Abstract
Background
Dexmedetomidine has dose-dependent selectivity for alpha 2 adrenoceptors. It is a good sedative with analgesic characteristics and good haemodynamic stability. Intranasal sedation is a non-invasive medication delivery method that is both safe and well accepted by both children and adults. One of the most common procedures in maxillofacial surgery is transalveolar extraction. In minor oral surgery, a painless transalveolar extraction with little post-operative pain would be ideal.
Aim
To examine the effectiveness of intranasal dexmedetomidine spray against intranasal normal saline spray in patients undergoing transalveolar extractions for anxiety relief.
Method
We compared sedation effect by Ramsay sedation scale, analgesia by visual analogue scale, monitored BP and pulse rate for anxiety, and spo2 levels for any complication in this prospective double-blinded randomized control study for two groups, A group with intranasal dexmedetomidine spray and the B group of intranasal NS spray for placebo effect at 0 min, 15 min, 30 min, and 45 min until transalveolar extraction.
Result
As a result of the intranasal spray of dexmedetomidine, there were no related problems such as respiratory depression. There was a substantial difference in sedation and analgesia between group A and the placebo group, as well as a significant decrease in pulse rate and hypotension in the dexmedetomidine group to reduce anxiety.
Conclusion
Intranasal injection of atomized dexmedetomidine (1.5 mcg/kg) for patient sedation having transalveolar extractions or other minor surgical operations in oral and maxillofacial surgery is clinically effective, convenient, lowers anxiety, and safe.
Clinical Trial Registration: No. CTRI/2021/07/035181.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Transalveolar extraction process is a frequently used technique in oral and maxillofacial surgery. A painless transalveolar extraction with minimum anxiety and minimal intra & post-operative pain would be desirable in minor oral surgery. The patients undergoing transalveolar extractions necessitates a reduction in the tension and anxiety associated with dental therapy [1]. Primary premedical anaesthesia and analgesics enable surgical experience easier and ensure that human disease-curing surgeries are successful. Benzodiazepines like midazolam, triazolam; and fentanyl are the most popular preoperative agents because of their anxiolytic effect. Post-operative pain and anxiety are observed in benzodiazepine derivatives. Premedication with use of sedative agents can reduce anxiety, pain, and irritation caused by injecting local anaesthetics. Dexmedetomidine is a versatile drug that is often used as a premedication for general anaesthesia. It can be injected into the body IM, IV, orally, or by the intranasal route [2]. It is dose-dependent having alpha-2 adrenoceptor selectivity. It is an excellent sedative with analgesic qualities and good haemodynamic stability. Because dexmedetomidine acts primarily on the locus coeruleus rather than the cerebrum, it is characterized by rapid as well as easy arousal, comparable to that of natural sleep [3, 4]. Nasal administration of dexmedetomidine is showing potential as a drug delivery method. Absorption is fast through the mucosa of the nose, so action is quick and avoids unpleasant stimuli to injections, easy administration, and outcome [4]. Intranasal sedation drugs for dental procedures are easy to use, effective, and have few adverse effects. The importance of the nasal atomization device is it delivers intranasal medication in a fine mist form, with the right amount of medication supplied, enhancing absorption and bioavailability for speedy and effective drug administration. Previous studies compared local anaesthetics with dexmedetomidine intra nasal or intra venous. Patients in the dexmedetomidine groups reported lower VAS pain scores and higher VAS satisfaction scores than patients in the local anesthesia group [5]. In our study, we compared the intra nasal normal saline spray and intra nasal administration of the dexmedetomidine in terms of reduction of the anxiety level and post-operative pain. Anxiety and discomfort are unavoidable side effects of transalveolar extractions in the absence of prior anaesthetic [6, 7]. Pre-medication that is just right, reduces patients’ concerns, resulting in a calm, sedated patient who has trouble-free transalveolar extractions and a speedy recovery in the post-operative phase [8]. The aim of our study is to examine the effectiveness of intra nasal dexmedetomidine spray against intra nasal normal saline spray in patients undergoing transalveolar extraction for anxiety relief.
