Abstract
Flood dynamics are important drivers of wetland biodiversity. With current climate and land-use changes affecting overall water cycling, many wetland ecosystems are at risk of degradation, affecting biodiversity support negatively. This emphasizes a need for understanding possible correlations between specific hydrological conditions and biodiversity support in wetlands, at least in terms of species composition. In this study, we used high resolution hydrological monitoring of water levels and insect sampling in a depressional wetland to investigate possible correlations between inundation patterns and insect abundance. Our results show that there is a high spatial and temporal heterogeneity in wetland inundation patterns and that this heterogeneity explains variation in insect abundance. This creates episodes of downstream wet and upstream dry conditions. In addition, the spatial variability was high between grid cells of 2 meter’s resolution. There were also indications that distance to stream affected insect community structure. The findings from this work show that that the local hydrological conditions can create heterogeneity in habitat conditions, which in turn lead to refuge habitats for species vulnerable to changes in inundation condition. This study also highlights the importance of acknowledging quantitative hydrological methods when assessing the relation to insect communities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Wetlands are important ecosystems for biodiversity conservation by providing living or breeding habitats for many species while also providing important services to mankind (Davidson et al. 2019; Cheng et al. 2020; Hambäck et al. 2023). Wetland ecosystem services and functions are to a large extent driven by hydrological and hydrogeological conditions (Mitsch and Gosselink 2015; Åhlén et al. 2020). For the abundance and diversity of wetland species, waterlogging dynamics play an important role (Kuiper et al. 2014; Lafage and Pétillon 2016; Żmihorski et al. 2016). Hence, changes in the water cycle from climate change and large-scale changes in land use threatens wetlands for degradation (Fay et al. 2016; Foden et al. 2018; Albert et al. 2020; Åhlén et al. 2021), which consequently exacerbate current global biodiversity losses (Millennium Ecosystem Assessment (Program) 2005; Dudgeon et al. 2006; Bellard et al. 2012).
The degree of water saturation and flood dynamics in wetlands are important for many species (De Szalay and Resh 2000; Davidson et al. 2012; Lafage and Pétillon 2016), because floods both disturb plant and animal communities and bring in nutrients that increase primary and secondary productivity (Pollock et al. 1998; Lenssen et al. 1999; Nicolet et al. 2004). As a consequence, previous studies show that species composition differ with wetland type as the level of water saturation, i.e., permanently flooded, semi-flooded or seasonally dry, varies between wetlands (Neckles et al. 1990; Whiles and Goldowitz 2005; McInerney et al. 2017). However, these studies seldom consider the specific hydrological conditions affecting different species. Likewise, studies that look at flood zones often consider them as a homogeneous habitat (e.g., Wantzen and Junk 2000; Porej and Hetherington 2005; Datry et al. 2014), and disregard small-scale topographic variation within wetlands that create small-scale refuges. To catch such small-scale heterogeneity of importance for species abundance and diversity in wetlands, it is necessary to also derive hydrological variables at the relevant spatial scale.
There are large variabilities in flood regimes both within and among wetlands. For large wetland areas in particular, which often can be attributed to floodplains, deltas and riparian wetlands, inundation patterns are known to vary both between season and in different parts of a wetland (Hess et al. 2003; Di Vittorio and Georgakakos 2018). This variability in inundation can also be an important hydrological feature for smaller depressional wetlands (e.g., Åhlén et al. 2022) in which annual flood regimes might be less evident compared to a floodplain. For wetland biodiversity support, even small scale spatial variability (of couple of meters) in inundation can significantly increase habitat heterogeneity (Rolls et al. 2018; Jordan et al. 1997). While habitat heterogeneity has been shown to affect species composition at a landscape scale level (Rolls et al. 2018; Fahrig 2020; Kačergytė et al. 2021), this effect might also be apparent on a smaller scale, such as within an individual wetland. To sharpen our understanding on how species depend on and respond to specific hydrological conditions, it is important to examine the habitat variation not only between but also within wetlands.
