Abstract
The decision to exercise is not only bound to rational considerations but also automatic affective processes. The affective–reflective theory of physical inactivity and exercise (ART) proposes a theoretical framework for explaining how the automatic affective process (type‑1 process) will influence exercise behavior, i.e., through the automatic activation of exercise-related associations and a subsequent affective valuation of exercise. This study aimed to empirically test this assumption of the ART with data from 69 study participants. A single-measurement study, including within-subject experimental variation, was conducted. Automatic associations with exercise were first measured with a single-target implicit association test. The somato-affective core of the participants’ automatic valuation of exercise-related pictures was then assessed via heart rate variability (HRV) analysis, and the affective valence of the valuation was tested with a facial expression (FE; smile and frown) task. Exercise behavior was assessed via self-report. Multiple regression (path) analysis revealed that automatic associations predicted HRV reactivity (β = −0.24, p = .044); the signs of the correlation between automatic associations and the smile FE score was in the expected direction but remained nonsignificant (β = −0.21, p = .078). HRV reactivity predicted self-reported exercise behavior (β = −0.28, p = .013) (the same pattern of results was achieved for the frown FE score). The HRV-related results illustrate the potential role of automatic negative affective reactions to the thought of exercise as a restraining force in exercise motivation. For better empirical distinction between the two ART type‑1 process components, automatic associations and the affective valuation should perhaps be measured separately in the future. The results support the notion that automatic and affective processes should be regarded as essential aspects of the motivation to exercise.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Regular physical activity and exercise are effective health-preventive (Rhodes, Janssen, Bredin, Warburton, & Bauman, 2017) and health-promoting strategies (Warburton, Nicol, & Bredin, 2006). However, the analysis of global trends in physical activity indicates that worldwide more than 27% of adults are insufficiently active, according to the recommendations of the WHO (2010). In high-income countries, the level of inactivity is even higher (>35%) and continues to rise (Guthold, Stevens, Riley, & Bull, 2018). Most health interventions today aim to change activity behavior by stimulating rational deliberation, e.g., about the health benefits of exercise. The approach underlying these interventions refers to the assumption that human decision-making is fundamentally rational (e.g., Brand & Cheval, 2019; Ekkekakis, Zenko, Ladwig, & Hartman, 2018). However, precisely this premise has never been without controversy (e.g., Simon, 1986). Hence, within exercise psychology over the past few years, the investigation of the role of automatic and affective processes in health behavior change and maintenance has become a flourishing field (e.g., Ekkekakis & Brand, 2019; Rebar et al., 2016). Related to this novel line of research, a couple of new theories and models have emerged (e.g., Cheval et al., 2018; Conroy & Berry, 2017), of which the affective–reflective theory (ART) of physical inactivity and exercise (Brand & Ekkekakis, 2018) is under study here.
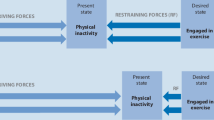
The ART is a dual-process theory with an explicit exercise psychological account that provides a theoretical framework for the interplay of automatic affective processing (type-1: fast and automatic in the sense that it requires minimal cognitive resources and effort; Evans & Stanovich, 2013) and reflective processing (type-2: generally slower and it requires controlled reasoning under the use of working memory; Evans & Stanovich, 2013) and their impact on exercise behavior. According to the ART, external (e.g., seeing somebody jogging) or internal stimuli (e.g., thinking of the intention to do more exercise) will trigger the automatic affective type‑1 process and activate spontaneous mental associations with memories of exercise. These automatic associations rely on mental associations between the concept of “exercise” and other semantic concepts in memory. It is believed that any activation of the “exercise” concept will automatically reactivate associated concepts (e.g., “displeasure,” “sweat,” “exertion”). Determined by the relative strength and type of activated associations (e.g., “running” and “displeasure”), an automatic affective valuation follows. The affective valuation emerges from somato-affective bonds formed through one’s past experiences with exercise (e.g., the experience of shortness of breath during a run). These bonds are re-actualized during the automatic processing of the exercise-related stimulus.
According to the ART, the automatic affective valuation includes core affective feelings of pleasure or displeasure that arise directly from the body. This relates to the definition of core affect as a “neuro-physiological state consciously accessible as a simple, primitive, nonreflective feeling most evident in mood and emotion but always available to consciousness” (Russell & Feldman Barrett, 2009, p. 104). Core affect can be dimensionally classified according to its affective valence (positive–negative) and level of arousal (activation; Russell, 1980). The affective valuation provides the basis for a reflective evaluation (type‑2 process; e.g., rational reflection about personal values and expected consequences of exercising) in a way that it can color subsequent reasoning, especially when self-regulatory resources are available. The type‑1 process is also connected with a direct impulse to change one’s actual state of physical inactivity; the type‑2 process can result in divergent or concurrent action plans.