Materials and Methods
This randomized control double blinded prospective study was performed after approval by our Deemed University’s Research Academy and Ethical Committee with completion no. SVIEC/ON/DENT/SRP/22015 and we also registered in clinical trials of India which was completed with No. CTRI/2021/07/035181. All patients have completed a form with informed consent to take part in this study.
Inclusion criteria:
-
Patients between 18 and 60 years of age
-
Healthy patients without any systemic illness.
Exclusion criteria:
-
Patients below the age of 18 and those over the age of 60
-
Patients with underlying systemic illness
-
Patients with cough, an URTI, blockage of nostrils, or dexmedetomidine allergies.
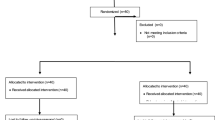
Fifty healthy participants between the ages of 18 and 60 who were scheduled for transalveolar extraction and were performed on all of the volunteers under local anaesthesia. The patients had not reported previous history of drug allergy used in this study. Before surgery, pre-operative radiographs were collected. Gender and ethnic differences were not considered.
Participants were assigned to one of two groups: Group A (25 participants) received 1.5 mcg/kg dexmedetomidine intranasally and group B (25 participants) received intranasal water (placebo group). Atomization of the drug was done to avoid the long procedure of drug delivery by nasal route. A nasal atomization device was used and the dose calculated was 1.5 mcg/kg as a standard protocol and as per studies done by Hiwarkar [9]. The study was conducted at the dental college in the department of oral and maxillofacial surgery. All aseptic measures, as well as COVID-19 precautions, were observed. After ten mins, the patient’s BP, oxygen saturation levels, and PR were tested for baseline levels. Patients in group A received 1.5 mcg/kg dexmedetomidine as an intranasal spray before local anaesthesia, while group B received intranasal normal saline spray as a placebo effect. Double blinding was applied at the patient and main researcher (operating surgeon) levels. To achieve the placebo effect, the patient had no awareness of which group they belonged to. The transalveolar extraction procedure was followed. The patient’s oxygen level, PR, and BP were measured immediately after the local anaesthetic injection and every 15 min until the surgery was done. The operating surgeon assessed the sedation score using Ramsay’s sedation scale [10] (as described below) every 15 min after the local anaesthesia was injected. The pain was assessed on a VAS every 15 min after the injection of local anaesthesia.
Ramsay sedation scale | |
|---|---|
1 | Patient is anxious and agitated or restless, or both |
2 | Patient is co-operative, oriented, and tranquil |
3 | Patient responds to commands only |
4 | Patient exhibits brisk response to light glabellar tap or loud auditory stimulus |
5 | Patient exhibits a sluggish response to light glabellar tap or loud auditory stimulus |
6 | Patient exhibits no response |
Statistical Analysis
IBM SPSS 20 for Windows statistical software was used to analyze the data. For quantitative data, statistical analysis was Chi-square test and paired sample t-test. P-values of 0.05 were considered significant in all statistical studies.
Result
The effectiveness, anxiety control, sedative, and analgesic effects of dexmedetomidine were investigated in a prospective, double-blind, and randomized control research. In our study, 50 patients met the inclusion criteria for transalveolar extractions, 28 of whom were male and 22 of whom were female, with a mean age of 28 years. The result shows after 15 min there was a significant difference in the spo2 with a p-value of 0.028 then at 30 min p-value is highly significant i.e., 0.001 and 0.002 at 45 min (Fig. 1). The result shows the difference is significant between the two groups. Mean of the BP at 15 min, 30 min, and 45 min with p-value 0.013, 0.002, and 0.001, respectively. There is a significant difference between 0, 15, 30, and 45 min with a p-value of 0.018, 0.002, 0.001, and 0.001, respectively. There is a significant difference between the two groups’ pain levels by VAS score and sedation by Ramsay sedation scale at 15 min, 30 min, and 45 min with a p-value less than 0.001 (Figs. 2, 3). Intranasal inhalation of dexmedetomidine can induce a decent sedative effect and a degree of postoperative analgesia with minimal side effects and problems. A decrease in the BP and pulse rate shows a reduction in the anxiety and calm state of the patient during the procedure (Fig. 4). Intranasal sprays of normal saline used as a placebo had no recordable effect in terms of anxiety control or sedation.