In this study, we used high resolution hydrological data to investigate the relation between hydrological properties and biodiversity patterns in a depressional wetland in Uppland County, Sweden. More specifically, by simultaneously monitoring the wetland’s spatial inundation pattern and insect species abundance, we tested the hypothesis that significant and quantifiable spatial variation in inundation pattern cause variation in insect abundance also within a wetland. This study aims at demonstrating a methodological approach that better correlates species abundance to different inundation gradients by answering following questions: (i) What are the spatial and temporal inundation pattern in the wetland throughout the growing season (spring, summer and autumn)? (ii) Are there spatial differences in insect communities? and (iii) Are there identifiable relations between different inundation states and insect abundances?
Methods
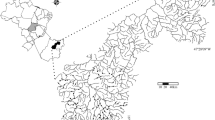
Rastmyran is a wetland located within the Vattholma catchment in Uppland County, Sweden, (60°7′35"N, 17°53′56"E) and is about 3.3 km2 large with an upstream catchment area of 37 km2. The topography of its catchment is undulating, adhering to a wave-like pattern that essentially follows the bedrock topography. The soil cover is thin, usually five meters or less, or even non-existent as reflected by frequently occurring bedrock outcrops (Antal et al. 1998). This is characteristic to large parts of southern Sweden, where the landscape supports multiple smaller streams that follow the local undulation pattern, in absence of more extensive valleys and stream floodplains. Wetlands and lakes are commonly located in depressions of the undulating landscape, and their levels are governed by multiple factors, such as the elevation of natural barriers (e.g. surficial bedrock) at their outlets, properties of adjacent shallow aquifers and bedrock properties including water conductive rock fracture zones. Hydrogeomorphologically, Rastmyran wetland can be classified as a depressional wetland, as it is located in a topographical depression with closed contours (Cole et al. 1997; Shaffer et al. 1999; see Fig. S4 in supplementary materials), close to the headwaters of the catchment. It is intersected by a small stream (about two meter wide) that flows through the wetland from south to north (Fig. 1). In contrast to, e.g., floodplain wetlands, overbank flow from the stream is just one of several sources of flooding, not least as groundwater and diffuse unmonitored flows in ditches and non-permanent stream branches can play an important role in water level changes, as shown previously for this type of wetlands and this type of landscape (Jarsjö et al. 2008).
The hydrological analysis was done by installing a pressure transducer at the outlet, based on which water levels were calculated from differential air–water pressures, recorded temperatures, the local gravitational constant (equal to 9.81 m/s2) and the temperature dependent water density. The monitoring of water levels in Rastmyran was done between March and October 2019 with 30 min intervals. The measured water levels were then combined with a digital elevation map (DEM, with a pixel resolution of 2 × 2 m; see Fig. S4 in supplementary materials for detailed information about the topographical features of Rastmyran and surrounding landscape) of the wetland (Swedish Mapping, Cadastral and Land Registration Authority 2019) to estimate the inundation frequency within each pixel. First, the daily water levels were estimated by converting the average water level for each day into meters above sea level (m.a.s.l) in order to match the unit of the DEM. Secondly, inundation for each day across the wetland was estimated by subtracting the water level to the elevation for each DEM pixel. Hence, all pixels with a lower m.a.s.l value than the current water level were considered as inundated. This procedure was repeated for all days of the monitoring period, giving a high resolution daily inundation map over the wetland for 217 days (from March to October). Finally, we estimated the inundation frequency by summing up the number of days each pixel was inundated for the whole period as well as seasonally. The inundation maps present the inundation frequency in percent of days for three periods; spring – 28 March to 31 May, summer – 1 June to 30 September and autumn – 1 October to 30 October.
