN), Meloidogyne spp. are the most agricultural pest causing quality, yield crop losses, and increased host sensitivity to abiotic and biotic stress worldwide. For decades, the control of these parasitic nematodes was largely based on the application of nematicides. Recently, many restrictions were set on the use of nematicides due to environmental and human health concerns. Therefore, developing alternative control strategies is becoming increasingly needful to manage root-knot nematodes. Recently, biocontrol is attracting attention as a safer alternative to chemical products. It involves a wide range of control approaches including cultural practices, stimulation or introduction of antagonistic organisms, their metabolites, or their bioactive compounds. The present review aims to point out the principal biological approaches to deal with root-knot nematodes. Firstly, an informative overview of these microscopic plant-parasitic nematodes (PPN) will be provided. Then, a comparison of the main cultural practices with their potential uses, their advantages, and limitations in the management of Meloidogyne spp. will be discussed. In addition, the antagonistic activity of plant essential oils as well as that of nematophagous bacteria and fungi against Meloidogyne spp. will be described. Their nematicidal effect is discussed as they have been proven as an efficient biological control agent. The present review is a benefit to all persons interested in alternative methods for Meloidogyne spp. management.

Similar content being viewed by others
References
Abad P, Williamson VM (2010) Plant nematode interaction: a sophisticated dialogue. In: Advances in Botanical Research. Elsevier, pp 147–192
Abd-elgawad M, Askary TH (2015) Impact of Phytonematodes on Agriculture Economy. In: Askary TH, Martinelli PRP (eds) Biocontrol Agents of Phytonematodes. CAB International: Wallingford, UK, pp 3–49
Abd-Elgawad MMM, Askary TH (2018) Fungal and bacterial nematicides in integrated nematode management strategies. Egypt J Biol Pest Control 28:74. https://doi.org/10.1186/s41938-018-0080-x
Affokpon A, Coyne DL, Htay CC et al (2011) Biocontrol potential of native Trichoderma isolates against root-knot nematodes in West African vegetable production systems. Soil Biol Biochem 43:600–608. https://doi.org/10.1016/j.soilbio.2010.11.029
Ali N, Chapuis E, Tavoillot J, Mateille T (2014) Plant-parasitic nematodes associated with olive tree (Olea europaea L.) with a focus on the Mediterranean Basin: a review. Comptes Rendus - Biol 337:423–442. https://doi.org/10.1016/j.crvi.2014.05.006
Almaghrabi OA, Massoud SI, Abdelmoneim TS (2013) Influence of inoculation with plant growth promoting rhizobacteria (PGPR) on tomato plant growth and nematode reproduction under greenhouse conditions. Saudi J Biol Sci 20:57–61. https://doi.org/10.1016/j.sjbs.2012.10.004
Andrés MF, González-Coloma A, Sanz J et al (2012) Nematicidal activity of essential oils: a review. Phytochem Rev 11:371–390. https://doi.org/10.1007/s11101-012-9263-3
Anke H, Stadler M, Mayer A, Sterner O (1995) Secondary metabolites with nematicidal and antimicrobial activity from nematophagous fungi and Ascomycetes. Can J Bot 73:932–939. https://doi.org/10.1139/b95-341
Ansari RA, Rizvi R, Mahmood I (2020) Management of Phytonematodes : Recent Advances and Future Challenges. Springer: Singapore. https://doi.org/10.1007/978-981-15-4087-5
Askary TH (2015) Nematophagous fungi as biocontrol agents of phytonematodes. In: Askary TH, Martinelli PRP (eds) Biocontrol agents of phytonematodes. CAB International, Wallingford, Oxfordshire, pp 81–125
Askary TH, Martineli PRP (2015) Biocontrol agents of phytonematodes. CAB International, UK
Avato P, Tursi F, Vitali C et al (2000) Allylsulfide constituents of garlic volatile oil as antimicrobial agents. Phytomedicine 7:239–243. https://doi.org/10.1016/S0944-7113(00)80010-0
Avato P, Laquale S, Argentieri MP et al (2017) Nematicidal activity of essential oils from aromatic plants of Morocco. J Pest Sci (2004) 90:711–722. https://doi.org/10.1007/s10340-016-0805-0
Baghaee Ravari S., Mahdikhani Moghaddam E. (2016) Efficacy of Bacillus thuringiensis Cry14 toxin against root knot nematode, Meloidogyne javanica Plant Protection Science 51(No. 1):46–51. https://doi.org/10.17221/93/2013-PPS
Bais HP, Weir TL, Perry LG et al (2006) The role of root exudates in rhizosphere interactions with plants and other organisms. Annu Rev Plant Biol 57:233–266. https://doi.org/10.1146/annurev.arplant.57.032905.105159
Bakkali F, Averbeck S, Averbeck D, Idaomar M (2008) Biological effects of essential oils - a review. Food Chem Toxicol 46:446–475. https://doi.org/10.1016/j.fct.2007.09.106
Baldwin KR (2006) Crop rotations on Organic farms. Org Prod Publ Ser Environ Farming Syst 2–14
Barker KR (1991) Rotation and cropping systems for nematode control: the North Carolina experience-introduction. J Nematol 23:342–343
Bedelu T, Gessesse A, Abate D (1998) Relation of protease production to nematode-degrading ability of two Arthrobotrys spp. World J Microbiol Biotechnol 14:731–734. https://doi.org/10.1023/A:1008842706070
