Abstract
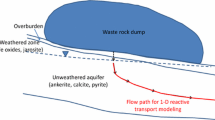
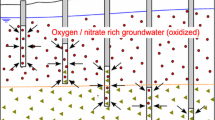
The present study is aimed to formulate a numerical model to simulate hydrogeochemical reactions controlling pyrite oxidation in unsaturated aquifers under non-uniform flow conditions. A coupled flow and multi-component transport model is solved using a finite difference scheme to simulate the concentration distribution of oxygen (O2), ferrous (Fe2+), ferric (Fe3+), sulphate (SO42−) and hydrogen (H+) ions. An attempt is made to quantify the transport characteristics of pyrite oxidation ions by analysing the spatial moments of concentration profiles. The simulation results suggest that the variable flow rate in the vertical direction significantly enhances the pyrite oxidation near the ground surface. Peak concentration of Fe2+, Fe3+, and SO42− near the ground surface is found to be approximately 2–3 times higher in the case of variable flow. The pH level near the ground surface drops considerably when variable flow conditions exist in the unsaturated zone. The variation in flow parameters such as porosity, and water saturation and reaction parameters such as oxygen diffusion rate and the oxygen consumption rate are found to affect the mass and mobility of Fe2+, Fe3+, and SO42− considerably. Although there has been substantial research on hydrogeochemical events associated with pyrite oxidation in the past, there has been very less focus on the transport qualities of the ions produced in groundwater. The mobility assessment of pyrite oxidation ions in aquifer presented in the study is beneficial to understand the groundwater quality risk associated with mining and to plan and implement effective remediation strategies for the restoration of affected aquifers.









Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Abdelghani FB, Aubertin M, Simon R, Therrien R (2015) Numerical simulations of water flow and contaminants transport near mining wastes disposed in a fractured rock mass. Int J Min Sci Technol 25:37–45. https://doi.org/10.1016/j.ijmst.2014.11.003
Ata A, Soner K (2006) Acid mine drainage (AMD): causes, treatment and case studies. J Clean Prod 14(12–13):1139–1145. https://doi.org/10.1016/j.jclepro.2004.09.006
Dold B (2014) Evolution of acid mine drainage formation in sulphidic mine tailings. Minerals 4:621–641. https://doi.org/10.3390/min4030621
Dos Santos EC, de Mendonça Silva JC, Duarte HA (2016) Pyrite oxidation mechanism by oxygen in aqueous medium. J Phys Chem C 120:2760–2768. https://doi.org/10.1021/acs.jpcc.5b10949
Doulati AF, Jodieri SB, Moradzadeh A, Shafaei SZ, Kakaei R (2011) Geochemical characterisation of pyrite oxidation and environmental problems related to release and transport of metals from a coal washing low grade waste dump, Shahrood, northeast Iran. Environ Monit Assess 181:41–55. https://doi.org/10.1007/s10661-011-1904-2
Doulati AF, Jannesar MS, Ziaedin SS et al (2014) A numerical multi-component reactive model for pyrite oxidation and pollutant transportation in a pyritic, carbonate-rich coal waste pile in Northern Iran. Mine Water Environ 33:121–132. https://doi.org/10.1007/s10230-014-0275-7
Dragon K, Marciniak M (2010) Chemical composition of groundwater and surface water in the Arctic environment (Petuniabukta region, centralSpitsbergen). J Hydrol 386:160–172. https://doi.org/10.2136/sssaj2017.02.0058
Elberling B, Nicholson RV, Scharer JM (1994) A combined kinetic and diffusion model for pyrite oxidation in tailings: a change in controls with time. J Hydrol 157(1):47–60
Farthing MW, Ogden FL (2017) Numerical solution of Richards’ equation: a review of advances and challenges. Soil Sci Soc Am J 81:1257–1269. https://doi.org/10.1016/j.jhydrol.2010.03.017
Karikari-Yeboah O, Skinner W, Addai-Mensah J (2019) Anaerobic pyrite oxidation in a naturally occurring pyrite-rich sediment under preload surcharge. Environ Monit Assess 191:216. https://doi.org/10.1007/s10661-019-7289-3
Kariman N, Johnston SG, Burton ED (2017) Acidity generation accompanying iron and sulfur transformations during drought simulation of freshwater re-flooded acid sulfate soils. Geoderma 285:117–131. https://doi.org/10.1016/j.geoderma.2016.09.030
Lefebvre R, Hockley D, Smolensky J, Lamonta A (2001) Multiphase transfer processes in waste rock piles producing acid mine drainage: 2. Applications of numerical simulation. J Contam Hydrol 52(1–4):165–186. https://doi.org/10.1016/S0169-7722(01)00157-7
Liang Z, Ting R, Ningbo W (2017) Groundwater impact of open cut coal mine and an assessment methodology: a case study in NSW. Int J Min Sci Technol 27(5):861–866. https://doi.org/10.1016/j.ijmst.2017.07.008
Mahmoud A, Mohamed S, Amal A, Shrouk MA, Elmontser MS (2019) Hydrogeochemical processes and evaluation of groundwater aquifer at Sohag city, Egypt. Sci Afr 6:e00196. https://doi.org/10.1016/j.sciaf.2019.e00196
Molson JW, Fala O, Aubertin M, Bussière B (2005) Numerical simulations of pyrite oxidation and acid mine drainage in unsaturated waste rock piles. J Contam Hydrol 78(4):343–371. https://doi.org/10.1016/j.jconhyd.2005.06.005