To estimate insect abundances, SLAM (Sea Land Air Malaise) traps were set out for 3–4 days in end of June 2019. A total of 28 traps was positioned in a grid relative to the stream but also along a gradient from the downstream part (northern) of the wetland in order to represent a hydrological gradient of distance from water discharge area of the wetland. Trapped insects were brought into the laboratory for sorting, counting and identification (Table S3 in supplementary materials). All specimens were identified to order, and some groups were further identified to family or species. Diptera were identified to family or lower levels for selected groups (excluding difficult groups). Hemiptera (true bugs) were subdivided into suborders (Heteroptera, Auchenorrhyncha and Sternorrhyncha) and to lower levels for Heteroptera and Auchenorrhyncha. Before analyses at the order level, we subdivided orders into those with (a) aquatic larvae, (b) terrestrial plant-feeding larvae and other c) mixed larval feeding mode (Table S2 in supplementary matierals). The latter groups include orders such as Diptera and Coleoptera where larval feeding mode differs greatly among families. For the separate analysis of Diptera, we similarly subdivided families into those with (a) aquatic larvae, (b) larvae in moist soil, (c) terrestrial plant-feeding larvae and (d) other terrestrial feeding modes.
To relate hydrological conditions and insect abundances, we used manyglm in the package mvabund (Wang et al. 2012) and posthoc univariate tests with Bonferroni-corrected significance values. These tests were run separately for each larval trait groups and both at the level of insect orders and for Diptera at the family level. In this analysis, the abundances of included taxa were used as a multivariate response variable with two hydrological variables (distance to stream and inundation frequency) as response variable and a negative binomial error distribution. In the tests, we used the hydrological variables estimated for each trap location from the corresponding pixel in the DEM. For each sampling point, we repeated the analysis the inundation frequency for all pixels within a 25 m radius in order to assess the nearby spread and variability of spatial inundation. All analyses were performed in R version 4.2.1.
Results
The estimated inundation patterns for the Rastmyran wetland showed that a majority of the area is water saturated for 75% to 100% of the time during spring and autumn (Fig. 2). Overall, the wetland can be divided into three distinct inundation patterns. The upstream parts of the wetland showed a more heterogeneous inundation pattern in space and time, with the exception of summer. During summer, inundation was infrequent (between 0 to 25% of the time) for the upstream parts of the wetland. In the middle part of the wetland, inundation was less frequent during summer, compared to spring. The frequency tended to increase towards the downstream part of the wetland, i.e. along the flow direction, reaching values above 50%. In contrast, there is no distinct spatial pattern in inundation frequency perpendicular to the stream, in the transverse direction of the flow (Fig. 2 and Fig. S3 in supplementary materials), which is a consequence of topographical depressions within the wetland creating sinks and barriers for water to fill up.
The insect sampling points were distributed across the three inundation regimes, reflecting different hydrological conditions in the downstream (sample points 1A-1E), middle (2A-4F) and upstream (5A-6F) parts of the wetland (Fig. 3). In addition, most sampling points were found in areas (within 25 m from the sampling point) that are subject to considerable spatial differences in local inundation frequencies (Fig. 3).
In total, we captured 10559 insect specimens, and the insect orders with the highest number of individuals in Rastmyran wetland were Diptera (flies and midges, N = 3679) and Hemiptera (true bugs, N = 4224). Some orders with very low abundance (< 10) including Mecoptera, Orthoptera, Neuroptera and Blattodea were excluded from further analysis (see Figs. S1 & S2 in supplementary materials). Diptera and Hymenoptera (wasps) were found in the highest abundances in sampling locations 5 and 6, whereas Auchenorrhyncha (leafhoppers, treehoppers etc.) showed the highest abundances in sampling locations 1 and 2 and Trichoptera (caddisflies) showed the highest abundances in sampling locations 3 and 4 (see Fig. S2 in supplementary material).
The multivariate analysis showed that insect communities at the order level varied both in relation to inundation frequency and to the distance from the stream, with the general pattern independent of the measured resolution of inundation frequencies (Table 1). The subsequent post-hoc analysis indicated that Auchenorrhyncha was the only group varying with inundation frequency, with the highest abundances in sites with a higher inundation frequency (Fig. 4 and Table S1 in supplementary materials). Similarly, Coleoptera was the only group that varied by distance from stream and abundances were greater further away from the stream (Fig. 4).