Béra-Maillet C, Arthaud L, Abad P, Rosso M-N (2000) Biochemical characterization of MI-ENG1, a family 5 endoglucanase secreted by the root-knot nematode Meloidogyne incognita. Eur J Biochem 267:3255–3263. https://doi.org/10.1046/j.1432-1327.2000.01356.x
Bird AF (1986) The influence of the actinomycete, Pasteuria penetrans, on the host-parasite relationship of the plant-parasitic nematode, Meloidogyne javanica. Parasitology 93:571–580. https://doi.org/10.1017/S0031182000081270
Bird DM, Opperman CH, Davies KG (2003) Interactions between bacteria and plant-parasitic nematodes: now and then. Int J Parasitol 33:1269–1276. https://doi.org/10.1016/S0020-7519(03)00160-7
Bird AF, Brisbane P., Mcclure S., Kimber RWL (1990) Studies on the properties of the spores of some populations Pasteuria penetrans. 55:169–178. https://doi.org/10.1016/0022-2011(90)90052-8
Bishop AH, Gowen SR, Pembroke B, Trotter JR (2007) Morphological and molecular characteristics of a new species of Pasteuria parasitic on Meloidogyne ardenensis. J Invertebr Pathol 96:28–33. https://doi.org/10.1016/j.jip.2007.02.008
Bonilla N, Gutiérrez-Barranquero JA, De Vicente A, Cazorla FM (2012) Enhancing soil quality and plant health through suppressive organic amendments. Diversity 4:475–491. https://doi.org/10.3390/d4040475
Bontempo AF, Fernandes RH, Lopes J, et al (2014) Pochonia chlamydosporia controls Meloidogyne incognita on carrot. Australas Plant Pathol 43:421–424. https://doi.org/10.1007/s13313-014-0283-x
Borah B, Ahmed R, Hussain M, et al (2018) Suppression of root-knot disease in Pogostemon cablin caused by Meloidogyne incognita in a rhizobacteria mediated activation of phenylpropanoid pathway. Biol Control 119:43–50. https://doi.org/10.1016/j.biocontrol.2018.01.003
Borrajo MP, Mondino EA, Maroniche GA et al (2021) Potential of rhizobacteria native to Argentina for the control of Meloidogyne javanica. Rev Argent Microbiol. https://doi.org/10.1016/j.ram.2021.02.010
Bouchagier P (2018) Survival of Root-Knot nematodes and their egg-parasitic fungus Pochonia chlamydosporia (Goddard) on weed roots. SDRP J Plant Sci 2:1–8. https://doi.org/10.25177/jps.2.2.4
Braga FR, De Araújo JV (2014) Nematophagous fungi for biological control of gastrointestinal nematodes in domestic animals. Appl Microbiol Biotechnol 98:71–82. https://doi.org/10.1007/s00253-013-5366-z
Bravo A, Gill SS, Soberón M (2007) Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control. Toxicon 49:423–435. https://doi.org/10.1016/j.toxicon.2006.11.022
Bravo A, Likitvivatanavong S, Gill SS, Soberón M (2011) Bacillus thuringiensis: a story of a successful bioinsecticide. Insect Biochem Mol Biol 41:423–431. https://doi.org/10.1016/j.ibmb.2011.02.006
Bravo A, Gómez I, Porta H et al (2012) Evolution of Bacillus thuringiensis Cry toxins insecticidal activity. Microb Biotechnol 6:17–26. https://doi.org/10.1111/j.1751-7915.2012.00342.x
Caillaud MC, Dubreuil G, Quentin M et al (2008) Root-knot nematodes manipulate plant cell functions during a compatible interaction. J Plant Physiol 165:104–113. https://doi.org/10.1016/j.jplph.2007.05.007
Castagnone-Sereno P, Piotte C, Uijthof J et al (1993) Phylogenetic relationships between amphimictic and parthenogenetic nematodes of the genus Meloidogyne as inferred from repetitive DNA analysis. Heredity (edinb) 70:195–204. https://doi.org/10.1038/hdy.1993.29
Castagnone-Sereno P, Danchin EGJ, Perfus-Barbeoch L, Abad P (2013) Diversity and evolution of root-knot nematodes, genus Meloidogyne : new insights from the genomic era. Annu Rev Phytopathol 51:203–220. https://doi.org/10.1146/annurev-phyto-082712-102300
Chen ZX, Dickson DW (1998) Review of Pasteuria penetrans: biology, ecology, and biological control potential. J Nematol 30:313–340
Chen P, Tsay TT (2006) Effect of crop rotation on Meloidogyne spp. and Pratylenchus spp. populations in strawberry fields in Taiwan. J Nematol 38:339–344
Cheng W, Yang J, Nie Q et al (2017) Volatile organic compounds from Paenibacillus polymyxa KM2501-1 control Meloidogyne incognita by multiple strategies. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-16631-8
Christaki E, Bonos E, Giannenas I, Florou-Paneri P (2012) Aromatic plants as a source of bioactive compounds. Agriculture 2:228–243. https://doi.org/10.3390/agriculture2030228
Chuan QB, Zhi LL, Qi ZL (2011) Nematicidal constituents from the essential oil of Chenopodium Ambrosioides aerial parts. E-Journal Chem 8:143–148. https://doi.org/10.1155/2011/470862
Collange B, Navarrete M, Peyre G et al (2011) Root-knot nematode (Meloidogyne) management in vegetable crop production: the challenge of an agronomic system analysis. Crop Prot 30:1251–1262. https://doi.org/10.1016/j.cropro.2011.04.016
Crickmore N (2005) Using worms to better understand how Bacillus thuringiensis kills insects. Trends Microbiol 13:347–350. https://doi.org/10.1016/j.tim.2005.06.002
Dalla Pasqua S, Dallemole-Giaretta R, dos Santos I, et al (2020) Combined application of Pochonia chlamydosporia and solid by-product of the wine industry for the control of Meloidogyne javanica. Appl Soil Ecol 147:103397. https://doi.org/10.1016/j.apsoil.2019.103397
Davies KG, Opperman CH (2006) A potential role for collagen in the attachment of Pasteuria penetrans to nematode cuticle. Multitrophic Interact Soil IOBC/wprs Bull 29:11–15
