Abstract
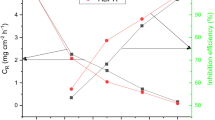
Corrosion protection of mild steel (MS) substrate is critical to prolonging the life of service equipment in refineries and chemical industries. In petroleum refineries, drilling and acidizing pipe work with vast amounts of 15% HCl that induce corrosion of MS pipelines. Here, a novel pentadecyl phenol (originated from cashew nut shell liquid)-functionalized conjugated polypyrrole (MPY) was synthesized and demonstrated for MS corrosion inhibition at a 15% HCl environment. Pentadecyl units provide hydrophobicity and form interdigitating layer structures to cover the metal substrate, which leads to better corrosion mitigation. Electrochemical impedance spectroscopy (EIS) and potentiodynamic polarization (PDP) studies reveal the superior corrosion inhibition performance of MPY on MS under 15% HCl conditions. The corrosion protection efficiency was determined to be 67.93% over MS substrate in the presence of MPY from PDP studies compared with pure MS.
Graphical Abstract









Similar content being viewed by others
References
Papavinasam S, Corrosion control in the oil and gas industry, 1st edn. Elsevier, Amsterdam (2003). https://doi.org/10.1016/C2011-0-04629-X
Rahimi A, Abdouss M, Farhadian A, Guo L, and Neshati J, Ind Eng Chem Res 60 (2021) 11030. https://doi.org/10.1021/acs.iecr.1c01946
Oyekunle D T, Agboola O, and Ayen A O, J Phy Conf Ser 1378 (2019) 032046. https://doi.org/10.1088/1742-6596/1378/3/032046
Guezennec J G, Int Biodeterior Biodegrad 34 (1994) 275. https://doi.org/10.1016/0964-8305(94)90088-4
Subasri R, Shinohara T, and Mori K, Sci Technol Adv Mater 6 (2005) 501. https://doi.org/10.1016/j.stam.2005.01.003
Cecchetto L, Delabouglise D, and Petit J P, Electrochim Acta 52 (2007) 3485. https://doi.org/10.1016/j.electacta.2006.10.009
Selvaraj R, Selvaraj M, and Iyer S V K, Prog Org Coat 64 (2009) 454. https://doi.org/10.1016/j.porgcoat.2008.08.005
Chauhan D S, Ansari K R, Sorour A A, Quraishi M A, Lgaz H, and Salghi R, Int J Biol Macromol 107 (2018) 1747. https://doi.org/10.1016/j.ijbiomac.2017.10.050
Townsend H E, Corros Sci 57 (2001) 497. https://doi.org/10.5006/1.3290374
Saxena N, Kumar S, and Mathur S P, Chem Eng Commun. 196 (2009) 1451. https://doi.org/10.1080/00986440902938881
Abdallah M, El-Etre A Y, Soliman M G, and Mabrouk E M, Anti-Corros Method M 53 (2006) 118. https://doi.org/10.1108/00035590610650820
Sabirneeza A A F, Geethanjali R, and Subhashini S, Chem Eng Commun 202 (2015) 232. https://doi.org/10.1080/00986445.2014.934448
Farhadian A, Varfolomeev M A, Shaabani A, Nasiri S, Vakhitov I, Zaripova Y F, Yarkovoi V V, and Sukhov A V, Carbohydr Polym 236 (2020) 116035. https://doi.org/10.1016/j.carbpol.2020.116035
Umoren S A, and Solomon M M, Corrosion inhibitors in oil and gas industry, Wiley, London (2020), p 303.
Spinks M, Dominis A J, and Wallace G G, J.Solid State Electrochem 6 (2002) 85. https://doi.org/10.1007/s100080100211
Kong P, Feng H, Chen N, Lu Y, and Wang P, RSC Adv 9 (2019) 9211. https://doi.org/10.1039/c9ra00029a
Biswas A, Pal S, and Udayabhanu G, Appl Surf Sci 353 (2015) 173. https://doi.org/10.1016/j.apsusc.2015.06.128
Yeganeh M, Saremi M, and Rezaeyan H, Prog Org Coat 77 (2014) 1428. https://doi.org/10.1016/j.porgcoat.2014.05.007
Wang X, and Xing J, Int J Electrochem Sci 15 (2020) 1606. https://doi.org/10.20964/2020.02.46
Chen B, Xu G, Wang L, Zhang C, Li C, Ju G, and Feng H, Res Sq (2021). https://doi.org/10.21203/rs.3.rs-958811/v1
Senthilkumar T, and Asha S K, Macromolecules 46 (2013) 2159. https://doi.org/10.1021/ma4000946
Brezoi D Ş, J Sci arts 1 (2010) 53.
Tsai P Y, Chen T E, and Lee Y L, Coatings 8 (2018) 250. https://doi.org/10.3390/coatings8070250
Prakashaiah B G, Kumara D V, Pandith A A, and Shetty A N, Corros Sci. 136 (2018) 326. https://doi.org/10.1016/j.corsci.2018.03.021
Kesari P, and Udayabhanu G, Ain Shams Eng J 14 (2023) 101920. https://doi.org/10.1016/j.asej.2022.101920
Prakashaiah B G, Shetty A N, and Amitharani B E, Surf Eng Appl Electrochem 54 (2018) 286. https://doi.org/10.3103/S1068375518030109
Acknowledgements
The authors thank the Analytical Sciences Division of CSIR-IIP for analytical support. Authors Sachin and Nishtha Arora thank UGC for the research fellowship.
Funding
This work was supported by the CSIR-IIP project No. OLP-1183 for the research funding.
Author information
Authors and Affiliations
Contributions
TS contributed to conceptualization and supervision; S and TS contributed to methodology; S, NA, BGP, and TS contributed to investigation; S, RCS, and TS contributed to writing and original draft preparation; RCS, BGP, SKG, and TS contributed to writing, reviewing, and editing; and RCS and TS contributed to visualization. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dua, S., Prakashaiah, B.G., Arora, N. et al. Functionalized Polypyrrole for Mild Steel Corrosion Protection in 15% HCl. Trans Indian Inst Met 77, 1313–1321 (2024). https://doi.org/10.1007/s12666-023-03107-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-023-03107-9