The multivariate analysis for Diptera families showed that the composition varied in relation to inundation frequency for all feeding modes and in relation to distance from stream for taxa with aquatic larvae and for taxa with larvae in moist soil (Table 2 and Fig. 5).
Discussion
Results from this study show that inundation patterns in Rastmyran wetland vary spatially and temporally. In Rastmyran, there is a frequently inundated downstream area and a drier upstream area within a 2 km distance, with no corresponding inundation gradient away from the stream. Hence, considering that some wetlands lack distinct riparian zone structures as well as encompass complex topographical features creating natural barriers, studying shore zones might not always be a good approximation for quantifying flooding conditions. The temporal inundation pattern furthermore showed that downstream areas of Rastmyran wetland remained water saturated even during summer season while most upstream areas were only partly inundated even during the high flood events in spring and autumn. These results support the proposition that temporal and spatial hydrological conditions in wetlands are more complex and heterogeneous than accounted for when using only wetland type or riparian zones as hydrological variables (Diamond et al. 2021). Future work coupling hydrology and biodiversity should therefore acknowledge quantitative and small-scale methods in the analysis process. Results from this work also challenge the way that wetland are classified and defined, suggesting that a single wetland might support a range of hydrological conditions that traditionally might have been attributed to different types of wetlands. In order to better understand the relation between hydrology and biodiversity, our study illustrates that a hydrological analysis should be performed prior to the sampling of ecological data.
Hydrological heterogeneity of the Rastmyran wetland is not only fragmented into downstream wet and upstream dry conditions. In most areas of the wetland, we found that inundation frequencies vary extensively within a 2 m distance. Such large variability in small scale spatial inundation conditions has shown to be important for biodiversity as well as create heterogeneity at landscape and regional scale (Datry et al. 2014; Manfrin et al. 2016). In the context of climate and land-use changes that may impact wetland hydrology, our present results indicate that the observed hydrological heterogeneity could potentially harbor microhabitats which can mitigate the effect of extreme flooding events on wetland species (Batzer and Wu 2020). More specifically, for insect groups and other macroinvertebrates with limited dispersal capacity, nearby habitat conditions can act as important refuges for species that are at risk of being flooded or exposed to drought conditions too difficult to survive (Plum 2005; Rothenbücher and Schaefer 2006). However, the exact mechanisms of how nearby conditions can help species to adapt when their environment changes cannot be fully understood from this study, as multiple factors may affect species in a flooding gradient, including biotic factors such as predation and competition (Sipura et al. 2002; Verschut and Hambäck 2018).
The multivariate tests identified spatial differences in insect communities in the Rastmyran wetland connected to both inundation frequency and distance from the stream. These patterns were apparent despite that we only captured flying insects, using SLAM traps, which presumably can move over larger areas. Notice however that many dipteran species are poor fliers and often aggregate close to their breeding grounds. It is of course also possible that the different habitat conditions are strong enough drivers of population growth and attraction to species to overcome eventual dispersal effects. The reality is that we know very little about the ecology and behavior of most wetland arthropods, which is even truer for poorly studied dipteran groups. In this study, we compared responses when integrating inundation over different scales (2 or 25 m) but patterns were not greatly different. However, it is possible that scale difference were too small.
The specific groups responding to hydrological conditions indicate that both direct and indirect effects determine species abundances in the wetland. The lower abundance of dance flies (Hybotidae), which develop in wet soils, may indicate that these species are sensitive to intense flooding that cause anoxic conditions for larvae. On the other hand, the higher abundance of Auchenorrhynchaspecies with plant-eating larvae [leafhoppers (Cicadellidae – Auchenorrhyncha), gall midges (Cecidomyiidae) and grass flies (Chloropidae)] in more inundated areas likely have an indirect explanation, which may be traced to changes in their respective host plant. These species mostly feed on either live or dead plant matter and their higher abundance in more inundated areas could be either because their host plants grow wetter or because quality of these plants are better in those areas, but an alternative could also be that arthropod predators are often scarcer in more inundated areas (Sipura et al. 2002; Verschut and Hambäck 2018). Such a trend could be seen in dance flies (Hybotidae) and hover flies (Syrphidae), where the former group is predatory as both adults and larvae whereas the latter group here is dominated by species with predatory larvae (Syrphinae). A different response was seen for another predatory group, stilt flies (Dolichopodidae), that decreased in abundance away from the stream. The connection between Dolichopodidae and wetlands is logical as the larvae of many species develop in aquatic conditions or in wet soil (Gelbič and Olejníček 2011) and previous studies also show an effect of proximity to water courses (Pfister et al. 2017). Interestingly, two groups with presumed aquatic larvae (Culicidae and Tabanidae) were captured more in drier areas or away from the stream. The pattern for Culicidae (mosquitoes) may be somewhat misleading as mosquitoes are often poorly captured by Malaise traps, but it is also evident that many species of both groups are strong fliers. The same could also be applied to species with aquatic larvae, such as for Trichoptera.