Davies K, Laird V, Kerry BR (1991) The motility, development and infection of Meloidogyne incognita encumbered with spores of the obligate hyperparasite Pasteuria penetrans. Rev Nématologie 14:611–618
Davies KG (2009) Understanding the interaction between an obligate hyperparasitic bacterium, Pasteuria penetrans and its obligate plant-parasitic nematode host, Meloidogyne spp. In: Advances in Parasitology, 1st edn. Elsevier Ltd, pp 211–245
Davis EL, Hussey RS, Mitchum MG, Baum TJ (2008) Parasitism proteins in nematode-plant interactions. Curr Opin Plant Biol 11:360–366. https://doi.org/10.1016/j.pbi.2008.04.003
de F Soares FE, Sufiate BL, de Queiroz JH (2018) Nematophagous fungi: far beyond the endoparasite, predator and ovicidal groups. Agric Nat Resour 52:1–8. https://doi.org/10.1016/j.anres.2018.05.010
De Maagd RA, Bravo A, Crickmore N (2001) How Bacillus thuringiensis has evolved specific toxins to colonize the insect world. Trends Genet 17:193–199. https://doi.org/10.1016/S0168-9525(01)02237-5
Degenkolb T, Vilcinskas A (2016) Metabolites from nematophagous fungi and nematicidal natural products from fungi as an alternative for biological control. Part I: metabolites from nematophagous ascomycetes. Appl Microbiol Biotechnol 100:3799–3812. https://doi.org/10.1007/s00253-015-7233-6
Dihingia S, Das D, Bora S (2017) Effect of microbial secrection on innhibotory effect of phytnematode: a review. Int J Inf Res Rev 04:4275–4280
Dong LQ, Zhang KQ (2006) Microbial control of plant-parasitic nematodes: a five-party interaction. Plant Soil 288:31–45. https://doi.org/10.1007/s11104-006-9009-3
Dong L, Huang C, Huang L et al (2012) Screening plants resistant against Meloidogyne incognita and integrated management of plant resources for nematode control. Crop Prot 33:34–39. https://doi.org/10.1016/j.cropro.2011.11.012
Du B, Xu Y, Dong H, et al (2020) Phanerochaete chrysosporium strain B-22, a nematophagous fungus parasitizing Meloidogyne incognita. PLoS One 15:1–14. https://doi.org/10.1371/journal.pone.0216688
Dutta TK, Khan MR, Phani V (2019) Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: current status and future prospects. Curr Plant Biol 17:17–32. https://doi.org/10.1016/j.cpb.2019.02.001
Echeverrigaray S, Zacaria J, Beltrão R (2010) Nematicidal activity of monoterpenoids against the root-knot nematode Meloidogyne incognita. Phytopathology 100:199–203. https://doi.org/10.1094/PHYTO-100-2-0199
Eisenback JD, Triantaphyllou HH (1991) Root-knot Nematodes: Meloidogyne species and races. In: Manual of Agricultural Nematology, WR Nickle. Marcel Dekker, New York, pp 191 – 274
Fassuliotis G (1985) The role of the nematologist in the development of resistant cultivars. In: J.N. Sasser and C.C. Carter (ed) An advanced treatise on Meloidogyne, Vol. I. Biology and control. N. C. State Univ., Graphics, Raleigh. pp 233–240
Favery B, Quentin M, Jaubert-Possamai S, Abad P (2016) Gall-forming root-knot nematodes hijack key plant cellular functions to induce multinucleate and hypertrophied feeding cells. J Insect Physiol 84:60–69. https://doi.org/10.1016/j.jinsphys.2015.07.013
Figueiredo AC, Barroso JG, Pedro LG, Scheffer JJC (2008) Chemical variability of the leaf oil of 113 hybrids from. 152–163. https://doi.org/10.1002/ffj
Gheysen G, Fenoll C (2002) Gene expression in nematode feeding sites. Annu Rev Phytopathol 40:191–219. https://doi.org/10.1146/annurev.phyto.40.121201.093719
Giannakou I, Pembroke B, Gowen S, Davies K (1997) Effects of long term storage and above normal temperatures on spore adhesion of Pasteuria penetrans and infection of the root-knot nematode Meloidogyne javanica. Nematologica 43:185–192
Glick BR (1995) The enhancement of plant growth by free-living bacteria. Can J Microbiol 41:109–117. https://doi.org/10.1139/m95-015
Gray EJ, Smith DL (2005) Intracellular and extracellular PGPR: commonalities and distinctions in the plant-bacterium signaling processes. Soil Biol Biochem 37:395–412
Gullino ML, Albajes R (2001) Integrated Pest and Disease Management in Greenhouse Crops. In: Gullino ML, Albajes R, Nicot PC (eds) Plant Pathology in 21st Century, 2nd edn. Springer, pp 759–760
Gupta G, Parihar SS, Ahirwar NK, et al (2015) Plant growth promoting Rhizobacteria (PGPR): current and future prospects for development of sustainable agriculture. J Microb Biochem Technol 07: https://doi.org/10.4172/1948-5948.1000188
Habazar T, Winarto Obel et al (2021) Biocontrol of Meloidogyne sp. on tomato plants by selected Bacillus spp. IOP Conf Ser Earth Environ Sci 757:012019. https://doi.org/10.1088/1755-1315/757/1/012019
Hamada N, Yimer HZ, Williamson VM, Siddique S (2020) Chemical hide and seek: nematode’s journey to its plant host. Mol Plant 13:541–543. https://doi.org/10.1016/j.molp.2020.03.005
Hassan S, Mathesius U (2012) The role of flavonoids in root-rhizosphere signalling: opportunities and challenges for improving plant-microbe interactions. J Exp Bot 63:3429–3444. https://doi.org/10.1093/jxb/err430
Hida H, Nishiyama H, Sawa S et al (2015) Chemotaxis assay of plant-parasitic nematodes on a gel-filled microchannel device. Sensors Actuators, B Chem 221:1483–1491. https://doi.org/10.1016/j.snb.2015.07.081
