Abstract
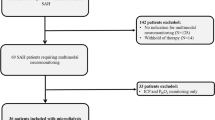
Pneumonia is a significant medical complication following aneurysmal subarachnoid hemorrhage (aSAH). The aSAH may initiate immune interactions leading to depressed immunofunction, followed by an increased risk of infection. It remains unclear as to whether there is a possible association between cerebral metabolism and infections. Clinical and microdialysis data from aSAH patients prospectively included in the CoOperative Study on Brain Injury Depolarisations protocol Berlin were analyzed. Levels of glucose, lactate, pyruvate, and glutamate were measured hourly using microdialysis in the cerebral extracellular fluid. The occurrence of pneumonia (defined by positive microbiological cultures) and delayed ischemic neurological deficits (DIND) was documented prospectively. Eighteen aSAH patients (52.7 ± 10.7 years), classified according to the World Federation of Neurological Surgeons in low (I–III, n = 9) and high (IV–V, n = 9) grades, were studied. Eight patients (45 %) experienced DIND, 10 patients (56 %) pneumonia (mean onset day 2.6). Lactate was elevated at day 3 in infected patients (n = 9, median = 6.82 mmol/L) vs. patient without infections (n = 6, median = 2.90 mmol/L, p = 0.036). The optimum cut-off point to predict pneumonia at day 3 was 3.57 mmol/L with a sensitivity of 0.77, and a specificity of 0.66 (area under curve was 0.833 with p = 0.034). Lactate at day 7 was higher in DIND patients compared to no-DIND-patients (p = 0.016). Early elevated lactate correlated with occurrence of bacterial pneumonia, while late elevations with DIND after aSAH. Future investigations may elucidate the relationship between cerebral lactate and markers of immunocompetence and more detailed to identify patients with higher susceptibility for infections.




Similar content being viewed by others
References
Oddo M, Levine JM, Frangos S, et al. Brain lactate metabolism in humans with subarachnoid hemorrhage. Stroke. 2012;43:1418–21.
Hilker R, Poetter C, Findeisen N, et al. Nosocomial pneumonia after acute stroke: implications for neurological intensive care medicine. Stroke. 2003;34:975–81.
Frontera JA, Fernandez A, et al. Impact of nosocomial infectious complications after subarachnoid hemorrhage. Neurosurgery. 2008;62:80–7.
Dirnagl U, Klehmet J, Braun JS, et al. Stroke-induced immunodepression: experimental evidence and clinical relevance. Stroke. 2007;38:770–3.
Harms H, Prass K, Meisel C, et al. Preventive antibacterial therapy in acute ischemic stroke: a randomized controlled trial. PloS One. 2008;3:2158.
Sarrafzadeh A, Schlenk F, Meisel A, et al. Immunodepression after aneurysmal subarachnoid hemorrhage. Stroke. 2011;42:53–8.
Sehba FA, Pluta R, et al. Metamorphosis of subarachnoid hemorrhage research: from delayed vasospasm to early brain injury. Mol Neurobiol. 2011;43:27–40.
Chaichana KL, Pradilla G, Huang J, Tamargo RJ. Role of inflammation (leukocyte-endothelial cell interactions) in vasospasm after subarachnoid hemorrhage. World Neurosurg. 2010;73:22–41.
Sercombe R, Dinh YR, Gomis P. Cerebrovascular inflammation following subarachnoid hemorrhage. Jpn J Pharmacol. 2002;88:227–49.
Sarrafzadeh AS, Nagel A, Czabanka M, et al. Imaging of hypoxic-ischemic penumbra with (18)F-fluoromisonidazole PET/CT and measurement of related cerebral metabolism in aneurysmal subarachnoid hemorrhage. J Cereb Blood Flow. 2010;30:36–45.
Mathiesen T, Andersson B, Loftenius A, et al. Increased interleukin-6 levels in cerebrospinal fluid following subarachnoid hemorrhage. J Neurosurg. 1993;78:562–7.
Osuka K, Suzuki Y, Tanazawa T, et al. Interleukin-6 and development of vasospasm after subarachnoid haemorrhage. Acta Neurochir. 1998;140:943–51.
Schoch B, Regel JP, Wichert M, et al. Analysis of intrathecal interleukin-6 as a potential predictive factor for vasospasm in subarachnoid hemorrhage. Neurosurgery. 2007;60:828–36. discussion 828-836.
