Abstract
LOXL2, an enzyme belonging to the LOX family, facilitates the cross-linking of extracellular matrix (ECM) elements. However, the roles of the LOXL2 gene in mechanisms of oncogenesis and tumor development have not been clearly defined. In this pan-cancer study, we examined the notable disparity in LOXL2 expression at the mRNA and protein levels among various cancer types and elucidated its interconnected roles in tumor progression, mutational profile, immune response, and cellular senescence. Apart from investigating the hyperexpression of LOXL2 being related to poorer prognosis in different types of tumors, this study also unveiled noteworthy connections between LOXL2 and genetic mutations, infiltration of tumor immune cells, and genes in immune checkpoint pathways. Further analysis revealed the participation of LOXL2 in multiple pathways related to cancer extracellular matrix remodeling and cellular senescence. Moreover, our investigation uncovered that the knockdown and inhibition of LOXL2 significantly attenuated the proliferation and migration of PC-9 and HCC-LM3 cells. The knock-down and inhibition of LOXL2 enhanced cellular senescence in lung and liver cancer cells, as confirmed by SA-β-Gal staining and quantitative RT-PCR analyses. This comprehensive analysis offers valuable insights on the functions of LOXL2 in different types of cancer and its role in regulating the senescence of cancer cells.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Globally, tumors have become a major disease threatening people’s health [1]. Despite substantial progress in the field of cancer treatment, the attainment of curative treatment remains elusive, resulting in persistently high mortality rates [2]. Targeted therapy and immunotherapy have reshaped the therapeutic landscape for cancer patients [3]. However, the therapeutic effects of these treatments exhibit significant variability among individuals and tumor types, thereby highlighting the existing disparity in gene mutation abundance and heterogeneity of the immune microenvironment [4, 5]. Hence, it is imperative to clarify the molecular pathogenesis and immune composition of tumors and conduct analytical validation of universal biomarkers.
The tumor microenvironment (TME) pertains to the internal surroundings that impact the progression and survival of tumor cells [6]. Cells synthesize and release a network of macromolecules, known as the extracellular matrix (ECM). Tumor cells, immune cells, stromal cells, and ECM components make up the TME [7]. The deposition and remodeling of ECM serve as the primary step and biochemical foundation for tumor malignant behaviors and immune cell infiltration [4, 8]. The roles of cellular senescence in tumor cell behaviors are controversial, senescent cells block tumor growth while maintaining a metabolically active state. Despite slowing down tumor progression, cellular senescence encourages metastasis, alters the TME, and affects treatment outcomes [9]. The innate immune system interacts with tumors in TME and plays a dual role in abnormal cellular senescence and elimination. This process involves the participation of CD4 + T cells, macrophages, neutrophils, and natural killer cells [10]. Therefore, it is necessary to find key molecules co-regulating immunity, senescent tumor cell and ECM.
Lysyl oxidase-like-2 (LOXL2), a copper-dependent amine oxidase, is a member of the LOX family [11]. A major role of LOXL2 is to facilitate and stabilize the cross-linking process of elastin and collagen within the ECM. Additionally, LOXL2 has been mainly researched in individual tumors, including breast cancer [12], hepatocellular carcinoma [13], non-small cell lung cancer [14], cervical cancer [15], colon cancer [16], and esophageal cancer [17], and several studies have found its association with tumor progression, metastasis, chemo-radiotherapy resistance, and worse prognosis [18,19,20,21]. However, progress towards humanized LOXL2 targeted drugs is not as expected [22, 23]. During replicative and stress-induced senescence, fibroblasts and epithelial cells exhibited elevated levels of LOXL2 expression, whereas its occurrence in oncogene-induced cellular senescence was rarely reported [24, 25]. Besides, the pathological effects and function of LOXL2 are underrecognized, and systematic pan-cancer analysis for LOXL2 is still lacking.
This study aimed to comprehensively examine the landscape of LOXL2 in pan-cancer, encompassing its expression level, prognostic value, genetic alteration, and immune infiltration. Additionally, we validated the impacts of LOXL2 on tumor proliferation, migration, and cellular senescence in vitro. The ultimate objective was to assess the potential of LOXL2 to be a prognostic and diagnostic biomarker, and afford new thought for personalized therapy.
2 Result
2.1 The expression of LOXL2 in pan cancer
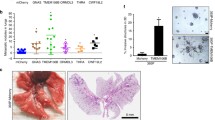
TCGA data revealed that unpaired tissue mRNA expression of LOXL2 was significantly higher in tumor tissues than in normal tissues, in ACC, BLCA, BRCA CHOL, COAD, DLBC, ESCA, GBM, HNSC, KIRC, KIRP, LAML, LGG, LIHC, LUAD, LUSC, PAAD, PCPG, READ, SKCM, STAD, TGCT, THCA, THYM, and UCS, besides being down-expressed in OV and PRAD (Fig. 1A). In paired tissue mRNA expression of pan-cancer, significantly increased expression of LOXL2 was found in tumor tissues of BLCA, BRCA, CHOL, COAD, ESCA, HNSC, KIRC, KIRP, LIHC, LUAD, LUSC, READ, STAD, THCA, and UCEC compared with their normal counterparts (Figure S1A). Additionally, the results of analysis in the CPTAC dataset showed that LOXL2 protein expression had a noteworthy increase in tumor tissues of liver cancer, breast cancer, brain cancer, colon cancer, head and neck cancer, kidney cancer, lung adenocarcinoma, ovarian cancer, pancreatic ductal adenocarcinoma, and uterine cancer, compared with corresponding normal samples (Fig. 1B). Based on the HPA database, the results of IHC in lung, breast, and liver cancer tissues and corresponding normal tissues revealed that LOXL2 protein was predominantly localized in the nucleus and ECM (Fig. 1C). Besides, the results of analysis in the TCGA database indicated that LOXL2 expression was markedly increased in BLCA, GBMLGG, LIHC, LUAD, and UVM with higher clinical or pathological stages (Figure S1B). Overall, a higher level of LOXL2 expression has been detected in tumor tissues, compared to normal tissues, indicating that LOXL2 may contribute to tumor pathogenesis through multiple potential mechanisms.
