Abstract
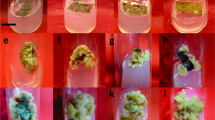
Jatropha curcas, the energy plant has attained great attention in recent years because of its biodiesel production potential; however, oil and deoiled cakes are toxic. A non-toxic variety of J. curcas is reported from Mexico. A simple and efficient protocol has been developed for plant regeneration using cotyledonary petiole explants of non-toxic variety of J. curcas. The percentage of induction of shoot buds (59.11%), and the number of shoot buds (5.01) per explant was achieved on Murashige and Skoog’s (MS) medium supplemented with 2.27 μM thidiazuron (TDZ). These induced shoot buds multiplied when subcultured on MS medium supplemented with 10 μM kinetin (Kn), 4.5 μM 6-benzyl aminopurine (BAP), and 5.5 μM α-naphthaleneacetic acid (NAA) for 4 weeks and subsequent elongation achieved on MS medium supplemented with 2.25 μM BAP and 8.5 μM indole-3-acetic acid (IAA). Shoots more than 2 cm long were harvested and cultured on MS medium containing different concentrations and combinations of IBA, IAA, NAA, and 0.25 mg L−1 activated charcoal, and 19.91% rooting was achieved in 15 μM IBA, 5.7 μM IAA, and 16.5 μM NAA after 4 weeks with more than 90% survival rate.
Similar content being viewed by others
References
Arockiasamy S, Prakash S, Ignacimuthu S. 2002. Direct organogenesis from mature leaf and petiole explants of Eryngium foetidum. Biol. Plant. 45: 129–132
Bhagwat B, Vieira LGE, Erickson LR. 1996. Stimulation of in vitro shoot proliferation from nodal explants of cassava by thidiazuron, benzyladenine and gibberellic acid. Plant Cell Tiss. Org. Cult. 46: 1–7
Christopher T. Rajam MV. 1996. Effect of genotype, explant and medium on in vitro regeneration of red pepper. Plant Cell Tiss. Org. Cult. 46: 245–250
da Camara Machado A, Frick NS, Kremen R, Katinger H, da Camara Machado ML. 1997. Biotechnological approaches to the improvement of Jatropha curcas. In Proceedings of the International Symposium on Jatropha, pp. 15. Managua, Nicaragua
Deore AC, Johnson, TS. 2008. High-frequency plant regeneration from leaf-disc cultures of Jatropha curcas L.: an important biodiesel plant. Plant Biotech. Rep. 2: 10–15
Fasolo F, Zimmerman RH, Fordham I. 1989. Adventitious shoot formation on excised leaves of in vitro grown shoots of apple cultivars. Plant Cell Tiss. Org. Cult. 16: 75–87
Huetteman CA, Preece JE. 1993. Thidiazuron: a potent cytokinin for woody plant tissue culture. Plant Cell Tiss. Org. Cult. Org. Cult. 33: 105–119
Jha T, Mukherjee P, Datta MM. 2007. Somatic embryogenesis in Jatropha curcas Linn. an important biofuel plant. Plant Biotech. Rep. 1: 135–140
Kaminek M. 1992. Progress in cytokinin research. Trends Biotech. 10: 159–162
Kumar N, Pamidimarri SDVN, Kaur M, Boricha G, Reddy MP. 2008. Effects of NaCl on growth, ion accumulation, protein, proline contents, and antioxidant enzymes activity in callus cultures of Jatropha curcas. Biologia 63: 378–382
Kumar N, Reddy MP. 2010. Plant regeneration through the direct induction of shoot buds from petiole explants of Jatropha curcas: a biofuel plant. Ann. Appl. Biol. 156: 367–375
Kumar N, Vijay Anand KG, Reddy MP. 2010. Shoot regeneration from cotyledonary leaf explants of Jatropha curcas: a biodesel plant. Acta. Physiol. Plant. (in press). DOI: 10.1007/s11738-010-0479-9
Makkar HPS, Becker K. 1997. Potential of Jatropha seed cake as protein supplement in livestock feed and constraints to its utilization. In Proceedings of Jatropha 97: International Symposium on Biofuel and Industrial Products from Jatropha curcas and Other Tropical Oil Seed Plants. Managua, Nicaragua, Mexico, 23–27
Makkar HPS, Aderibigbe AO, Becker K. 1998. Comparative evaluation of non-toxic and toxic varieties of Jatropha curcas for chemical composition, digestibility, protein degradability and toxic factors. Food Chem. 62: 207–215
Martin KP. 2003. Rapid axillary bud proliferation and ex vitro rooting of Eupatorium triplinerve. Biol. Plant. 47: 589–591
Mithila J, Hall JC, Victor JMR, Saxena PK. 2003. Thidiazuron induces shoot organogenesis at low concentrations and somatic embryogenesis at high concentrations of leaf and petiole explants of African violet (Saintpaulia ionantha Wendl.). Plant Cell Rep. 21: 408–414
Murashige T, Skoog F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–479
Ozaslan M, Can C, Aytekin T. 2005. Effect of explant source on in vitro propagation of Paulownia tomentosa Steud. Biotech. Biotech. Equip. 19: 20–26
Preece JE, Imel MR. 1991. Plant regeneration from leaf explants of Rhododendron ‘P.J.M. Hybrids’. Sci. Hort. 48: 159–170
Rajore S, Batra A. 2007. An alternative source for regenerable organogenic callus induction in Jatropha curcas. Ind. J. Biotech. 6: 545–548
Sharma R, Wakhlu A. 2001. Adventitious shoot regeneration from petiole explants of Heracleum candicans wall. In Vitro Cell. Dev. Biol. Plant 37: 794–797
Singh A, Reddy MP, Chikara J, Singh S. 2010. A simple regeneration protocol from stem explants of Jatropha curcas-A Biodisel Plant. Ind. Crops Prod. 31: 209–213
Singh A, Reddy MP, Patolia JS. 2008. An improved protocol for micropropagation of elite genotypes of Simmondsia chinensis (Link) Schneider. Biol. Plant. 52: 538–540
Sujatha M, Makkar, HPS, Becker K. 2005. Shoot bud proliferation from axillary nodes and leaf sections of non-toxic Jatropha curcas L. Plant Growth Regul. 47: 83–90
Sujatha M, Mukta N. 1996. Morphogenesis and plant regeneration from tissue cultures of Jatropha curcas. Plant Cell Tiss. Org. Cult. 44: 135–141
Venkataiah P, Christopher T, Subhash K. 2003. Thidiazuron-induced adventitious shoot bud formation and plant regeneration in Capsicum annuum L. J. Plant. Biotech. 5: 245–250
Wei Q, Wei-Da L, Liao Y, Shu-Lin P, Xu Y, Tang L, Fang C. 2004. Plant regeneration from epicotyl explants of Jatropha curcas. J. Plant Physiol. Mol. Biol. 30: 475–478
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kumar, N., Vijay Anand, K.G. & Reddy, M.P. In vitro plant regeneration of non-toxic Jatropha curcas L.: Direct shoot organogenesis from cotyledonary petiole explants. J. Crop Sci. Biotechnol. 13, 189–194 (2010). https://doi.org/10.1007/s12892-010-0039-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-010-0039-2