Recent empirical studies have investigated aspects of automatic processes very similar to the way they are defined in the ART. For instance, several independent studies have shown that those who exercise more have stronger positive automatic associations with exercise compared to those who exercise less (for systematic reviews see Cheval et al., 2018; and Schinkoeth & Antoniewicz, 2017). Automatic associations were shown to be related to exercise decision preferences (e.g., Brand & Schweizer, 2015; Zenko & Ekkekakis, 2019). Activated automatic associations with exercise have been shown to lead to self-imposed physical load in a brief exercise session immediately after the activation (Antoniewicz & Brand, 2016). Consistent with this, the more negative the automatic associations were, the less future exercise behavior was reported by participants (Eves, Scott, Hoppé, & French, 2007).
Very few studies have tried to approach the automatic affective valuation of exercise and its affective core, as defined in the ART, as yet. One study relies on data from the Affective Misattribution Procedure (AMP; Payne, Cheng, Govorun, & Steward, 2005) and found that indoor fitness center exercisers had more positively valenced affect after subliminally presented fitness center primes, compared with participants who preferred to exercise in different settings (Antoniewicz & Brand, 2014). Core affect as a neuro-physiological state is accompanied by activity patterns in the autonomic nervous system (ANS), as well as facial changes (Russell, 2003). Two more recent studies (Brand & Ulrich, 2019; Schinkoeth, Weymar, & Brand, 2019) used approaches with biometric data to assess these psychophysiological and behavioral changes associated with the arousal and valence of the automatic valuations affective core. Both studies play essential roles in the empirical study presented here (study descriptions below).
This study aims to disentangle and relate to each other the ART’s two type‑1 process components, i.e., automatic associations and affective valuation of exercise. Accordingly, it is necessary to have tests that allow the separate measurement of these two constructs.
Automatic associations with exercise-related stimuli
Automatic (evaluative) associations between the mental concept of “exercise” and positive or negative attributes (e.g., “pleasure” and “displeasure”) are often referred to in the literature as automatic affective evaluations of exercise (e.g., Conroy & Berry, 2017; Rebar et al., 2016). The majority of studies that have investigated automatic associations with exercise have used variants of the Implicit Association Test (IAT; Greenwald, McGhee, & Schwartz, 1998) to measure these associations (Cheval et al., 2018; Chevance, Bernard, Chamberland, & Rebar, 2019; Schinkoeth & Antoniewicz, 2017).
The standard version of the IAT (Greenwald et al., 1998) uses stimuli from a category with two complementary objects of evaluation (e.g., the target concept “exercise” and the comparison concept “physical inactivity,” with words like “training,” “workout,” “gym,” and “read,” “sit,” “sleep” as word stimuli) and an evaluative category with the two concepts “good” and “bad” (e.g., “pleasure,” “joy,” “fun,” and “terrible,” “disgusting,” “sad” as word stimuli). Study participants are required to sort the stimuli presented on a computer screen as fast and accurately as possible to their respective concept by pressing a key on the keyboard, in a test setup in which two concepts from the two categories share the same response key (e.g., “exercise” and “good” paired on one response key, and “physical inactivity” and “bad” on the other). Combinations of concepts vary across test blocks. The test rests on the assumption that those who have positive associations with exercise will sort exercise-related stimuli faster when “exercise” and “good” (vs. “exercise” and “bad”) share the same response key since the response is compatible with their mental associations.
Although it is known that IAT scores can be considered as only partly automatic (Gawronski & De Houwer, 2014), today the IAT is almost regarded as a standard method for measuring automatic associations. Some meta-analyses support the validity of the IAT (e.g., criterion-related validity: Greenwald, Poehlman, Uhlmann, & Banaji, 2009; and convergent validity: Hofmann, Gawronski, Gschwendner, Le, & Schmitt, 2005), while other reviews have reached more skeptical conclusions (e.g., Schimmack, 2019) with regard to whether the test can predict individual differences in behavior.