A For unpaired normal and tumor samples, mRNA expression levels of LOXL2 in different cancers were analyzed via the TCGA database, MESO and UVM lack normal tissue data; B Protein expression of LOXL2 in different normal and tumor tissues was analyzed via the CPTAC database; C The IHC staining results of LOXL2 expression in lung, breast, and liver cancers were obtained via the HPA database. * p < 0.05, **p < 0.01, and ***p < 0.001
2.2 Prognostic Signature of LOXL2
In order to examine the prognostic value of LOXL2 in pan-cancer patients, we analyzed its expression in relation to patients’ outcomes, concentrating on overall survival (OS), progression-free interval (PFI), disease-free interval (DFI), and disease specific survival (DSS). The results revealed that high-expressed LOXL2 was related to poor OS in GBMLGG, MESO, LGG, KIPAN, LUAD, CESC, UVM, PAAD, BLCA, THCA, ACC, SARC, KIRP, LAML, and LIHC. In patients with CESC, PAAD, BLCA, LUAD, MESO, LGG, UVM, and LIHC, elevated LOXL2 expression indicated decreased survival time according to Kaplan–Meier survival analysis (Fig. 2A), and non-significant correlations were shown in Figure S2. LOXL2 expression was correlated with DSS in various cancers, including LUAD, GBMLGG, and other 15 types of cancer (Fig. 2B). It also revealed that LOXL2 expression was significantly associated with DFI in 13 types of cancers (Fig. 2C). Moreover, the expression of LOXL2 was remarkably related to PFI in 24 types of cancer (Fig. 2D). These results suggested that LOXL2 might be a useful biomarker in predicting patient prognosis, especially in LUAD, CESC, PAAD, and ACC, and the contribution of LOXL2 in tumor progression is yet to be fully elucidated.
A The relationship between LOXL2 expression and OS was analyzed using the univariate Cox regression method in pan-cancer. In the TCGA database, Kaplan–Meier methodology was used to compare the expression of LOXL2 in different cancer types, showing OS curves for high and low expression; B–D The association between LOXL2 expression and DSS, DFI, and PFI was analyzed using univariate Cox regression analysis
2.3 Genetic alteration of LOXL2
It is acknowledged that the accumulation of genetic mutations is a key factor of human cancer and also a feature of cellular senescence [9, 26]. The results of analysis in cBioPortal showed that genetic alteration types and frequencies of LOXL2 differ in multiple cancers. The main genetic alteration type of LOXL2 in pan-cancer is “Deep Deletion” and the top 3 cancer types with high alteration frequencies were UCEC, SKCM, and PRAD. The LOXL2 gene alterations were all “Deep Deletion” in UCS, DLBC, CHOL, UVM, and TGCT. In PCPG and THYM, the LOXL2 gene alterations were all “Amplification” (Fig. 3A).
A The mutation profile of LOXL2 revealed “Deep Deletion” was the major type of mutation by TCGA; B The mutational landscape of LOXL2 across various cancers was examined by TCGA; C Genetic alteration disparities in UCEC, SKCM, COAD, and ESCA with varying expression levels of LOXL2 by TCGA; D, E The correlation of survival with LOXL2 SNV and CNV in pan-cancer via GSCA.
LOXL2 mutates extensively in various cancer species (Fig. 3B). The top 3 high-frequency mutations were 5.9% in SKCM, 4.6% in UCEC, and 3.5% in COAD (Fig. 3B). Furthermore, we evaluated the differences in gene alteration frequencies of tumors with higher expression of LOXL2, among the forementioned four types of tumors (UCEC, SKCM, COAD, and ESCA), APOB, USH2A, VPS13B, and KMT2D had the highest alteration frequency in the above cancer types (Fig. 3C). Besides, cancer patients with LOXL2 alternations of stable nuclear variant (SNV) had worse OS and PFS than those with no alterations in BRCA and CESC, while the copy number variation (CNV) of LOXL2 is associated with a worse prognosis in KIRP, UCEC, and BRCA (Fig. 3D, E). The above results show that the genetic alteration of LOXL2 may play a crucial role in tumorigenesis and tumor progression.
2.4 Immunological role of LOXL2
The relationship between immune cell infiltration and LOXL2 expression was examined using the xCell and TIMER algorithms. The correlation between immune cell infiltration, specifically B cell in 12 types, CD4 + T cell in 23 types, CD8 + T cell in 18 types, neutrophil in 29 types, macrophage in 30 types, and dendritic cell in 31 types, and LOXL2 expression was examined in 38 types of tumors using the TIMER method (Fig. 4A). Our analysis revealed associations between the majority of the 64 immune cell subtypes and LOXL2 expression in various tumor types (Fig. 4B). The role of immune checkpoint genes in tumor immunotherapy is crucial [27]. We conducted investigations to examine the association between the expression of LOXL2 and the genes in immune checkpoint pathways [28]. Our findings indicated a significant relationship between LOXL2 expression and the majority of the genes in immune checkpoint pathways, like VEGFA, CD276, TGFB1, and IL10 in pan-cancer. Furthermore, we observed a positive correlation between LOXL2 expression and genes in major checkpoint pathways in KIPAN, BLCA, KICH, READ, COAD, PRAD, LIHC, OV, UVM, GBMLGG, and LGG (Fig. 4C). Consequently, the immune infiltration scores, stromal scores and Pearson correlation of LOXL2 expression in pan-cancer have been calculated via the ESTIMATE algorithm. Among them, the LOXL2 pattern has a positive correlation with immune scores, and stromal scores in PAAD, LIHC, LGG, BLCA, COAD, and ESCA (Fig. 4D). LOXL2 expression was positively associated with the number of CD4 + T and CD8 + T cells. The stromal scores in 31 tumors also have a positive correlation with the RNA levels of LOXL2 via the xCell algorithm (Fig. 4B). TME associated fibroblast, pericyte, and endothelial cell also showed significant negative associations in multiple cancers (Fig. 4B). Above all, LOXL2 has the potential to regulate stromal cells, modify the tumor immune microenvironment, and impact the response of immunotherapy in multiple tumors.