To conclude, using high resolution hydrological data, this study presents a quantitative method for estimating relationships between species abundance and inundation conditions. In addition, our study shows that the scale of the analysis matters as one wetland contain multiple microhabitats due to small-scale variations in hydrological conditions over both time and space. This result opens for the consideration that even one single and heterogeneous wetland can provide refuges to sensitive species when changes in environmental conditions in proximate areas becomes prohibitive for organism survival. At last, the findings of this paper show that wetland hydrology is complex, which is not reflected in wetland classification systems and therefore need to be better acknowledged in ecological studies involving freshwater and wetland systems.
Data Availability
The data used for this study is available from Figshare repository: https://doi.org/10.6084/m9.figshare.21995609 & https://doi.org/10.6084/m9.figshare.21995555.
References
Åhlén I, Hambäck P, Thorslund J, Frampton A, Destouni G, Jarsjö J (2020) Wetlandscape size thresholds for ecosystem service delivery: Evidence from the Norrström drainage basin, Sweden. Science of the Total Environment 704:135452. https://doi.org/10.1016/j.scitotenv.2019.135452
Åhlén I, Vigouroux G, Destouni G, Pietroń J, Ghajarnia N, Anaya J, Blanco J, Borja S, Chalov S, Chun KP, Clerici N, Desormeaux A, Girard P, Gorelits O, Hansen A, Jaramillo F, Kalantari Z, Labbaci A, Licero-Villanueva L, Livsey J, Maneas G, Pisarello KLM, Pahani DM, Palomino-Ángel S, Price R, Ricaurte-Villota C, Fernanda Ricaurte L, Rivera-Monroy VH, Rodriguez A, Rodriguez E, Salgado J, Sannel B, Seifollahi-Aghmiuni S, Simard M, Sjöberg Y, Terskii P, Thorslund J, Zamora DA, Jarsjö J (2021) Hydro-climatic changes of wetlandscapes across the world. Scientific Reports 11:2754. https://doi.org/10.1038/s41598-021-81137-3
Åhlén I, Thorslund J, Hambäck P, Destouni G, Jarsjö J (2022) Wetland position in the landscape: Impact on water storage and flood buffering. Ecohydrology n/a:e2458. https://doi.org/10.1002/eco.2458
Albert JS, Destouni G, Duke-Sylvester SM, Magurran AE, Oberdorff T, Reis RE, Winemiller KO, Ripple WJ (2020) Scientists’ warning to humanity on the freshwater biodiversity crisis. Ambio. https://doi.org/10.1007/s13280-020-01318-8
Antal I, Bergman S, Gierup J, Persson C, Thunholm B (1998) Översiktsstudie av Uppsala län - Geologiska förutsättningar. Sven. Kärnbränslehantering AB SKB Rapport R-98-32
Batzer DP, Wu H (2020) Ecology of terrestrial arthropods in freshwater wetlands. Annual Review of Entomology 65:101–119. https://doi.org/10.1146/annurev-ento-011019-024902
Bellard C, Bertelsmeier C, Leadley P, Thuiller W, Courchamp F (2012) Impacts of climate change on the future of biodiversity. Ecology Letters 15:365–377. https://doi.org/10.1111/j.1461-0248.2011.01736.x
Cheng FY, Van Meter KJ, Byrnes DK, Basu NB (2020) Maximizing US nitrate removal through wetland protection and restoration. Nature 588:625–630. https://doi.org/10.1038/s41586-020-03042-5
Cole CA, Brooks RP, Wardrop DH (1997) Wetland hydrology as a function of hydrogeomorphic (HGM) subclass. Wetlands 17:456–467. https://doi.org/10.1007/BF03161511