Molson J, Aubertin M, Bussière B (2012) Reactive transport modelling of acid mine drainage within discretely fractured porous media: plume evolution from a surface source zone. Environ Model Soft 38:259270. https://doi.org/10.1016/j.apgeochem.2015.02.008
Nordstrom D, Blowes D, Ptacek C (2015) Hydrogeochemistry and microbiology of mine drainage: an update. Appl Geochem 57:3–16. https://doi.org/10.1016/j.apgeochem.2015.02.008
Phoon KK, Tan TS, Chong PC (2007) Numerical simulation of Richards equation in partially saturated porous media: under-relaxation and mass balance. Geotech Geol Eng 25:525–541. https://doi.org/10.1007/s10706-007-9126-7
Pruess K, Oldenburg C, Moridis G (2012) TOUGH2 User’s Guide, Version 2.1, Report LBNL-43134. Lawrence Berkeley National Laboratory, Berkeley
Ramasamy M, Power C (2019) Evolution of acid mine drainage from a coal waste rock pile reclaimed with a simple soil cover. Hydrology 6(4):83. https://doi.org/10.3390/hydrology6040083
Shahhoseiny M, Doulati AF, Shafaei SZ, Noaparast M, Hamidi D (2012) Geochemical and mineralogical characterization of a pyritic waste pile at the Anjir Tangeh Coal Washing Plant, Zirab, Northern Iran. Mine Water Environ 32(2):84–96
Shingo T, Toshifumi I (2022) The potential threat of mine drainage to groundwater resources. Cur Opin Environ Sci Health 27:100347. https://doi.org/10.1016/j.coesh.2022.100347
Skousen J, Ziemkiewicz P, McDonald L (2018) Acid mine drainage formation, control and treatment: approaches and strategies. Extract Ind Soc 6(1):241–249. https://doi.org/10.1016/j.exis.2018.09.008
Subba Rao N, Vidyasagar G, Surya RP et al (2014) Assessment of hydrogeochemical processes in a coastal region: application of multivariate statistical model. J Geol Soc India 84:494–500. https://doi.org/10.1007/s12594-014-0155-6
Tu Z, Wu Q, He H, Zhou S, Liu J, He H, Liu C, Dang Z, Reinfelder JR (2022) Reduction of acid mine drainage by passivation of pyrite surfaces: a review. Sci Tot Environ 832:155116. https://doi.org/10.1016/j.scitotenv.2022.155116
Van Genuchten MT (1980) A closed-form equation for predicting the hydraulic conductivity of unsaturated soils. Soil Sci Soc Am J 44:892–898. https://doi.org/10.2136/sssaj1980.03615995004400050002x
Verron H, Sterpenich J, Bonnet J, Bourdelle F, Mosser-Ruck R, Lorgeoux C, Randi A, Michau N (2019) Experimental study of pyrite oxidation at 100 °C: implications for deep geological radwaste repository in claystone. Minerals 9(7):427. https://doi.org/10.3390/min9070427
Wang Q, Dong S, Wang H et al (2020) Hydrogeochemical processes and groundwater quality assessment for different aquifers in the Caojiatan coal mine of Ordos Basin, Northwestern China. Environ Earth Sci 79:199. https://doi.org/10.1007/s12665-020-08942-3
Wei Y, Cao X (2022) A COMSOL-PHREEQC coupled python framework for reactive transport modeling in soil and groundwater. Groundwater 60:284–294. https://doi.org/10.1111/gwat.13144
Wei T, Yu Y, Hu Z, Cao Y, Gao Y, Yang Y, Wang X, Wang P (2013) Research progress of acid mine drainage treatment technology in China. Appl Mech Mat 409–410:214–220. https://doi.org/10.4028/www.scientific.net/AMM.409-410.214
Woonghee L, Etienne B, Seongnam A, Ilka W, Vincent P, Seunghak L, Peter KK (2020) Spatiotemporal evolution of iron and sulfate concentrations during riverbank filtration: Field observations and reactive transport modeling. J Contam Hydrol 234:103697. https://doi.org/10.1016/j.jconhyd.2020.103697
Wunderly MD, Blowes DW, Frind EO, Ptacek CJ (1996) Sulfide mineral oxidation and subsequent reactive transport of oxidation products in mine tailings impoundments: a numerical model. Water Resour Res 32(10):3173–3187. https://doi.org/10.1029/96WR02105
Xu Y, Plaza FJ, Liang X, Davis TW, Nichols J, Fu JK, Koranchie-Boah P (2019) A hydro-thermal-geochemical modeling framework to simulate reactive transport in a waste coal area under amended and non-amended conditions. Heliyon 6(1):e02803. https://doi.org/10.1016/j.heliyon.2019.e02803
Molson JW, Aubertin M, Frind EO, Blowes D (2006) POLYMIN, User Guide, a 2D multicomponent reactive transport model with geochemical speciation and kinetic sulphide oxidation. Dept. of Civil, Geological and Mining Engineering, Ecole Polytechnique, Montreal
Parkhurst DL, Appelo CAJ (2013) Description of input and examples for PHREEQC Version 3—a computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations, 6–A43. U.S. Geological Survey Techniques and Methods, Reston
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
GR: conceptualization, methodology, model development, simulation and result analysis, and manuscript writing. RV: conceptualization, supervision, methodology, result analysis, manuscript writing and editing. The first draft of the manuscript was written by both authors. All the authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Roy, G., Valsala, R. Hydrogeochemical modelling of pyrite oxidation ion mobility in unsaturated mine waste rock piles. Environ Earth Sci 83, 127 (2024). https://doi.org/10.1007/s12665-023-11414-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12665-023-11414-z