A Heatmap showing correlations of LOXL2 expression with immune infiltration level in various TCGA cancer types using TIMER; B Heatmap of relationships of LOXL2 expression with infiltrated cells in various TCGA cancer types using xCell; C The connection between LOXL2 expression and genes in immune checkpoint pathways; D The correlation of LOXL2 expression and stromal scores, immune scores in pan-cancer via ESTIMATE and TCGA databases
2.5 Enrichment analysis of LOXL2-related genes
In order to comprehensively elucidate the protein co-expression, potential biological functions, and signaling pathways associated with LOXL2, an analysis was performed using GeneMANIA and GSEA. The protein–protein interaction (PPI) network generated from GeneMANIA uncovered interactions between LOXL2 and SNAI1, LOX, LOXL1, LOXL3, EGFL7, and VEGFC (Fig. 5A). Among them, there was ample evidence that SNAI1 has a physical interaction with LOXL2. Meanwhile, the functional analysis indicated that LOXL2 and its molecular partners were primarily focused on protein oxidation, oxidoreductase function, acting on the CH-NH2 group of donors, oxygen as acceptor, extracellular matrix organization, complex of collagen trimers, and collagen-containing extracellular matrix (Fig. 5A).
A Through GeneMANIA, LOXL2 and its molecular partners form a PPI network; B KEGG enrichment analysis of the gene set from GEPIA2; C GSEA analysis of LOXL2 in different cancers. Various colored curves represent distinct functions or pathways controlled by various types of cancer. Positive regulation is indicated by peaks on the ascending curve, while negative regulation is indicated by peaks on the descending curve.
The top 100 genes with the most comparable expression patterns to LOXL2 in pan-cancer were extracted from GEPIA2. The above 100 genes were closely related to cell migration, adhesion, and cellular senescence based on the KEGG enrichment analysis (Fig. 5B). Based on the median LOXL2 mRNA expression level, TCGA samples were divided into high and low expression levels for LOXL2, and enriched gene collections were identified. As a result of the GSEA analysis, the high LOXL2 expression group showed altered cancer-associated (Cell Cycle, TP53 Regulation), TME-associated (Collagen Degradation) and cellular senescence-associated (DNA Repair, Telomere Maintenance, Metabolic pathway) pathways in PAAD, LIHC, LUAD, and UVM (Fig. 5C).
2.6 LOXL2 expression effects cell proliferation and migration
According to the previous bioinformatic data, the occurrence and development of tumors in pan-cancer are strongly correlated with the expression of LOXL2. The effects of LOXL2 on proliferation and migration were examined in PC-9 and HCC-LM3 cells using CCK-8 and wound healing assays. The expression of LOXL2 was down-regulated by siRNA and LOXL2 inhibitors in PC-9 and HCC-LM3 cells (Figure S3). PC-9 and HCC-LM3 cells were significantly impeded in proliferation by decreasing LOXL2 expression in the CCK-8 assay at 48 h, 72 h and 96 h. This finding was further supported by the administration of a LOXL2 inhibitor (1 µM) (Fig. 6A). Furthermore, the wound healing experiments demonstrated that LOXL2 knockdown impaired the migration of PC-9 and HCC-LM3 cells. Consistent outcomes were observed in wound healing assays following treatment with the LOXL2 inhibitor (1 µM) for 48 h (Fig. 6B). The findings suggested that LOXL2 has the ability to suppress the growth and migration of tumor cells.
A The impact of LOXL2 on cell proliferation in PC-9 and HCC-LM3 cells was assessed using the CCK8 assay at 24, 48, 72, and 96 h; B The migration ability of PC-9 and HCC-LM3 cells was compromised by both LOXL2 knockdown and the LOXL2 inhibitor. Scale bar = 500 µm. The statistical findings from the scratch wound-healing assays at 24 h are displayed as the average plus standard deviation (three replicates). * P < 0.05; ** P < 0.01; *** P < 0.001; **** P < 0.0001
2.7 Down-regulation of LOXL2 promotes cellular senescence
LOXL2 has been associated with replicative and stress-induced senescence in previous studies of skeletal muscle and lung diseases, whereas it is rarely reported in oncogene induced cellular senescence [24, 25]. Moreover, GSEA results showed that LOXL2 was involved in “DNA Repair”, “Telomere Maintenance” and “Collagen Degradation”, which were closely related to cellular senescence.
To examine the influence of LOXL2 on cellular senescence in tumors, we performed β-galactosidase (SA-β-gal) staining and real-time quantitative PCR assays in lung cancer and liver cancer cells. The SA-β-Gal staining results indicated that knock-down of LOXL2 facilitated cellular senescence in lung and liver cancer cells (Fig. 7A). Furthermore, knockdown of LOXL2 resulted in elevated levels of the cellular senescence markers CDKN1A and CDKN2A (positively correlated with cellular senescence) (Fig. 7B). These findings were consistently observed after treatment with LOXL2 inhibitor. Therefore, our results suggest that LOXL2 may depress cellular senescence in multiple tumors.
3 Method
3.1 Pan-cancer expression
To compare the LOXL2 (ENSG00000134013) expression in tumor and normal tissues, we extracted the mRNA expression data of pan-cancer from the Cancer Genome Atlas (TCGA) database via the GDC portal (https://portal.gdc.cancer.gov/repository). The data were converted into relative expression (transcripts per million /TPM) and the expression value was transformed into log2(data + 1) form. The R software package “ggplot2” (version 3.3.3, https://cran.r-project.org/web/packages/ggplot2/index.html) was utilized to display LOXL2 expression levels in cancer and adjacent normal tissues. The significance of the difference in expression between tumor and normal tissues was assessed through the Wilcoxon rank sum test. Additionally, the protein expression of LOXL2 in normal and primary tumor tissues was examined through the Clinical Proteomic Tumor Analysis Consortium (CPTAC) module within the UALCAN portal [29] (https://ualcan.path.uab.edu/analysis-prot.html). Besides, we present an overview of LOXL2 protein expression across lung, breast, and liver cancers from the immunohistochemistry (IHC) pictures in the HPA database (https://www.proteinatlas.org/).
3.2 Prognostic signature
The survival data of LOXL2 in 44 tumors was obtained from the UCSC [30] (https://xenabrowser.net/) of TCGA PANCAN with TARGET PANCAN and GTEx (N = 19,131, G = 60,499). We also excluded the cancers with less than 10 samples in a single cancer, and finally obtained the expression data and the overall survival data of all samples in the 44 cancers, as shown above. The univariate Cox proportional hazards regression model was utilized to examine the association between gene expression and clinical outcome in the R software package “survival” (version 3.2–7). The prognostic significance of LOXL2 expression was assessed using a log-rank test. R packages “survminer” and “ggplot2” [31] generated Kaplan–Meier curves and forest plots to present the outcomes of OS, PFI, DFI, and DSS.