Datry T, Corti R, Belletti B, Piégay H (2014) Ground-dwelling arthropod communities across braided river landscape mosaics: a Mediterranean perspective. Freshwater Biology 59:1308–1322. https://doi.org/10.1111/fwb.12350
Davidson TA, Mackay AW, Wolski P, Mazebedi R, Murray-Hudson M, Todd M (2012) Seasonal and spatial hydrological variability drives aquatic biodiversity in a flood-pulsed, sub-tropical wetland. Freshwater Biology 57:1253–1265. https://doi.org/10.1111/j.1365-2427.2012.02795.x
Davidson NC, van Dam AA, Finlayson CM, McInnes RJ, Davidson NC, van Dam AA, Finlayson CM, McInnes RJ (2019) Worth of wetlands: revised global monetary values of coastal and inland wetland ecosystem services. Marine and Freshwater Research 70:1189–1194. https://doi.org/10.1071/MF18391
De Szalay FA, Resh VH (2000) Factors influencing macroinvertebrate colonization of seasonal wetlands: responses to emergent plant cover. Freshwater Biology 45:295–308. https://doi.org/10.1111/j.1365-2427.2000.00623.x
Di Vittorio CA, Georgakakos AP (2018) Land cover classification and wetland inundation mapping using MODIS. Remote Sensing of Environment 204:1–17. https://doi.org/10.1016/j.rse.2017.11.001
Diamond JS, Epstein JM, Cohen MJ, McLaughlin DL, Hsueh Y-H, Keim RF, Duberstein JA (2021) A little relief: ecological functions and autogenesis of wetland microtopography. WIREs Water 8:e1493. https://doi.org/10.1002/wat2.1493
Dudgeon D, Arthington AH, Gessner MO, Kawabata Z-I, Knowler DJ, Lévêque C, Naiman RJ, Prieur-Richard A-H, Soto D, Stiassny MLJ, Sullivan CA (2006) Freshwater biodiversity: importance, threats, status and conservation challenges. Biological Reviews 81:163–182. https://doi.org/10.1017/S1464793105006950
Fahrig L (2020) Why do several small patches hold more species than few large patches? Global Ecology and Biogeography 29:615–628. https://doi.org/10.1111/geb.13059
Fay PA, Guntenspergen GR, Olker JH, Johnson WC (2016) Climate change impacts on freshwater wetland hydrology and vegetation cover cycling along a regional aridity gradient. Ecosphere 7:e01504. https://doi.org/10.1002/ecs2.1504
Foden WB, Young BE, Akçakaya HR, Garcia RA, Hoffmann AA, Stein BA, Thomas CD, Wheatley CJ, Bickford D, Carr JA, Hole DG, Martin TG, Pacifici M, Pearce-Higgins JW, Platts PJ, Visconti P, Watson JEM, Huntley B (2018) Climate change vulnerability assessment of species. Wiley Interdisciplinary Reviews Climate Change: e551. https://doi.org/10.1002/wcc.551
Gelbič I, Olejníček J (2011) Ecology of Dolichopodidae (Diptera) in a wetland habitat and their potential role as bioindicators. Open Life Sciences 6:118–129. https://doi.org/10.2478/s11535-010-0098-x
Hambäck PA, Dawson L, Geranmayeh P, Jarsjö J, Kačergytė I, Peacock M, Collentine D, Destouni G, Futter M, Hugelius G, Hedman S, Jonsson S, Klatt BK, Lindström A, Nilsson JE, Pärt T, Schneider LD, Strand JA, Urrutia-Cordero P, Åhlén D, Åhlén I, Blicharska M (2023) Tradeoffs and synergies in wetland multifunctionality: A scaling issue. Science of the Total Environment 862:160746. https://doi.org/10.1016/j.scitotenv.2022.160746
Hess LL, Melack JM, Novo EMLM, Barbosa CCF, Gastil M (2003) Dual-season mapping of wetland inundation and vegetation for the central Amazon basin. Remote Sensing of Environment 87:404–428. https://doi.org/10.1016/j.rse.2003.04.001. (Large Scale Biosphere Atmosphere Experiment in Amazonia)