Hill NS (1988) Cultural practices for the management of plant parasitic nematodes. Ornamentals Northwest Arch 12:7–9
Hsueh YP, Mahanti P, Schroeder FC, Sternberg PW (2013) Nematode-trapping fungi eavesdrop on nematode pheromones. Curr Biol 23:83–86. https://doi.org/10.1016/j.cub.2012.11.035
Huang G, Dong R, Allen R et al (2005) Two chorismate mutase genes from the root-knot nematode Meloidogyne incognita. Mol Plant Pathol 6:23–30. https://doi.org/10.1111/J.1364-3703.2004.00257.X
Huang Y, Xu CK, Ma L et al (2010) Characterisation of volatiles produced from Bacillus megaterium YFM3.25 and their nematicidal activity against Meloidogyne incognita. Eur J Plant Pathol 126:417–422. https://doi.org/10.1007/s10658-009-9550-z
Huettel RN (1986) Chemical communicators in nematodes. J Nematol 18:3–8
Hussey RS (1989) Disease-inducing secretions of plant-parasitic nematodes. Annu Rev Phytopathol 27:123–141. https://doi.org/10.1146/annurev.py.27.090189.001011
Hyde K, Swe A, Zhang K (2014) Nematode-trapping fungi. In: KQ Z, K H (eds) Nematode-trapping fungi, Fungal Div. Springer
Iftikhar Y, Sajid A, Shakeel Q et al (2020) Biological antagonism: a safe and sustainable way to manage plant diseases. In: Haq IU, Ljaz S (eds) Plant Disease Management Strategies for Sustainable Agriculture through Traditional and Modern Approaches. Springer, Cham, pp 137–172
Isman MB (2000) Plant essential oils for pest and disease management. Crop Prot 19:603–608. https://doi.org/10.1016/S0261-2194(00)00079-X
James K, Back M, Prior T (2019) A literature review of the root-knot nematodes (Meloidogyne species) that pose a threat to potato production in GB
Jamshidnejad V, Sahebani N, Etebarian H (2013) Potential biocontrol activity of Arthrobotrys oligospora and Trichoderma harzianum BI against Meloidogyne javanica on tomato in the greenhouse and laboratory studies. Arch Phytopathol Plant Prot 46:1632–1640. https://doi.org/10.1080/03235408.2013.778476
Jatala P (1986) Biological control of plant-parasitic nematodes. Ann Rev Pathopathol 24:453–489
Jones JT, Haegeman A, Danchin EGJ et al (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. https://doi.org/10.1111/mpp.12057
Jones MGK, Goto DB (2011) Root-knot nematodes and giant cells. In: J. J, G. G, C. F (eds) genomics and molecular genetics of plant-nematode interactions. Springer, Dordrecht, pp 83–100
Kalaiselvi D, Mohankumar A, Shanmugam G et al (2019) Altitude-related changes in the phytochemical profile of essential oils extracted from Artemisia nilagirica and their nematicidal activity against Meloidogyne incognita. Ind Crops Prod 139:111472. https://doi.org/10.1016/j.indcrop.2019.111472
Kaloshian I, Teixeira M (2019) Advances in plant_nematode interactions with emphasis on the notorious nematode genus Meloidogyne. Phytopathology 109:1988–1996. https://doi.org/10.1094/PHYTO-05-19-0163-IA
Kamran M, Javed N, Khan SA, et al (2014) Efficacy of Pasteuria penetrans on Meloidogyne incognita reproduction and growth of tomato. Pak J Zool 46:1651–1655
Kariuki GM, Dickson DW (2007) Transfer and development of Pasteuria penetrans. J Nematol 39:55–61
Karthik Raja R, Arun A, Touray M et al (2020) Antagonists and defense mechanisms of entomopathogenic nematodes and their mutualistic bacteria. Biol Control 152:104452. https://doi.org/10.1016/j.biocontrol.2020.104452
Kimbaris AC (2017) Biocidal compounds from Mentha sp. essential oils and their structure–activity relationships. Chem Biodivers 14(3):e1600270. https://doi.org/10.1002/cbdv.201600270
Khyami-horani H, Al-Banna L (2006) Efficacy of Bacillus thuringiensis jordanica against Meloidogyne javanica infecting tomato. Phytopathol Mediterr 45:153–157. https://doi.org/10.14601/Phytopathol_Mediterr-1826
Kloepper JW, Zablotowicz RM, Tipping EM, Lifshitz R (1991) Plant growth promotion mediated by bacterial rhizosphere colonizers. Rhizosph Plant Growth 1978:315–326. https://doi.org/10.1007/978-94-011-3336-4_70
Kloepper JW, Rodríguez-Kábana R, McInroy JA, Young RW (1992) Rhizosphere bacteria antagonistic to soybean cyst (Heterodera glycines) and root-knot (Meloidogyne incognita) nematodes: identification by fatty acid analysis and frequency of biological control activity. Plant Soil 139:75–84. https://doi.org/10.1007/BF00012844
Koch DW, Gray FA, Krall JM (1998) Nematode-resistant oil radish for Heterodera schachtii control. J Sugarbeet Res 35:63–75. https://doi.org/10.5274/jsbr.35.1.63
Krebs B, Höding B, Kübart S et al (1998) Use of Bacillus subtilis as biocontrol agent. I. Activities and characterization of Bacillus subtilis strains. Z Pflanzenkrankh Pflanzenschutz 105:181–197
Kundan R, Pant G, Jadon N, Agrawal PK (2015) Plant growth promoting Rhizobacteria: mechanism and current prospective. J Biofertilizers Biopestic 06: https://doi.org/10.4172/jbfbp.1000155
Kyndt T, Vieira P, Gheysen G, de Almeida-Engler J (2013) Nematode feeding sites: unique organs in plant roots. Planta 238:807–818. https://doi.org/10.1007/s00425-013-1923-z
Laquale S, Candido V, Avato P et al (2015) Essential oils as soil biofumigants for the control of the root-knot nematode Meloidogyne incognita on tomato. Ann Appl Biol 167:217–224. https://doi.org/10.1111/aab.12221