3.3 Genetic alteration
To uncover the genetic alteration of LOXL2, we utilized the cBioPortal (http://www.cbioportal.org/) database, an open-access database designed for interactive exploration of pan-cancer genomics datasets. The cohort of “TCGA Pan-Cancer Atlas Studies” was chosen for analysis. Additionally, we obtained comprehensive cancer genomic data from the UCSC Xena database (https://xena.ucsc.edu/), specifically the TCGA TARGET GTEx Pan-Cancer data, and conducted an analysis on the genomic alteration of LOXL2 across various cancer types. Moreover, we employed the GDC portal, MuTect2 software [32], and the R software package “maftools” [33] to integrate, calculate, and display the simple nucleotide mutations of UCEC, SKCM, COAD, and ESCA genomic data, which were sourced from the TCGA database. The results of genomic alteration were depicted using bar plots, and the genomic alteration rate was visualized by a lollipop plot. The results of genomic alteration (stable nuclear variant (SNV) and copy number variation (CNV)) correlation with survival were calculated via GSCA [34] (https://guolab.wchscu.cn/GSCA/#/mutation).
3.4 Immune infiltration landscape
The Tumor Immune Estimation Resource (TIMER) [35] is a useful tool for systematically analyzing immune infiltrates within various cancer types. In this study, we employed the TIMER and xCell methods from the R package “IOBR” [36] to examine the connection between LOXL2 expression and the quantity of immune cells in each patient. Furthermore, we performed a co-expression analysis of LOXL2 with genes in immune checkpoint pathways (inhibitory (24) and stimulatory (36)) [28] using Spearman correlation, with statistical significance defined as p < 0.05. To further evaluate the relation of LOXL2 with immune and stromal scores in pan-cancer, we employed R package “ESTIMATE” (version 1.0.13) and the xCell method. To visualize the results, we employed the R packages “limma”, “reshape2” and “RColorBreyer”.
3.5 Gene ontology enrichment analysis
To investigate the intrinsic mechanisms and signaling routes linked to LOXL2, we utilized GeneMANIA (http://genemania.org/), an online database [37] renowned for building protein–protein interaction (PPI) networks, to forecast the protein that interacted with LOXL2 based on fundamental parameters such as co-expression, co-location, and physical interaction. The top 20 genes were identified and employed to construct the PPI network. To uncover the potential molecular partners in pan-cancer, the TCGA dataset was analyzed using the Similar Gene Detection module of GEPIA2 (https://gepia2.cancer-pku.cn/) to identify genes exhibiting expression patterns that closely resemble LOXL2. The GO and KEGG databases were utilized for gene and pathway annotation, while the R packages “clusterProfiler” [38] and “ggplot2” were employed to visualize the enrichment results. In addition, we conducted the gene set enrichment analysis (GSEA, https://www.gsea-msigdb.org/gsea/downloads.jsp). High- and low-expression groups of LOXL2 were defined based on patient mRNA levels, and subsequently subjected to GSEA enrichment analysis using the Rectome database (https://reactome.org/) and Wiki pathways gene sets (http://wikipathways.org/). Pathways that showed significant enrichment were identified using the following criteria: |NES|> 1, p < 0.05, and FDR q < 0.25.
3.6 Cell culture
Human lung cancer cell line PC-9 and human hepatocellular carcinoma cell line HCC-LM3 were acquired from the Cell Bank of China Academy of Sciences (Shanghai, China), and cultured in RPMI1640 and DMEM media, respectively. Transfection of siRNAs was performed using the Lipofectamine™ 3000 kit (Invitrogen, USA), according to the manufacturers’ instructions. All siRNA sequences were designed and synthesized via Sangon Biotech (Shanghai, China). The siRNA sequence targeting LOXL2 included sense: 5’-GAAACCCTCCAGTCTATTATA-3’; antisense: 5’-TATAATAGACTGGAGGGTCTT-3’. (2-Chloropyridin-4-yl) methanamine hydrochloride (MCE, State of New Jersey, USA) is a selective inhibitor for LOXL2 [39, 40].
3.7 Real-time quantitative PCR
Total RNA was extracted with the RNA fast2000 kit (Fastagen, shanghai, China), reversed-transcribed into complementary DNA (cDNA) using the Reverse Transcription kit (Takara, Dalian, China) following the manufacturer’s protocol. Real-time quantitative PCR was conducted with the Universal SYBR qPCR Master Mix (Vazyme, Nanjing, China) and Bio-Rad CFX96TM Real-time System. The β-actin gene was used as an endogenous control. The experiment was done with three technical replicates. Primer sequences are listed in Table 1.
3.8 Cell counting kit-8 (CCK8) assay
Cell proliferation was measured using a CCK-8 kit (NCM Biotech, Suzhou, China). Transfected cells or 1 μM inhibitor treated cells were plated in 96-well plates at a density of 2000–3000 cells/well, and the optical density of the samples was determined at 450 nm with a microplate reader at 0, 24 h, 48 h, 72 h, and 96 h after staining with CCK-8 reagent. Statistical significance was determined using two-tailed Student's t-tests.
3.9 SA-β-galactosidase (SA-β-Gal) staining
The plates were filled with cells at a concentration of 7 × 104 per well in 48-well plates. The experimental groups consisted of control siRNA, siRNA-LOXL2, control, and inhibitor (1 μM, seeded at 48 h) Subsequently, the cells underwent SA-β-Gal staining (Beyotime, Beijing, China) following the guidelines provided by the manufacturer. Senescent cells were characterized by blue staining.
3.10 Wound healing assay
Cells transfected or treated with 1 μM inhibitor were quantified and seeded in 6-well plates. Straight lines were drawn on the plate surface using a 200 μL pipette tip. Following two rounds of PBS cleaning, serum-free medium was introduced for subsequent cell culture. The experimental groups consisted of control siRNA, siRNA-LOXL2, control, and inhibitor (1 µM). Subsequently, photographs were captured after rinsing the wells with PBS at the 24-h mark, and the migration areas were subsequently analyzed using ImageJ software.