Jarsjö J, Shibuo Y, Destouni G (2008) Spatial distribution of unmonitored inland water discharges to the sea. Journal of Hydrology 348:59–72. https://doi.org/10.1016/j.jhydrol.2007.09.052
Jordan F, Jelks HL, Kitchens WM (1997) Habitat structure and plant community composition in a northern Everglades wetland landscape. Wetlands 17:275–283. https://doi.org/10.1007/BF03161415
Kačergytė I, Petersson E, Arlt D, Hellström M, Knape J, Spens J, Żmihorski M, Pärt T (2021) Environmental DNA metabarcoding elucidates patterns of fish colonisation and co-occurrences with amphibians in temperate wetlands created for biodiversity. Freshwater Biology 66:1915–1929. https://doi.org/10.1111/fwb.13800
Kuiper JJ, Janse JH, Teurlincx S, Verhoeven JTA, Alkemade R (2014) The impact of river regulation on the biodiversity intactness of floodplain wetlands. Wetlands Ecology and Management 22:647–658. https://doi.org/10.1007/s11273-014-9360-8
Lafage D, Pétillon J (2016) Relative importance of management and natural flooding on spider, carabid and plant assemblages in extensively used grasslands along the Loire. Basic and Applied Ecology 17:535–545. https://doi.org/10.1016/j.baae.2016.04.002
Lenssen JPM, Menting FBJ, van der Putten WH, Blom CWPM (1999) Effects of sediment type and water level on biomass production of wetland plant species. Aquatic Botany 64:151–165. https://doi.org/10.1016/S0304-3770(99)00012-1
Manfrin A, Traversetti L, Pilotto F, Larsen S, Scalici M (2016) Effect of spatial scale on macroinvertebrate assemblages along a Mediterranean river. Hydrobiologia 765:185–196. https://doi.org/10.1007/s10750-015-2412-0
McInerney PJ, Stoffels RJ, Shackleton ME, Davey CD (2017) Flooding drives a macroinvertebrate biomass boom in ephemeral floodplain wetlands. Freshwater Science 36:726–738. https://doi.org/10.1086/694905
Millennium Ecosystem Assessment (Program) (Ed.) (2005) Ecosystems and human well-being: wetlands and water synthesis: a report of the Millennium Ecosystem Assessment. World Resources Institute, Washington, DC
Mitsch WJ, Gosselink JG (2015) Wetlands [Elektronisk resurs]. Wiley
Neckles HA, Murkin HR, Cooper JA (1990) Influences of seasonal flooding on macroinvertebrate abundance in wetland habitats. Freshwater Biology 23:311–322. https://doi.org/10.1111/j.1365-2427.1990.tb00274.x
Nicolet P, Biggs J, Fox G, Hodson MJ, Reynolds C, Whitfield M, Williams P (2004) The wetland plant and macroinvertebrate assemblages of temporary ponds in England and Wales. Biological Conservation 120:261–278. https://doi.org/10.1016/j.biocon.2004.03.010
Pfister SC, Sutter L, Albrecht M, Marini S, Schirmel J, Entling MH (2017) Positive effects of local and landscape features on predatory flies in European agricultural landscapes. Agriculture, Ecosystems & Environment 239:283–292. https://doi.org/10.1016/j.agee.2017.01.032
Plum N (2005) Terrestrial invertebrates in flooded grassland: A literature review. Wetlands 25:721–737. https://doi.org/10.1672/0277-5212(2005)025[0721:TIIFGA]2.0.CO;2
Pollock MM, Naiman RJ, Hanley TA (1998) Plant species richness in Riparian Wetlands—a test of biodiversity theory. Ecology 79:94–105. https://doi.org/10.1890/0012-9658(1998)079[0094:PSRIRW]2.0.CO;2
Porej D, Hetherington TE (2005) Designing wetlands for amphibians: the importance of predatory fish and shallow littoral zones in structuring of amphibian communities. Wetlands Ecology and Management 13:445–455. https://doi.org/10.1007/s11273-004-0522-y