Discussion
Transalveolar extraction techniques in oral and maxillofacial surgery rely heavily on patients’ cooperation, as many patients are anxious about dental treatment, phobias of syringes, and other factors, surgeons commonly do minor oral surgical operations under LA rather than general anaesthesia because general sedation is a more invasive process [11]. With the right use of sedatives as well as analgesics, anxiety-provoking experiences and painful stimuli can be made tolerable. Conscious sedation, specifically, is an outpatient method available to oral surgeons that can help patients tolerate and accept unpleasant operations [12, 13]. Dexmedetomidine does not have any taste, colour, or odour [14] and it acts selectively on alpha-2 adrenergic receptors causing sedation and little analgesic effect in the system. Because of its properties, it can be used as an adjuvant for premedication [9, 15].
Its efficacy, anxiety-controlling activities, and sedative and analgesic effects were evaluated in a prospective, double-blind, randomized controlled experiment. Fifty patients satisfied the requirements for transalveolar extractions in our study, 28 of whom were men and 22 of whom were female, with an average age of 28 years.
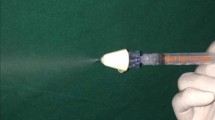
Controlled dexmedetomidine, a central and peripheral vasodilating drug, can be used to reduce BP effectively and safely. It is a near-ideal hypotensive medication because of its ease of administration, accuracy with anaesthetic medicines, and lack of toxic effect while maintaining adequate perfusion of essential organs. With dexmedetomidine-controlled hypotension, spinal fusion operations for scoliosis, septoplasty and tympanoplasty surgeries, and maxillofacial operations have all been safely performed. The results of our study demonstrate a significant change in systolic pressure comparing groups A and B at 45 min, with a p-value of 0.013. Furthermore, with a probability value of 0.013, 0.002, and 0.001, mean diastolic pressure was substantially different at 15 min, 30 min, and 45 min. This study looked into the use of atomized dexmedetomidine intranasally to sedate adult patients undergoing transalveolar extractions. According to the objective and subjective results, 1.5 mcg/kg dexmedetomidine provided effective sedation with minor side effects. The use of intranasally administered atomized dexmedetomidine to sedate adult patients having transalveolar extractions was investigated in this study (Fig. 5). About 1.5 mcg/kg dexmedetomidine produced effective sedation with few adverse effects, according to objective and subjective data. This study’s subjects were entirely normal healthy patients, which is unusual. The use of an atomized version with intranasal dexmedetomidine, which might help with bioavailability issues, is another noteworthy component of this study. A device called a Mucosal Atomizer device is used to achieve atomized intranasal delivery (Fig. 6). The Mucosal Atomization device is given intranasally, which is not only safe and patient-friendly but also very convenient. Higher drug serum levels are achieved when a drug is administered in its atomized form due to greater drug diffusion via the nasal mucosa and enhanced bioavailability. Furthermore, atomized medication is less likely to be blown backwards out of the nose because it is supplied as a mist. In children under the age of 18, Talon et al. [15] compared intranasal dexmedetomidine delivered via atomizer to oral midazolam. For general anaesthetic induction, both medications exhibited equal preoperative sedation and anxiolysis effects, with no notable side effects and minor haemodynamic effects. Yuen et al. [16] studied the duration to onset and period of sedative effects for nasal dexmedetomidine delivered via droplets and found that the sedative efficacy and pharmacological outcomes were similar.