Laquale S, Avato P, Argentieri MP et al (2018) Nematotoxic activity of essential oils from Monarda species. J Pest Sci 91:1115–1125. https://doi.org/10.1007/s10340-018-0957-1
Li B, Xie GL, Soad A, Coosemans J (2005) Suppression of Meloidogyne javanica by antagonistic and plant growth-promoting rhizobacteria. J Zhejiang Univ Sci 6(B):496–501. https://doi.org/10.1631/jzus.2005.B0496
Li J, Zou C, Xu J et al (2015) Molecular mechanisms of nematode-nematophagous microbe interactions: basis for biological control of plant-parasitic nematodes. Annu Rev Phytopathol 53:67–95. https://doi.org/10.1146/annurev-phyto-080614-120336
Liu W, Park SW (2018) Underground mystery: interactions between plant roots and parasitic nematodes. Curr Plant Biol 15:25–29. https://doi.org/10.1016/j.cpb.2018.11.004
Liu X, Xiang M, Che Y (2009) The living strategy of nematophagous fungi. Mycoscience 50:20–25. https://doi.org/10.1007/s10267-008-0451-3
Lopez-llorca LV, Macia-vicente J, Jansson H-B (2008) Mode of action and interactions of nematophagous fungi bt - integrated management of plant pests and diseases. Integrated Management and Biocontrol of Vegetable and Grain Crops Nematodes. Springer, Dordrecht, pp 51–76
Lugtenberg B, Kamilova F (2009) Plant-growth-promoting Rhizobacteria. Annu Rev Microbiol 63:541–556. https://doi.org/10.1146/annurev.micro.62.081307.162918
Malinovsky FG, Fangel JU, Willats WGT (2014) The role of the cell wall in plant immunity. Front Plant Sci 5:1–12. https://doi.org/10.3389/fpls.2014.00178
Mateille T, Trudgill DL, Trivino C et al (2002) Multisite survey of soil interactions with infestation of root-knot nematodes (Meloidogyne spp.) by Pasteuria penetrans. Soil Biol Biochem 34:1417–1424. https://doi.org/10.1016/S0038-0717(02)00085-8
Mateille T, Dabiré KR, Fould S, Diop MT (2010) Host-parasite soil communities and environmental constraints: modelling of soil functions involved in interactions between plant-parasitic nematodes and Pasteuria penetrans. Soil Biol Biochem 42:1193–1199. https://doi.org/10.1016/j.soilbio.2010.04.010
Matthiessen J, Kirkegaard J (2006) Biofumigation and enhanced biodegradation: opportunity and challenge in soilborne pest and disease management. CRC Crit Rev Plant Sci 25:235–265. https://doi.org/10.1080/07352680600611543
Mejias J, Truong NM, Abad P, et al (2019) Plant proteins and processes targeted by parasitic nematode effectors. Front Plant Sci 10: https://doi.org/10.3389/fpls.2019.00970
Messa VR, Torres da Costa AC, Kuhn OJ, Stroze CT (2020) Nematophagous and endomycorrhizal fungi in the control of Meloidogyne incognita in soybean. Rhizosphere 15:100222. https://doi.org/10.1016/j.rhisph.2020.100222
Mhatre PH, Karthik C, Kadirvelu K et al (2019) Plant growth promoting rhizobacteria (PGPR): a potential alternative tool for nematodes bio-control. Biocatal Agric Biotechnol 17:119–128. https://doi.org/10.1016/j.bcab.2018.11.009
Mhatre PH, Eapen SJ, Chawla G, et al (2020) Isolation and characterization of Pasteuria parasitizing root-knot nematode, Meloidogyne incognita, from black pepper fields in India. Egypt J Biol Pest Control 30: https://doi.org/10.1186/s41938-020-00296-z
Moens M, Perry RN, Starr JL (2009) Meloidogyne species - a diverse group of novel and important plant parasites. Root-knot Nematodes 1–17. https://doi.org/10.1079/9781845934927.0001
Mohammed SH, El Saedy MA, Enan MR, et al (2008) Biocontrol efficiency of Bacillus thuringiensis toxins against root-knot nematode, meloidogyne incognita. J Cell Mol Biol 7:57–66
Moosavi MR, Zare R (2012) Fungi as biological control agents of plant-parasitic nematodes. In: Plant Defence: Biological Control. pp 67–107
Motisi N, Doré T, Lucas P, Montfort F (2010) Dealing with the variability in biofumigation efficacy through an epidemiological framework. Soil Biol Biochem 42:2044–2057. https://doi.org/10.1016/j.soilbio.2010.08.016
Mostafanezhad H, Sahebani N, Nourinejhad Zarghani S (2014) Control of root-knot nematode (Meloidogyne javanica) with combination of Arthrobotrys oligospora and salicylic acid and study of some plant defense responses. Biocontrol Sci Technol 24:203–215. https://doi.org/10.1080/09583157.2013.855166
Mouton L, Traunecker E, McElroy K et al (2009) Identification of a polymorphic collagen-like protein in the crustacean bacteria Pasteuria ramosa. Res Microbiol 160:792–799. https://doi.org/10.1016/j.resmic.2009.08.016
Mukhtar T, Arshad Hussain M, Zameer Kayani M (2013) Biocontrol potential of Pasteuria penetrans, Pochonia chlamydosporia, Paecilomyces lilacinus and Trichoderma harzianum against Meloidogyne incognita in okra. Phytopathol Mediterr 52:66–76. https://doi.org/10.14601/Phytopathol_Mediterr-11305
Nasu É das GC, Amora DX, Monteiro TSA, et al (2018) Pochonia chlamydosporia applied via seed treatment for nematode control in two soil types. Crop Prot 114:106–112. https://doi.org/10.1016/j.cropro.2018.08.010
Negi PS (2012) Plant extracts for the control of bacterial growth: efficacy, stability and safety issues for food application. Int J Food Microbiol 156:7–17. https://doi.org/10.1016/j.ijfoodmicro.2012.03.006