Rolls RJ, Heino J, Ryder DS, Chessman BC, Growns IO, Thompson RM, Gido KB (2018) Scaling biodiversity responses to hydrological regimes. Biological Reviews 93:971–995. https://doi.org/10.1111/brv.12381
Rothenbücher J, Schaefer M (2006) Submersion tolerance in floodplain arthropod communities. Basic and Applied Ecology 7:398–408. https://doi.org/10.1016/j.baae.2006.05.005
Shaffer PW, Kentula ME, Gwin SE (1999) Characterization of wetland hydrology using hydrogeomorphic classification. Wetlands 19:490–504. https://doi.org/10.1007/BF03161688
Sipura M, Ikonen A, Tahvanainen J, Roininen H (2002) Why does the Leaf Beetle Galerucella Lineola F. Attack Wetland Willows? Ecology 83:3393–3407. https://doi.org/10.1890/0012-9658(2002)083[3393:WDTLBG]2.0.CO;2
Swedish Mapping, Cadastral and Land Registration Authority (2019) Höjddata, grid 50+ [WWW Document]. Lantmateriet.se. https://www.lantmateriet.se/sv/Kartor-och-geografisk-information/geodataprodukter/hojddata-grid-50/. Accessed 7.16.19
Verschut TA, Hambäck PA (2018) A random survival forest illustrates the importance of natural enemies compared to host plant quality on leaf beetle survival rates. BMC Ecology 18:33. https://doi.org/10.1186/s12898-018-0187-7
Wang Y, Naumann U, Wright S, Warton D (2012) mvabund–an R package for model-based analysis of multivariate abundance data. Methods Ecol Evol 3:471–474. https://doi.org/10.1111/j.2041-210X.2012.00190.x
Wantzen K, Junk W (2000) The importance of stream-wetland-systems for biodiversity: a tropical perspective. Biodiversity in Wetlands: Assessment, Function and Conservation 1:11–34
Whiles MR, Goldowitz BS (2005) Macroinvertebrate communities in central Platte River wetlands: Patterns across a hydrologic gradient. Wetlands 25:462–472
Żmihorski M, Pärt T, Gustafson T, Berg Å (2016) Effects of water level and grassland management on alpha and beta diversity of birds in restored wetlands. Journal of Applied Ecology 53:587–595. https://doi.org/10.1111/1365-2664.12588
Funding
Open access funding provided by Stockholm University. This research was supported by the Bolin Center for Climate Research, Research Area 7 (Landscape Processes and Climate) and Research Area 4 (Biogeochemical Cycles and Climate) and by a Formas grant to PAH (#2018-0843). This work was also supported by Albert and Maria Bergströms fond, Lillemor och Hans W:son Ahlmanns fond and Carl Mannerfelt fond.
Author information
Authors and Affiliations
Contributions
I.Å, J.J and P.H initiated the paper; I.Å collected the data in field with help from P.H and J.J; I.Å conducted the analysis and led the writing of the manuscript including the results interpretation with input from P.H; P.H & J.J helped improve the analysis, interpretation and manuscript text, contributing overall with their expertise.
Corresponding author
Ethics declarations
Competing Interest
The author declare that they have no competing financial interests or personal relationships that could have appeared to influence the work in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Åhlén, I., Jarsjö, J. & Hambäck, P.A. Connecting Wetland Flooding Patterns to Insect Abundance Using High-Resolution Inundation Frequency Data. Wetlands 43, 74 (2023). https://doi.org/10.1007/s13157-023-01716-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01716-0