The LA injection was given 15 min after dexmedetomidine, which was administered intranasally in our trial. The Ramsay sedation scale demonstrated considerable sedation compared to placebo at 30 and 45 min after dexmedetomidine infusion, with a p-value less than 0.001. Dexmedetomidine’s analgesic effectiveness is debatable. In a recent randomized, double-blind research, dexmedetomidine cannot provide more analgesic advantage than midazolam when used as IV sedation during third molar surgery under LA. Midazolam has side effects like apnoea, respiratory distress. Dexmedetomidine injection before and after surgery resulted in significant analgesia on the VAS when the combined average of all the participants in our trial was compared to placebo. There is a significant change in pain levels at 15 min, 30 min, and 45 min with a p-value less than 0.001. In addition, Ryu et al. [17] looked at the effects of parenteral and intranasal dexmedetomidine administration in third molar surgery and found that both routes had equal sedative effects. Intranasal inhalation of dexmedetomidine has sedative properties and can be employed as a non-invasive means of delivery in dentistry patients with preoperative anxiety. Respiratory depression is seen in most patients as a major side effect of dexmedetomidine medication according to Hiwarkar et al. As a result, monitoring oxygen saturation throughout the treatment and for the first 30 min afterwards is crucial [18]. We detected no concerns in our experiment, such as respiratory distress or a sudden decline in oxygen saturation; however, to avoid difficulties, oxygen saturation must be monitored during the treatment.
Conclusion
Inhalation of dexmedetomidine 30 min before transalveolar extraction via nasal mucosa can create reasonable sedation and adequate analgesia intra & post operatively with few side effects and complications. Normal saline intranasal sprays exhibited no placebo impact on anxiety control or drowsiness. Administration of 1.5 mcg/kg atomized dexmedetomidine intranasally produce significant action in pain-relieving, is easy to use, reduces anxiety, and is safe for patients of transalveolar extractions or other small surgical techniques in oral and maxillofacial surgery.
Abbreviations
- IM:
-
Intramuscular
- IV:
-
Intravenous
- VAS:
-
Visual analogue scale
- URTI:
-
Upper respiratory tract infection
- BP:
-
Blood pressure
- PR:
-
Pulse rate
- LA:
-
Local anaesthesia
References
Watanabe Y, Higuchi H, Ishii-Maruhama M et al (2016) Effect of a low dose of midazolam on high blood pressure in dental patients: a randomised, double-blind, placebo-controlled, two-centre study. Br J Oral Maxillofac Surg 54(4):443–448. https://doi.org/10.1016/j.bjoms.2016.02.006
Pasin L, Febres D, Testa V et al (2015) Dexmedetomidine vs midazolam as preanesthetic medication in children: a meta-analysis of randomized controlled trials. Paediatr Anaesth 25(5):468–476. https://doi.org/10.1111/pan.12587
Persson MP, Nilsson A, Hartvig P (1988) Relation of sedation and amnesia to plasma concentrations of midazolam in surgical patients. Clin Pharmacol Ther 43(3):324–331. https://doi.org/10.1038/clpt.1988.39
Chokshi AA, Patel VR, Chauhan PR, Patel DJ, Chadha IA, Ramani MN (2013) Evaluation of intranasal Midazolam spray as a sedative in pediatric patients for radiological imaging procedures. Anesth Essays Res 7(2):189–193. https://doi.org/10.4103/0259-1162.118954
Ryu DS, Lee DW, Choi SC, Oh IH (2016) Sedation protocol using dexmedetomidine for third molar extraction. J Oral Maxillofac Surg 74(5):926.e1-926.e9267. https://doi.org/10.1016/j.joms.2015.12.021