Nielsen DC, Calderón FJ (2015) Fallow effects on soil. Soil Manag Build a Stable Base Agric 287–300. https://doi.org/10.2136/2011.soilmanagement.c19
Niu XM, Zhang KQ (2011) Arthrobotrys oligospora: a model organism for understanding the interaction between fungi and nematodes. Mycology 2:59–78. https://doi.org/10.1080/21501203.2011.562559
Noling JW (2016) Nematode management in tomatoes, peppers and eggplant. Univ Florida, IFAS Ext ENY-032:1–15
Nordbring-hertz B (2004) Morphogenesis in the nematode-trapping fungus. 18:125–133. https://doi.org/10.1017/S0269915XO4003052
Nordbring-hertz B (2006) Nematophagous fungi. 1–11. https://doi.org/10.1038/npg.els.0004293
Ntalli NG, Ferrari F, Giannakou I, Menkissoglu-Spiroudi U (2010) Phytochemistry and nematicidal activity of the essential oils from 8 greek lamiaceae aromatic plants and 13 terpene components. J Agric Food Chem 58:7856–7863. https://doi.org/10.1021/jf100797m
Ntalli NG, Menkissoglu-Spiroudi U (2011) Pesticides of botanical origin: a promising tool in plant protection. In: Prof. Margarita Stoytcheva (ed) Pesticides - Formulations, Effects, Fate. InTech
Ntalli NG, Nasiou E, Menkissoglu-Spiroudi U (2013) Evaluation of Essential Oils from Rosemary, Orange, Lavandula and False Yellowhead on Hatching and Motility of Root-Knot Nematode. J Agric Sci Technol A 3:603–616
Oerke EC (2006) Crop losses to pests. J Agric Sci 144:31–43. https://doi.org/10.1017/S0021859605005708
Oka Y (2001) Nematicidal activity of essential oil components against the root-knot nematode Meloidogyne javanica. Nematology 3:159–164. https://doi.org/10.1163/156854101750236286
Oka Y, Nacar S, Putievsky E, et al (2000) Nematicidal activity of essential oils and their components against the root- nematicidal activity of essential oils and their components against the root-knot nematode. https://doi.org/10.1094/PHYTO.2000.90.7.710
Onkendi EM, Kariuki GM, Marais M, Moleleki LN (2014) The threat of root-knot nematodes (Meloidogyne spp.) in Africa: A review. Plant Pathol 63:727–737. https://doi.org/10.1111/ppa.12202
Oostendorp M, Dickson DW, Mitchell DJ (1990) Host range and ecology of isolates of Pasteuria spp. from the southeastern United States. J Nematol 22:525–52531
Oota M, Tsai AYL, Aoki D et al (2020) Identification of naturally occurring polyamines as root-knot nematode attractants. Mol Plant 13:658–665. https://doi.org/10.1016/j.molp.2019.12.010
Palma L, Muñoz D, Berry C et al (2014) Bacillus thuringiensis toxins: an overview of their biocidal activity. Toxins (basel) 6:3296–3325. https://doi.org/10.3390/toxins6123296
Palomares-Rius JE, Escobar C, Cabrera J et al (2017) Anatomical alterations in plant tissues induced by plant-parasitic nematodes. Front Plant Sci 8:1–16. https://doi.org/10.3389/fpls.2017.01987
Pankaj S, Sharma HK, Prasad J (2010) The rice root-knot nematode, Meloidogyne graminicola: an emerging problem in rice-wheat cropping system. Indian J Nematol 40:1–11
Perry RN, Moens M, Starr JL (2010) Root-knot nematodes. In: Nematology. pp 483–484
Perry RN, Moens M (2011) Introduction to plant-parasitic nematodes; modes of parasitism. Genomics Mol Genet Plant-Nematode Interact. https://doi.org/10.1007/978-94-007-0434-3
Persidis A, Lay JG, Manousis T et al (1991) Characterisation of potential adhesins of the bacterium Pasteuria penetrans, and of putative receptors on the cuticle of Meloidogyne incognita, a nematode host. J Cell Sci 100:613–622
Quentin M, Abad P, Favery B (2013) Plant parasitic nematode effectors target host defense and nuclear functions to establish feeding cells. Front Plant Sci 4:1–8. https://doi.org/10.3389/fpls.2013.00053
Ravari SB, Moghaddam EM (2015) Efficacy of bacillus thuringiensis Cry14 toxin against root knot nematode, meloidogyne javanica. Plant Prot Sci 51:46–51. https://doi.org/10.17221/93/2013-PPS
Roberts P. (2003) Concepts and consequences of resistance. In: J.L. Starr, R. Cook and J B (ed) Plant resistance to parasitic nematodes. pp 6–8
Rodiuc N, Vieira P, Banora MY, de Almeida EJ (2014) On the track of transfer cell formation by specialized plant-parasitic nematodes. Front Plant Sci 5:1–14. https://doi.org/10.3389/fpls.2014.00160
Sacchetti G, Maietti S, Muzzoli M et al (2005) Comparative evaluation of 11 essential oils of different origin as functional antioxidants, antiradicals and antimicrobials in foods. Food Chem 91:621–632. https://doi.org/10.1016/j.foodchem.2004.06.031
Santana O, Andrés MF, Sanz J et al (2014) Valorization of essential oils from Moroccan aromatic plants. Nat Prod Commun 9:1109–1114. https://doi.org/10.1177/1934578x1400900812
Savary S, Ficke A, Aubertot J-N, Hollier C (2012) Crop losses due to diseases and their implications for global food production losses and food security. Food Secur 4:519–537. https://doi.org/10.1007/s12571-012-0200-5
Sayre RM, Starr MP (1985) Pasteuria penetrans (ex Thorne, 1940) nom. rev., comb. n., sp. n., a mycelial and endospore-forming bacterium parasitic in plant-parasitic nematodes. Proc Helminthol Soc Wash 52:149–165
Sayre RM, Wergin WP (1977) Bacterial parasite of a plant nematode: morphology and ultrastructure. J Bacteriol 129:1091–1101. https://doi.org/10.1128/jb.129.2.1091-1101.1977