4 Discussion
Cancer is the major burden and fatal disease alongside population growth and aging. With the development of new innovative agents and treatment modalities in oncology, clinical practice has greatly benefited over the last few years [41]. The utilization of genetic testing and targeted therapy in a clinical setting demonstrates that the molecular characterization of oncogenes serves as the initial step in the ongoing investigation of underlying mechanisms and intervention strategies [42, 43]. A comprehensive analysis across different types of cancer may uncover shared characteristics and provide therapeutic strategies for cancers with similar features, and pan-cancer analysis has the potential to unveil a universal gene signature across diverse cancer types.
LOXL2 has been identified as a carcinoma promotor since 2003 [44], for its underlying roles in maturation and remodeling of ECM as an extracellular enzyme. Subsequently, a multitude of studies have elucidated the association between elevated expression of LOXL2 and diminished overall survival rates, as well as exacerbated clinicopathological attributes of tumors [12, 45, 46]. Furthermore, new roles of LOXL2 have been identified in the cytoplasm, perinuclear region, and nucleus, most of which are linked to its amine oxidase activity [18, 47,48,49]. In the extracellular milieu, the presence of LOXL2 was found to enhance stromal stiffness and facilitate the promotion of adjacent fibroblasts, thereby establishing a supportive niche within the cancer microenvironment [50,51,52]. This phenomenon is also viable in the case of distant metastasis, as the secretion of LOXL2 upon reaching distant organs can stimulate the restructuring of the ECM and initiate the development of premetastatic niches [53,54,55]. However, the efficacy of LOXL2-targeted therapy was found to be suboptimal, as evidenced by the unsatisfactory patient outcomes observed in multiple randomized phase II clinical trials involving the humanized LOXL2 antibody Simtuzumab [22, 23], and more targeted agents and combinational regimens are needed to enter clinical studies [20]. The current understanding of molecular biology in LOXL2 is not yet complete. Notwithstanding the significance of regulation in the context of cancer, it appears that this is the inaugural pan-cancer analysis of LOXL2 conducted thus far. By combining data from various types of cancer, we extensively investigated the panorama of LOXL2 in pan-cancer and elucidated its interconnected role in tumor progression, mutational profile, immune response, and cellular senescence.
Our initial comprehensive examination of LOXL2 across various cancer types revealed its prevalent overexpression and association with unfavorable clinical results. Depending on this research, LOXL2 was markedly increased in numerous tumors when analyzing paired (16 types of tumors) and unpaired (27 types of tumors) samples at the mRNA level and 10 tumor types of unpaired protein samples, which aligns with previous reports [20, 21], especially for liver, lung, breast, kidney, uterine, head and neck cancers. Cox and Kaplan–Meier curve analysis indicated a significant association between elevated LOXL2 expression and an increased incidence of poor OS (15 tumor types), DSS (17 tumor types), DPI (13 tumor types), and PFI (24 tumor types). The findings across multiple types of cancer are in line with previous results obtained from studies focused on a singular type of cancer, and indicating LOXL2 as a possible predictive biomarker for multiple cancers [12,13,14,15,16, 49]. Besides, the secreted characteristics of the LOXL2 protein determine its promising role in cancer screening and prognostic evaluation, indicating that LOXL2 can be considered a predictive and prognostic indicator for specific types of malignancies. More samples and high-quality clinical experiments are needed to be conducted.
The occurrence and advancement of cancer result from a sequence of genetic alteration and changes in oncogene molecular feature that are of great significance to guide therapy and monitor therapeutic effects [26]. We examined the genetic alteration of LOXL2 in various types of cancer. The main genetic alteration type of LOXL2 in pan-cancer is “Deep Deletion” and UCEC, SKCM, and COAD had the highest mutated frequencies of 5.9%, 4.6%, and 3.5%, respectively. LOXL2 gene mutations were all “Deep Deletion” in USC, DLBC, CHOL, and UM, but were all “Amplification” in PCPG and THYM. However, a report revealed that LOXL2 overexpression is more common than mutation [20], follow-up studies are required for specific cancer types. Variations in LOXL2 expression led to passenger mutations in APOB, USH2A, VPS13B, and KMT2D genes. Mutations of APOB, USH2A, and KMT2D were reported to be associated with poor prognosis in several cancer types, and could be considered targets for combination therapy [56,57,58]. Patients with LOXL2 alternations of SNV had a worse prognosis than those with no alterations in BRCA and CESC, while the CNV is associated with a worse prognosis in KIRP, UCEC, and BRCA. The presence of LOXL2 mutations has a substantial effect on the progression of tumors, resulting in tumor heterogeneity. Identifying specific genetic mutations and molecular profiles prior to targeted therapy will maximize cancer patients' benefits [59].
It is less known how LOXL2 affects the tumor immune milieu. We conducted research to explore the correlations between the expression of LOXL2 and the immune environment in various types of cancer. The correlation between immune cell infiltration and LOXL2 expression was examined in 38 types of tumors using the TIMER method. The xCELL method determined the association between the majority of the 64 immune cell subtypes and LOXL2 expression in different tumor types. LOXL2 expression was positively associated with the number of CD4 + T and CD8 + T cells, indicating that T cell infiltration could be promoted by LOXL2. Different single tumor studies showed disparate effects of LOXL2 on immune cell infiltration [60, 61]. A reciprocal influence exists between LOXL2 and immune cells, a more in-depth study may be required to clarify a specific mechanism. LOXL2 expression was positively correlated with genes in major checkpoint pathways, like VEGFC, CD276, TGFB1, and IL10, which may result in an immunosuppression microenvironment. Notably, LOXL2 expression exhibited a strong correlation with fibroblast, pericyte, and endothelial cells in various malignancies. LOXL2 protein as an ECM modifying factor, may alter TME by affecting stromal cells, because of its regulatory roles in vasculogenic mimicry and cell adhesion. Therefore, immunotherapy in cancer patients with LOXL2 overexpression requires more factors for consideration in personalized medicine.