Fassbender K, Hodapp B, Rossol S, et al. Inflammatory cytokines in subarachnoid haemorrhage: association with abnormal blood flow velocities in basal cerebral arteries. J Neurol Neurosurg Psychiatry. 2001;70:534–7.
Graetz D, Nagel A, et al. High ICP as trigger of proinflammatory IL-6 cytokine activation in aneurysmal subarachnoid hemorrhage. Neurol Res. 2009;32:728–35.
Schneider UC, Schiffler J, et al. Functional analysis of pro-inflammatory properties within the cerebrospinal fluid after subarachnoid hemorrhage in vivo and in vitro. J Neuroinflammation. 2012;9:28.
Teasdale GM, Drake CG, Hunt W, Kassell N, et al. A universal subarachnoid hemorrhage scale: report of a committee of the World Federation of Neurosurgical Societies. J Neurol Neurosurg Psych. 1988;51:1457.
Hunt WE, Hess RM. Surgical risk as related to time of intervention in the repair of intracranial aneurysms. J Neurosurg. 1968;28:14–20.
Fisher CM, Kistler JP, Davis JM. Relation of cerebral vasospasm to subarachnoid hemorrhage visualized by computerized tomographic scanning. Neurosurgery. 1980;6:1–9.
Vergouwen MDI, Vermeulen M, van Gijn J, et al. Definition of delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage as an outcome event in clinical trials and observational studies: proposal of a multidisciplinary research group. Stroke. 2010;41:2391–5.
Dreier JP, Major S, Pannek H-W, et al. Spreading convulsions, spreading depolarization and epileptogenesis in human cerebral cortex. Brain J Neurol. 2012;135:259–75.
Sarrafzadeh A, Haux D, Plotkin M, et al. Bedside microdialysis reflects dysfunction of cerebral energy metabolism in patients with aneurysmal subarachnoid hemorrhage as confirmed by 15 O-H2 O-PET and 18 F-FDG-PET. J Neuroradiol. 2005;32:348–51.
Drenckhahn C, Brabetz C, Major S, et al. Criteria for the diagnosis of noninfectious and infectious complications after aneurysmal subarachnoid hemorrhage in DISCHARGE-1. Acta Neurochir Suppl. 2013;115:153–9.
Pillai KC. Some new test criteria in multivariate analysis. Ann Math Statist. 1955;26:117–21.
Olson C. Comparative robustness of six tests in multivariate analysis of variance. J Am Stat Assoc. 1974;69:894–908.
Hillman J, Aneman O, Persson M, et al. Variations in the response of interleukins in neurosurgical intensive care patients monitored using intracerebral microdialysis. J Neurosurg. 2007;106:820–5.
Hanafy KA, Grobelny B, Fernandez L, et al. Brain interstitial fluid TNF-alpha after subarachnoid hemorrhage. J Neurol Sci. 2010;291:69–73.
Goodman JC, Valadka AB, Gopinath SP, et al. Extracellular lactate and glucose alterations in the brain after head injury measured by microdialysis. Crit Care Med. 1999;27:1965–73.
Nilsson OG, Brandt L, Ungerstedt U, Säveland H. Bedside detection of brain ischemia using intracerebral microdialysis: subarachnoid hemorrhage and delayed ischemic deterioration. Neurosurgery. 1999;45(5):1176–84.
Perry L, Love CP. Screening for dysphagia and aspiration in acute stroke: a systematic review. Dysphagia. 2001;16:7–18.
Prass K, Braun JS, Dirnagl U, Meisel C, et al. Stroke propagates bacterial aspiration to pneumonia in a model of cerebral ischemia. Stroke. 2006;37:2607–12.
Komotar RJ, Schmidt JM, Starke RM, et al. Resuscitation and critical care of poor-grade subarachnoid hemorrhage. Neurosurgery. 2009;64:397–410.
Acknowledgments
This study is supported by grants from the Deutsche Forschungsgemeinschaft (DFG DR 323/5-1) and the Bundesministerium für Bildung und Forschung (Center for Stroke Research Berlin, 01 EO 0801; Bernstein Center for Computational Neuroscience Berlin 01GQ1001C B2) to Dr. Dreier.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Radolf, S., Smoll, N., Drenckhahn, C. et al. Cerebral Lactate Correlates with Early Onset Pneumonia after Aneurysmal SAH. Transl. Stroke Res. 5, 278–285 (2014). https://doi.org/10.1007/s12975-013-0292-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-013-0292-z