Neuman G, SwedTobia R, Koren L, Leiba R, Shavit I (2018) Single dose oral midazolam for minor emergency department procedures in children: a retrospective cohort study. J Pain Res 11:319–324. https://doi.org/10.2147/JPR.S156080
FallahinejadGhajari M, Ansari G, Soleymani AA, Shayeghi S, Fotuhi Ardakani F (2015) Comparison of oral and intranasal midazolam/ketamine sedation in 3–6-year-old uncooperative dental patients. J Dent Res Dent Clin Dent Prospects 9(2):61–5. https://doi.org/10.15171/joddd.2015.013
Goldman DR (2006) Intranasal drug delivery for children with acute illness. Curr Drug Ther. https://doi.org/10.2174/157488506775268470
Hiwarkar S, Kshirsagar R, Singh V et al (2018) Comparative evaluation of the intranasal spray formulation of midazolam and dexmedetomidine in patients undergoing surgical removal of impacted mandibular third molars: a split mouth prospective study. J Maxillofac Oral Surg 17(1):44–51. https://doi.org/10.1007/s12663-016-0992-5
Dawson R, von Fintel N, Nairn S (2010) Sedation assessment using the Ramsay scale. Emerg Nurse 18(3):18–20. https://doi.org/10.7748/en2010.06.18.3.18.c7825
Shetty SK, Aggarwal G (2016) Efficacy of intranasal dexmedetomidine for conscious sedation in patients undergoing surgical removal of impacted third molar: a double-blind split mouth study. J Maxillofac Oral Surg 15(4):512–516. https://doi.org/10.1007/s12663-016-0889-3
Chatrath V, Kumar R, Sachdeva U, Thakur M (2018) Intranasal fentanyl, midazolam and dexmedetomidine as premedication in pediatric patients. Anesth Essays Res 12(3):748–753. https://doi.org/10.4103/aer.AER_97_18
Xavier SA, Wahab A, Sivakumar M (2020) Evaluation of intranasal administration of dexmedetomidine during therapeutic extraction. J Pharm Res Int 32(18):112–119
Kurmanji JM, Sulaiman SA, Kah LK, Chandrasekaran PK (2010) Depression and low bone mineral density: The correlation among Chinese. Asian J Psychiatr 3(3):134–137. https://doi.org/10.1016/j.ajp.2010.07.013
Talon MD, Woodson LC, Sherwood ER, Aarsland A, McRae L, Benham T (2009) Intranasal dexmedetomidine premedication is comparable with midazolam in burn children undergoing reconstructive surgery. J Burn Care Res 30(4):599–605. https://doi.org/10.1097/BCR.0b013e3181abff90
Yuen VM, Hui TW, Irwin MG, Yuen MK (2008) A comparison of intranasal dexmedetomidine and oral midazolam for premedication in pediatric anesthesia: a double-blinded randomized controlled trial. Anesth Analg 106(6):1715–1721. https://doi.org/10.1213/ane.0b013e31816c8929
Cheung CW, Ying CL, Chiu WK, Wong GT, Ng KF, Irwin MG (2007) A comparison of dexmedetomidine and midazolam for sedation in third molar surgery. Anaesthesia 62(11):1132–1138. https://doi.org/10.1111/j.1365-2044.2007.05230.x
Nooh N, Sheta SA, Abdullah WA, Abdelhalim AA (2013) Intranasal atomized dexmedetomidine for sedation during third molar extraction. Int J Oral Maxillofac Surg 42(7):857–862. https://doi.org/10.1016/j.ijom.2013.02.003
Acknowledgements
The authors would like to thank Dr. Gulam Naviwala for his comments, insightful suggestions and careful reading of the manuscript.
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any conflict of interest.
Ethical Approval
Our Deemed University’s Research Academy and Ethical Committee with completion No. SVIEC/ON/DENT/SRP/22015.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mulay, M., Mahajan, A., Shah, N. et al. Comparative Evaluation of Intranasal Dexmedetomidine Spray Versus Intranasal Normal Saline Spray in Patients Undergoing Transalveolar Extractions for Anxiety Reduction: A Randomized Control Study. J. Maxillofac. Oral Surg. 22, 627–633 (2023). https://doi.org/10.1007/s12663-023-01933-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12663-023-01933-4