Abstract
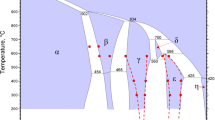
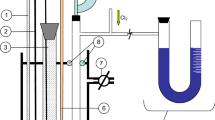
The diffusion behaviour of Al and Cu in Cu(Al) solid solution phase was investigated in the temperature range 1023–1223 K, by employing single-phase diffusion couples of Cu/Cu–10 at% Al. The intrinsic diffusion coefficient of Al was found to be higher than that of Cu in the solid solution phase. The thermodynamic factor for the Cu(Al) phase was calculated as a function of composition and temperature, assuming a regular solution behaviour. The tracer diffusion coefficients and mobilities of Al and Cu were also evaluated. The activation energies for intrinsic diffusion were 127.2 kJ mol−1 and 146.7 kJ mol−1, and those for tracer diffusion were 133.2 kJ mol−1 and 152.6 kJ mol−1 for Al and Cu, respectively. The correlation factor for Cu remained almost constant, while that of Al was found to have strong temperature dependence. The values of the correlation factors indicated a comparatively low concentration of vacancies involved in the diffusion process and the jump ratio of Al was an order of magnitude higher than that of the Cu.









Similar content being viewed by others
References
Zobac O, Kroupa A, Zemanova A, and Richter K W, Metall Mater Trans A 50A (2019) 3805.
Massalski T B, Okamoto H, Subramanian P R, and Kacprzak L, Binary Alloy Phase Diagrams, 2nd edn., ASM International, Materials Park (1990).
Adorno A T, Guerreiro M R, and Benedetti A V, J Alloys Compd 315 (2001)150.
Gaudig W and Warlimont H, Acta Metall 26 (1978) 709.
Popplewell J M and Crane J, Metall Trans B 2 (1971) 3411.
Trieb L and Vieth G, Acta Mater 26 (1978) 185.
Varschavski A, Thermochim Acta 203 (1992) 391.
Rhines F N and Mehl R F, Trans. AIME 128 (1938) 185.
Oikawa H, Obara T, and Karashima S, Metall Trans 1 (1970) 2969.
Matsuno N and Oikawa H, Metall Trans A 6A (1975) 2191.
Laik A, Bhanumurthy K, and Kale G B, Defect Diffusion Forum 279 (2008) 63.
Liu D, Zhang L, Du Y, Xu H, Liu S, and Liu L, Calphad 33 (2009) 761.
Boltzmann L, Annal Phys, 289 (1894) 959.
Matano C, Jpn J Phys 8 (1933) 109.
Darken L S, Trans. AIME 175 (1948) 184.
Philibert J, Atom Movements: Diffusion and Mass Transport in Solids, Les Editions de Physique, Les Ulis (1991).
Manning J R, Diffusion Kinetics for Atoms in Crystals, D. Van Nostrand Company, Princeton (1968).
Shewmon P G, Diffusion in Solids, 2nd edn., The Minerals, Metals and Materials Society, Pennsylvania (1989).
Miettinen J, Metall. Mater. Trans A 33A (2002) 1639.
Wilder T C, Trans. AIME 233 (1965)1202.
Kucera J and Million B, Metall Trans 1 (1970), 2599.
Franke P and Deickermann R, J Appl Phys 70 (1991) 787.
Laik A, Bhanumurthy K, and Kale G B, J Alloy Compd 372 (2004) 176.
Acknowledgements
The authors acknowledge the contributions of Dr G. B. Kale, former colleague at Materials Science Division, BARC, who initiated the present investigation. They are thankful to Dr V. Kain, Director, Materials Group for his constant support. The authors would also like to express their respectful gratitude to Late Dr Srikumar Banerjee, former Chairman, Atomic Energy Commission and Secretary, Department of Atomic Energy for his motivation and encouragement towards the programme on fundamental studies on diffusion in metals and alloys.
Funding
Funding was provided by Department of Atomic Energy, Government of India.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Laik, A., Tewari, R. Intrinsic Diffusion, Mobility and Correlation Effect in Cu(Al) Solid Solution. Trans Indian Inst Met 75, 895–905 (2022). https://doi.org/10.1007/s12666-021-02515-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12666-021-02515-z