GeneMANIA examined the impact of LOXL2 on the downstream signaling pathways. Multiple cellular senescence pathways (including DNA Repair, Telomere Maintenance, and Metabolic pathway) were significantly enriched. In the above bioinformatic research, LOXL2 was proven to have a significant association with the advancement of tumors and cellular senescence, subsequently, we performed CCK-8, wound healing, and SA-β-gal staining assays. Down-regulation of LOXL2 expression promoted cellular senescence and limited the proliferation and migration abilities of PC9 and HCC-LM3 cells. LOXL2 was identified as being associated with tumor progression and cellular senescence. LOXL2 has been reported in the process of senescent morphogenesis in fibroblast and epithelial cells, leading to skeletal muscle fibrosis and lung diseases [24, 25], but is rarely analyzed in tumor research. LOXL2-induced cellular senescence was reported for the first time in pan-cancer within the present study, which may aid in related research and the development of new cancer therapies.
5 Limitation
Some limitations warrant further deliberation. Our study data is based on public databases, which may be constrained due to inadequate sample size for specific studies. By far, we cannot definitively state that the strong connections we observed in pan-cancer are equally prevalent in every type of cancer. The effects of LOXL2 on tumor infiltrated immune cells and oncogenic senescence have not yet been elucidated, it eagerly anticipates future exploration of the mechanisms underlying tumor advancement and the transition to a senescent state. The development of therapeutically viable drugs targeting LOXL2 poses challenges due to the presence of various active mechanisms and barriers associated with inhibiting specific domains. Additional research is necessary to illustrate the underlying mechanism of senescence and further elicit tumorigenesis, thereby optimizing corresponding therapeutic interventions.
6 Conclusion
Our initial comprehensive analysis of LOXL2 across diverse cancer types revealed its prevalent overexpression and association with unfavorable clinical results, subsequent genetic alterations, and immunological context. Furthermore, the roles of LOXL2 in tumor progression and cellular senescence were partly verified in vitro. Overall, LOXL2 could be a potential prognostic and diagnostic biomarker, and afford new thought for tumor personalized therapy.
Data availability
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Abbreviations
- ACC:
-
Adrenocortical carcinoma
- BLCA:
-
Bladder urothelial carcinoma
- BRCA:
-
Breast invasive carcinoma
- CESC:
-
Cervical squamous cell carcinoma and endocervical adenocarcinoma
- CHOL:
-
Cholangiocarcinoma
- COAD:
-
Colon adenocarcinoma
- DLBC:
-
Lymphoid neoplasm diffuse large B-cell lymphoma
- ESCA:
-
Esophageal carcinoma
- GBM:
-
Glioblastoma multiforme
- HNSC:
-
Head and neck squamous cell carcinoma
- KICH:
-
Kidney Chromophobe
- KIPAN:
-
Pan-kidney cohort (KICH + KIRC + KIRP)
- KIRC:
-
Kidney renal clear cell carcinoma
- KIRP:
-
Kidney renal papillary cell carcinoma
- LAML:
-
Acute myeloid leukemia
- LGG:
-
Brain lower grade glioma
- LIHC:
-
Liver hepatocellular carcinoma
- LUAD:
-
Lung adenocarcinoma
- LUSC:
-
Lung squamous cell carcinoma
- MESO:
-
Mesothelioma
- OV:
-
Ovarian serous cystadenocarcinoma
- PAAD:
-
Pancreatic adenocarcinoma
- PCPG:
-
Pheochromocytoma and paraganglioma
- PRAD:
-
Prostate adenocarcinoma
- READ:
-
Rectum adenocarcinoma
- SARC:
-
Sarcoma
- SKCM:
-
Skin cutaneous melanoma
- STAD:
-
Stomach adenocarcinoma
- TGCT:
-
Testicular germ cell tumor
- THCA:
-
Thyroid carcinoma
- THYM:
-
Thymoma
- UCEC:
-
Uterine corpus endometrial carcinoma
- UCS:
-
Uterine carcinosarcoma
- UVM:
-
Uveal melanoma
References
Soerjomataram I, Bray F. Planning for tomorrow: global cancer incidence and the role of prevention 2020–2070. Nat Rev Clin Oncol. 2021;18:663–72. https://doi.org/10.1038/s41571-021-00514-z.
Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer Statistics, 2021. CA Cancer J Clin. 2021;71:7–33. https://doi.org/10.3322/caac.21654.
Robert L, Ribas A, Hu-Lieskovan S. Combining targeted therapy with immunotherapy. Can 1+1 equal more than 2? Semin Immunol. 2016;28:73–80. https://doi.org/10.1016/j.smim.2016.01.001.
Junttila MR, de Sauvage FJ. Influence of tumour micro-environment heterogeneity on therapeutic response. Nature. 2013;501:346–54. https://doi.org/10.1038/nature12626.
Fane M, Harris L, Smith AG, Piper M. Nuclear factor one transcription factors as epigenetic regulators in cancer. Int J Cancer. 2017;140:2634–41. https://doi.org/10.1002/ijc.30603.
Huang RH, Wang LX, He J, Gao W. Application and prospects of single cell sequencing in tumors. Biomark Res. 2021;9:88. https://doi.org/10.1186/s40364-021-00336-2.
Theocharis AD, Skandalis SS, Gialeli C, Karamanos NK. Extracellular matrix structure. Adv Drug Deliv Rev. 2016;97:4–27. https://doi.org/10.1016/j.addr.2015.11.001.
Zhang Q, Wang W, Zhou Q, et al. Roles of circRNAs in the tumour microenvironment. Mol Cancer. 2020;19:14. https://doi.org/10.1186/s12943-019-1125-9.
Kim YH, Park TJ. Cellular senescence in cancer. BMB Rep. 2019;52:42–6. https://doi.org/10.5483/BMBRep.2019.52.1.295.
Serrano M. Final act of senescence. Nature. 2011;479:481–2. https://doi.org/10.1038/479481a.
Wen B, Xu L-Y, Li E-M. LOXL2 in cancer: regulation, downstream effectors and novel roles. Biochim Biophys Acta Rev Cancer. 2020;1874: 188435. https://doi.org/10.1016/j.bbcan.2020.188435.
Deshpande H (2023) Levoleucovorin inhibits LOXL2 (lysyl oxidase like-2) to control breast cancer proliferation: a repurposing approach. J Biomol Struct Dyn 1–10. https://doi.org/10.1080/07391102.2023.2224894
Zhao N, Chen C, Guo Y, et al. LOXL2 serves as a prognostic biomarker for hepatocellular carcinoma by mediating immune infiltration and vasculogenic mimicry. Dig Liver Dis. 2023;55:661–72. https://doi.org/10.1016/j.dld.2022.09.003.