Schneider SM, Rosskopf EN, Leesch JG et al (2003) United States Department of Agriculture - Agricultural Research Service research on alternatives to methyl bromide: pre-plant and post-harvest. Pest Manag Sci 59:814–826. https://doi.org/10.1002/ps.728
Schnepf E, Crickmore N, Van Rie J et al (1998) Bacillus thuringiensis and its pesticidal crystal proteins. Microbiol Mol Biol Rev 62:775–806. https://doi.org/10.1128/mmbr.62.3.775-806.1998
Scholte K (2000) Effect of potato used as a trap crop on potato cyst nematodes and other soil pathogens and on the growth of a subsequent main potato crop. Ann Appl Biol 136:229–238. https://doi.org/10.1111/j.1744-7348.2000.tb00029.x
Sharar M, Bozeya A, Al-Banna L, Al-Bawab A (2017) Fungicidal and nematicidal activities for essential oils formulated in Janus emulsion. Green Chem Lett Rev 10:121–128. https://doi.org/10.1080/17518253.2017.1306613
Siddiqui ZA, Mahmood I (1996) Biological control fo plant parasitic nematodes by fungi: a review. Bioresour Technol 58:229–239
Siddiqui ZA, Mahmood I (1999) Role of bacteria in the management of plant parasitic nematodes: a review. Bioresour Technol 69:167–179. https://doi.org/10.1016/S0960-8524(98)00122-9
Siddiqui IA, Haas D, Heeb S (2005b) Extracellular protease of Pseudomonas fluorescens CHA0, a biocontrol factor with activity against the root-knot nematode Meloidogyne incognita. Appl Environ Microbiol 71:5646–5649. https://doi.org/10.1128/AEM.71.9.5646-5649.2005
Siddiqui IA, Shaukat SS (2003) Suppression of root-knot disease by Pseudomonas fluorescens CHA0 in tomato : importance of bacterial secondary metabolite, 35:1615–1623. https://doi.org/10.1016/j.soilbio.2003.08.006
Siddiqui IA, Haas D, Heeb S (2005a) Extracellular protease of Pseudomonas fluorescens CHA0 , a biocontrol factor with activity against the root-knot nematode Meloidogyne incognita. 71:5646–5649. https://doi.org/10.1128/AEM.71.9.5646
Siddiqui ZA (2006) PGPR: Biocontrol and Biofertilization. Springer, Dordrecht, The Netherlands
Sikora RA, Bridge J, Starr JL (2005) Management Practices: an Overview of Integrated Nematode Management Technologies. In: M. L, R.A. S, J. B (eds) Plant parasitic nematodes in subtropical and tropical agriculture. pp 793–825
Sikora RA, Molendijk LPG, Desaeger J (2021) Integrated nematode management and crop health: future challenges and opportunities. Integr nematode Manag state-of-the-art visions Futur 3–10. https://doi.org/10.1079/9781789247541.0001
Sikora R (1988) Interrelationship between plant health promoting rhizobacteria, plant parasitic nematodes and soil microorganisms. Commun Agric Appl Biol Sci 53:867–878
Singh UB, Sahu A, Singh RK et al (2012) Evaluation of biocontrol potential of Arthrobotrys oligospora against Meloidogyne graminicola and Rhizoctonia solani in rice (Oryza sativa L.). Biol Control 60:262–270. https://doi.org/10.1016/j.biocontrol.2011.10.006
Singh R, Phulera S (2015) Plant parasitic nematodes: the hidden enemies of farmers. In: Environmental Issues for Socio-ecological Development. pp 68–81
Soliman MS, El-Deriny MM, Ibrahim DSS et al (2021) Suppression of root-knot nematode Meloidogyne incognita on tomato plants using the nematode trapping fungus Arthrobotrys oligospora Fresenius. J Appl Microbiol. https://doi.org/10.1111/jam.15101
Sosa ME, Lancelle HG, Tonn CE et al (2012) Insecticidal and nematicidal essential oils from Argentinean Eupatorium and Baccharis spp. Biochem Syst Ecol 43:132–138. https://doi.org/10.1016/j.bse.2012.03.007
Srivastava A, Mohan S, Mauchline TH, Davies KG (2018) Evidence for diversifying selection of genetic regions of encoding putative collagen-like host-adhesive fibers in Pasteuria penetrans. FEMS Microbiol Ecol 95:1–8. https://doi.org/10.1093/femsec/fiy217
Stapleton JJ, Duncan RA (1998) Soil disinfestation with cruciferous amendments and sublethal heating: effects on Meloidogyne incognita, Sclerotium rolfsii and Pythium ultimum. Plant Pathol 47:737–742
Starr MP, Sayre RM (1988) Pasteuria thornei sp. nov. and Pasteuria penetrans sensu stricto emend., mycelial and endospore-forming bacteria parasitic, respectively, on plant-parasitic nematodes of the genera Pratylenchus and Meloidogyne. Птицы 139:11–31
Stirling G, Nicol J, Reay F (2002) Advisory Services for Nematode Pests - Operational Guidelines. Biol Crop Prot Pty Ltd 111
Stirling G, Bird A, Cakurs A (1986) Attachment of Pasteuria panetrans spores to the cuticle of root-knot nematodes. Rev Nematol.9: 251–260
Stirling GR (1985) Host specificity of Pasteuria Penetrans within the genus Meloidogyne. 31:203–209
Stirling GR (2011) Biological Control of Plant-Parasitic Nematodes: An Ecological Perspective, a Review of Progress and Opportunities for Further Research. In: Davies, K., Spiegel Y (eds) Biological Control of Plant-Parasitic Nematodes: Progress in Biological Control. Springer, Dordrecht, p 311
Stirling GR (2014) Biological control of plantparasitic nematodes: soil ecosystem management in sustainable agriculture, 2nd edition
Subedi S, Thapa B, Shrestha J (2020) Root-knot nematode (Meloidogyne incognita) and its management: a review. J Agric Nat Resour 3:21–31. https://doi.org/10.3126/janr.v3i2.32298