Zhan P, Lv X-J, Ji Y-N, et al. Increased lysyl oxidase-like 2 associates with a poor prognosis in non-small cell lung cancer. Clin Respir J. 2018;12:712–20. https://doi.org/10.1111/crj.12584.
Peng T, Lin S, Meng Y, et al. LOXL2LOXL2 small molecule inhibitor restrains malignant transformation of cervical cancer cells by repressing -induced epithelial-mesenchymal transition (EMT). Cell Cycle. 2022;21:1827–41. https://doi.org/10.1080/15384101.2022.2073047.
Torres S, Garcia-Palmero I, Herrera M, et al. LOXL2 is highly expressed in cancer-associated fibroblasts and associates to poor colon cancer survival. Clin Cancer Res. 2015;21:4892–902. https://doi.org/10.1158/1078-0432.CCR-14-3096.
Jiao J-W, Zhan X-H, Wang J-J, et al. LOXL2-dependent deacetylation of aldolase A induces metabolic reprogramming and tumor progression. Redox Biol. 2022;57: 102496. https://doi.org/10.1016/j.redox.2022.102496.
Umana-Diaz C, Pichol-Thievend C, Marchand MF, et al. Scavenger Receptor Cysteine-Rich domains of Lysyl Oxidase-Like2 regulate endothelial ECM and angiogenesis through non-catalytic scaffolding mechanisms. Matrix Biol. 2020;88:33–52. https://doi.org/10.1016/j.matbio.2019.11.003.
Zheng G-L, Liu Y-L, Yan Z-X, et al. Elevated LOXL2 expression by LINC01347/miR-328-5p axis contributes to 5-FU chemotherapy resistance of colorectal cancer. Am J Cancer Res. 2021;11:1572–85.
Cano A, Eraso P, Mazón MJ, Portillo F. LOXL2 in Cancer: A Two-Decade Perspective. Int J Mol Sci. 2023;24:14405. https://doi.org/10.3390/ijms241814405.
Liburkin-Dan T, Toledano S, Neufeld G. Lysyl Oxidase Family Enzymes and Their Role in Tumor Progression. Int J Mol Sci. 2022;23:6249. https://doi.org/10.3390/ijms23116249.
Raghu G, Brown KK, Collard HR, et al. Efficacy of simtuzumab versus placebo in patients with idiopathic pulmonary fibrosis: a randomised, double-blind, controlled, phase 2 trial. Lancet Respir Med. 2017;5:22–32. https://doi.org/10.1016/S2213-2600(16)30421-0.
Harrison SA, Abdelmalek MF, Caldwell S, et al. Simtuzumab is ineffective for patients with bridging fibrosis or compensated cirrhosis caused by nonalcoholic steatohepatitis. Gastroenterology. 2018;155:1140–53. https://doi.org/10.1053/j.gastro.2018.07.006.
Pascal T, Debacq-Chainiaux F, Chretien A, et al. Comparison of replicative senescence and stress-induced premature senescence combining differential display and low-density DNA arrays. FEBS Lett. 2005. https://doi.org/10.1016/j.febslet.2005.05.056.
Müller K-C, Welker L, Paasch K, et al. Lung fibroblasts from patients with emphysema show markers of senescence in vitro. Respir Res. 2006. https://doi.org/10.1186/1465-9921-7-32.
Xiao Y, Yu D. Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther. 2021;221: 107753. https://doi.org/10.1016/j.pharmthera.2020.107753.
Gu D, Ao X, Yang Y, et al. Soluble immune checkpoints in cancer: production, function and biological significance. J Immunother Cancer. 2018;6:132. https://doi.org/10.1186/s40425-018-0449-0.
Thorsson V, Gibbs DL, Brown SD, et al. The Immune Landscape of Cancer. Immunity. 2018;48:812-830.e14. https://doi.org/10.1016/j.immuni.2018.03.023.
Chandrashekar DS, Bashel B, Balasubramanya SAH, et al. UALCAN: A Portal for Facilitating Tumor Subgroup Gene Expression and Survival Analyses. Neoplasia. 2017;19:649–58. https://doi.org/10.1016/j.neo.2017.05.002.
Navarro Gonzalez J, Zweig AS, Speir ML, et al. The UCSC Genome Browser database: 2021 update. Nucleic Acids Res. 2021;49:D1046–57. https://doi.org/10.1093/nar/gkaa1070.
Gustavsson EK, Zhang D, Reynolds RH, et al. (2022) ggtranscript: an R package for the visualization and interpretation of transcript isoforms using ggplot2. Bioinformatics. 2022;38(15):3844–6. https://doi.org/10.1093/bioinformatics/btac409.
Beroukhim R, Mermel CH, Porter D, et al. The landscape of somatic copy-number alteration across human cancers. Nature. 2010;463:899–905. https://doi.org/10.1038/nature08822.
Mayakonda A, Lin D-C, Assenov Y, et al. Maftools: efficient and comprehensive analysis of somatic variants in cancer. Genome Res. 2018;28:1747–56. https://doi.org/10.1101/gr.239244.118.
Liu C-J, Hu F-F, Xie G-Y, et al. GSCA: an integrated platform for gene set cancer analysis at genomic, pharmacogenomic and immunogenomic levels. Brief Bioinform. 2023;24:558. https://doi.org/10.1093/bib/bbac558.
Li T, Fan J, Wang B, et al. TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 2017;77:e108–10. https://doi.org/10.1158/0008-5472.CAN-17-0307.
Zeng D, Ye Z, Shen R, et al. IOBR: multi-omics immuno-oncology biological research to decode tumor microenvironment and signatures. Front Immunol. 2021;12: 687975. https://doi.org/10.3389/fimmu.2021.687975.
Tang Z, Kang B, Li C, et al. GEPIA2: an enhanced web server for large-scale expression profiling and interactive analysis. Nucleic Acids Res. 2019;47:W556–60. https://doi.org/10.1093/nar/gkz430.
Yu G, Wang L-G, Han Y, He Q-Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS. 2012;16:284–7. https://doi.org/10.1089/omi.2011.0118.
Hutchinson JH, Rowbottom MW, Lonergan D, et al. Small Molecule Lysyl Oxidase-like 2 (LOXL2) Inhibitors: The Identification of an Inhibitor Selective for LOXL2 over LOX. ACS Med Chem Lett. 2017;8:423–7. https://doi.org/10.1021/acsmedchemlett.7b00014.
Chang J, Lucas MC, Leonte LE, et al. Pre-clinical evaluation of small molecule LOXL2 inhibitors in breast cancer. Oncotarget. 2017;8:26066–78. https://doi.org/10.18632/oncotarget.15257.
Whitcomb DC. What is personalized medicine and what should it replace? Nat Rev Gastroenterol Hepatol. 2012;9:418–24. https://doi.org/10.1038/nrgastro.2012.100.
Michiels S, Ternès N, Rotolo F. Statistical controversies in clinical research: prognostic gene signatures are not (yet) useful in clinical practice. Ann Oncol. 2016;27:2160–7. https://doi.org/10.1093/annonc/mdw307.
Chen F, Wendl MC, Wyczalkowski MA, et al. Moving pan-cancer studies from basic research toward the clinic. Nat Cancer. 2021;2:879–90. https://doi.org/10.1038/s43018-021-00250-4.
Akiri G, Sabo E, Dafni H, et al. Lysyl oxidase-related protein-1 promotes tumor fibrosis and tumor progression in vivo. Cancer Res. 2003;63:1657–66.
Wu L, Zhu Y. The function and mechanisms of action of LOXL2 in cancer (Review). Int J Mol Med. 2015;36:1200–4. https://doi.org/10.3892/ijmm.2015.2337.
Zhang X, Huang J, You F, et al. Prognostic and clinicopathological significance of LOXL2 in cancers: A systematic review and meta-analysis. J Cell Physiol. 2019;234:21369–79. https://doi.org/10.1002/jcp.28746.
Zhan X-H, Jiao J-W, Zhang H-F, et al. LOXL2 Upregulates Phosphorylation of Ezrin to Promote Cytoskeletal Reorganization and Tumor Cell Invasion. Cancer Res. 2019;79:4951–64. https://doi.org/10.1158/0008-5472.CAN-19-0860.
Almacellas-Rabaiget O, Monaco P, Huertas-Martinez J, et al. LOXL2 promotes oncogenic progression in alveolar rhabdomyosarcoma independently of its catalytic activity. Cancer Lett. 2020;23:45.
Lu X, Xin DE, Du JK, et al. Loss of LOXL2 Promotes Uterine Hypertrophy and Tumor Progression by Enhancing H3K36ac-Dependent Gene Expression. Cancer Res. 2022;474:1–14. https://doi.org/10.1016/j.canlet.2019.12.040.
Barker HE, Bird D, Lang G, Erler JT. Tumor-secreted LOXL2 activates fibroblasts through FAK signaling. Mol Cancer Res. 2013;11:1425–36. https://doi.org/10.1158/1541-7786.MCR-13-0033-T.
Wang C, Xu S, Tian Y, et al. Lysyl Oxidase-Like Protein 2 Promotes Tumor Lymphangiogenesis and Lymph Node Metastasis in Breast Cancer. Neoplasia. 2019;21:413–27. https://doi.org/10.1016/j.neo.2019.03.003.
Mahjour F, Dambal V, Shrestha N, et al. Mechanism for oral tumor cell lysyl oxidase like-2 in cancer development: synergy with PDGF-AB. Oncogenesis. 2019;8:34. https://doi.org/10.1038/s41389-019-0144-0.
Wong CC-L, Tse AP-W, Huang Y-P, et al. Lysyl oxidase-like 2 is critical to tumor microenvironment and metastatic niche formation in hepatocellular carcinoma. Hepatology. 2014;60:1645–58. https://doi.org/10.1002/hep.27320.
Canesin G, Cuevas EP, Santos V, et al. Lysyl oxidase-like 2 (LOXL2) and E47 EMT factor: novel partners in E-cadherin repression and early metastasis colonization. Oncogene Oncogene. 2015;34:951–64. https://doi.org/10.1038/onc.2014.23.
Zhu G, Wang L, Meng W, et al. LOXL2-enriched small extracellular vesicles mediate hypoxia-induced premetastatic niche and indicates poor outcome of head and neck cancer. Theranostics. 2021. https://doi.org/10.7150/thno.62455.
Lee G, Jeong YS, Kim DW, et al. Clinical significance of APOB inactivation in hepatocellular carcinoma. Exp Mol Med. 2018;50:1–12. https://doi.org/10.1038/s12276-018-0174-2.
Sun Y, Li L, Yao W, et al. USH2A Mutation is Associated With Tumor Mutation Burden and Antitumor Immunity in Patients With Colon Adenocarcinoma. Front Genet. 2021;12: 762160. https://doi.org/10.3389/fgene.2021.762160.
Morcillo-Garcia S, Noblejas-Lopez MDM, Nieto-Jimenez C, et al. Genetic mutational status of genes regulating epigenetics: Role of the histone methyltransferase KMT2D in triple negative breast tumors. PLoS ONE. 2019;14: e0209134. https://doi.org/10.1371/journal.pone.0209134.
Jin J, Wu X, Yin J, et al. Identification of Genetic Mutations in Cancer: Challenge and Opportunity in the New Era of Targeted Therapy. Front Oncol. 2019;9:263. https://doi.org/10.3389/fonc.2019.00263.
Peng DH, Rodriguez BL, Diao L, et al. Collagen promotes anti-PD-1/PD-L1 resistance in cancer through LAIR1-dependent CD8 T cell exhaustion. Nat Commun. 2020;11:4520. https://doi.org/10.1038/s41467-020-18298-8.
Zhang X, Wu X, Sun Y, et al. TRIM44 regulates tumor immunity in gastric cancer through LOXL2-dependent extracellular matrix remodeling. Cell Oncol. 2023;46:423–35. https://doi.org/10.1007/s13402-022-00759-5.
Funding
This work was supported by National Natural Science Foundation of China (Grant Numbers 82141105 and 82173028).
Author information
Authors and Affiliations
Contributions
Material preparation, data collection and analysis were performed by C. Y., S. J. and T. Z. The first draft of the manuscript was written by C. Y. and S. H., J. C. and J. X. made the final revision and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ye, C., Jiang, S., Zeng, T. et al. The role of LOXL2 in tumor progression, immune response and cellular senescence: a comprehensive analysis. Discov Onc 15, 245 (2024). https://doi.org/10.1007/s12672-024-01107-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12672-024-01107-9