Tazi H, Hamza MA, Hallouti A, et al (2021) Biocontrol potential of nematophagous fungi against Meloidogyne spp. infecting tomato. Org Agric 11:63–71. https://doi.org/10.1007/s13165-020-00325-z
Tazi H, Hamza MA, Hallouti A et al (2021) Biocontrol potential of nematophagous fungi against Meloidogyne spp. infecting tomato. Org Agric 11:63–71. https://doi.org/10.1007/s13165-020-00325-z
Thenail C, Joannon A, Capitaine M et al (2009) The contribution of crop-rotation organization in farms to crop-mosaic patterning at local landscape scales. Agric Ecosyst Environ 131:207–219. https://doi.org/10.1016/j.agee.2009.01.015
Tian B, Yang J, Zhang K-Q (2007) Bacteria used in the biological control of plant-parasitic nematodes: populations, mechanisms of action, and future prospects. FEMS Microbiol Ecol 61:197–213. https://doi.org/10.1111/j.1574-6941.2007.00349.x
Timper P (2009) Population dynamics of meloidogyne arenaria and pasteuria penetrans in a long-term crop rotation study. J Nematol 41:291–299
Trivedi P, Barker K (1986) Management of nematodes by cultural practices. Nematropica 16:213–236
Truong NM, Nguyen C-N, Abad P, et al (2015) Function of root-knot nematode effectors and their targets in plant parasitism. In: Carolina Escobar CF (ed) Plant Nematode Interactions A View on Compatible Interrelationships. Elsevier Ltd, pp 293–324
Tsai AYL, Higaki T, Nguyen CN et al (2019) Regulation of root-knot nematode behavior by seed-coat mucilage-derived attractants. Mol Plant 12:99–112. https://doi.org/10.1016/j.molp.2018.11.008
Uysal G, Özdemir FGG (2020) Bacteria in the management of plant parasitic. Turkish J Sci Rev 13:53–72
Van de Braak SAA., Leijten GCJ. (1999) Essential oils and oleoresins : a survey in the Netherlands and other major markets in the European Union. CBI, Cent Promot imports from Dev Ctries 116
Vejan P, Abdullah R, Khadiran T et al (2016) Role of plant growth promoting rhizobacteria in agricultural sustainability-a review. Molecules 21:1–17. https://doi.org/10.3390/molecules21050573
Verdejo-Lucas S, Talavera M (2019) Root-knot nematodes on zucchini (Cucurbita pepo subsp. pepo): pathogenicity and management. Crop Prot 126:104943. https://doi.org/10.1016/j.cropro.2019.104943
Viaene N, Coyne DL, Davies KG (2013) Biological and cultural management. Plant nematology. CABI, Wallingford, pp 383–410
Vikram, Walia RK (2015) Efficacy of Bacterial Parasite, Pasteuria penetrans Application as Seed Treatment against Root-Knot Nematode, Meloidogyne javanica. Indian J Nematol 45:1–6
Wei B-Q, Xue Q-Y, Wei L-H, Niu D-D, Liu H-X, Chen L-F, Guo J-H (2009) A novel screening strategy to identify biocontrol fungi using protease production or chitinase activity against Meloidogyne root-knot nematodes. Biocontrol Science and Technology 19(8):859–870. https://doi.org/10.1080/09583150903165636
Wei JZ, Hale K, Carta L et al (2003) Bacillus thuringiensis crystal proteins that target nematodes. Proc Natl Acad Sci U S A 100:2760–2765. https://doi.org/10.1073/pnas.0538072100
Westerdahl BB (2011) Cultural methods for managing nematodes on vegetables and ornamentals. Acta Hortic 911:185–198. https://doi.org/10.17660/ActaHortic.2011.911.18
Whipps JM (2001) Microbial interactions and biocontrol in the rhizosphere. J Exp Bot 52:487–511
Xiang N, Lawrence KS, Donald PA (2018) Biological control potential of plant growth-promoting rhizobacteria suppression of Meloidogyne incognita on cotton and Heterodera glycines on soybean: a review. J Phytopathol 166:449–458. https://doi.org/10.1111/jph.12712
Yang J, Tian B, Liang L, Zhang KQ (2007) Extracellular enzymes and the pathogenesis of nematophagous fungi. Appl Microbiol Biotechnol 75:21–31. https://doi.org/10.1007/s00253-007-0881-4
Yang JI, Loffredo A, Borneman J, Becker JO (2012) Biocontrol efficacy among strains of pochonia chlamydosporia obtained from a root-knot nematode suppressive soil. J Nematol 44:67–71
Zhai Y, Shao Z, Cai M, et al (2018) Multiple modes of nematode control by volatiles of Pseudomonas putida 1A00316 from Antarctic soil against Meloidogyne incognita. Front Microbiol 9: https://doi.org/10.3389/fmicb.2018.00253
Zhang F, Peng D, Ye X, et al (2012) In vitro uptake of 140 kDa Bacillus thuringiensis nematicidal crystal proteins by the second stage juvenile of Meloidogyne hapla. PLoS One 7: https://doi.org/10.1371/journal.pone.0038534
zi-Quan Y, Qian-Lan W, Bin L et al (2008) Bacillus thuringiensis crystal protein toxicity against plant-parasitic nematodes. Chinese J Agric Biotechnol 5:13–17. https://doi.org/10.1017/S1479236208002003
Zuckerman BM (1984) Nematode chemotaxis of host/prey recognition. Annu Rev Phytopathol 95:95–113
Author information
Authors and Affiliations
Contributions
L.A. wrote the manuscript text. M.E.M.E.B read the article, reviewed it, and provided feedback before the manuscript’s submission. E.H.M and M.B reviewed the plan of the review and provided guidance throughout the process of creating this review.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Azlay, L., El Boukhari, M., Mayad, E. et al. Biological management of root-knot nematodes (Meloidogyne spp.): a review. Org. Agr. 13, 99–117 (2023). https://doi.org/10.1007/s13165-022-00417-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13165-022-00417-y