Heart rate variability as a somatic correlate of the affective valuation of exercise
Peripheral correlates of core affective feelings can be measured on the somatic level as activity patterns in the ANS, for example, as changes in heart rate (HR; e.g., Russell, 2003) and HRV (Koval et al., 2013). HR is a somatic indicator of the combined regulatory processes in both divisions of the ANS, i.e., the sympathetic and parasympathetic systems. Higher HR often occurs when the activity of the sympathetic system is higher than parasympathetic activity, but coinhibition or coactivation of the two divisions can occur as well (Berntson, 2019). Hence, an affective stimulus can produce an accelerated, decelerated, or even unchanged HR response depending upon which activational input is larger (Berntson, Cacioppo, & Quigley, 1991). As a result, empirical studies investigating links between affect and changes in HR have found inconclusive results (Brouwer, van Wouwe, Mühl, van Erp, & Toet, 2013; Cacioppo, Berntson, Larson, Poehlmann, & Ito, 2000).
HRV is another candidate for the investigation of affective components at the somatic level. It is defined as the variation in the periods between successive heartbeats. Evidence suggests that HRV is a valid measure of parasympathetic (vagal) activity when respiration rate and depth are sufficiently controlled. According to the neurovisceral integration model (Thayer & Lane, 2000), reactivity in HRV (phasic HRV) can be interpreted in terms of the self-regulation of affect via prefrontal–subcortical routes (Thayer, Åhs, Fredrikson, Sollers, & Wager, 2012).
To the authors’ knowledge, only one study has so far analyzed HRV reactivity to investigate individual differences in participants’ affective responses to exercise-related stimuli (Schinkoeth et al., 2019). In that laboratory study, participants were confronted with exercise-related and neutral control pictures, while their HRV was continuously recorded. Ratings of affective valence and arousal of all exercise-related and control pictures were measured with self-assessment manikins (Bradley & Lang, 1994). The participants’ habitual exercise volumes in everyday life were obtained via self-report. Results indicated that those who reported less exercise and evaluated the exercise-related pictures as less pleasant showed a decrease in HRV in response to exercise stimuli. The authors suggested that the observed HRV reactivity could be interpreted as a somatic component of the affective valuation of exercise.
Facial expression analysis and the affective valence of the automatic valuation of exercise
The valence of affective feelings is sometimes associated with characteristic facial action (e.g., Cacioppo et al., 2000; Lang, Greenwald, Bradley, & Hamm, 1993). This has often been measured with facial electromyography (EMG; e.g., Cacioppo et al., 2000). Viewing unpleasant pictures, for example, from the international affective picture system database (IAPS; Lang, Bradley, & Cuthbert, 1997) has been shown to increase corrugator (“frown-muscle”) activity. In contrast, viewing pleasant pictures prompted zygomatic (“smile-muscle”) tension (Lang et al., 1993). Recent technological innovations allow the automatic computerized coding and analysis of facial actions in the whole face from video recordings (Kulke, Feyerabend, & Schacht, 2018; Timme & Brand, 2020).
To the best of the authors’ knowledge, only one study to date has used automatic FE analysis as a means to identify the valence of inter-individually different automatic affective valuations of exercise (Brand & Ulrich, 2019). There, participants worked through an emotional Stroop task, in which they were asked to respond to exercise-related and control stimuli with either a positive (smile, pleasure) or a negative (frown, displeasure) expression on their face depending on how the respective stimulus was presented. It was hypothesized that participants are faster in giving compatible responses (e.g., if someone who does not like exercise has to react with a frown to an exercise-related photo) than in giving incompatible responses (e.g., if the same person has to react to the same picture with a smile since it was presented differently). Results showed that the participants who reported lower exercise volumes and a more negative reflective evaluation of exercise were faster in giving frowns after exercise-related pictures (compared with those who exercised more). The authors interpreted this finding as evidence for a negative automatic affective valuation on reminders of exercise.
This study
The aim of the present study was to empirically analyze the relationship between the two components of the type‑1 process (automatic association and affective valuation of exercise) postulated in the ART (Brand & Ekkekakis, 2018). Different measurement methods were used for these components in an effort to avoid common method bias (Podsakoff, MacKenzie, Lee, & Podsakoff, 2003). Automatic associations were assessed with a reaction time-based test (a recoding-free variant of the single-target IAT; Brand & Utesch, 2019). Two separate measures were used to identify the participants’ automatic affective valuations of exercise. The somatic component of the affective valuation was addressed by the HRV reactivity test of Schinkoeth et al. (2019), and the affective valence of the reaction was evaluated with the FE task of Brand and Ulrich (2019). The authors hypothesized that more negative (vs. positive) automatic associations with exercise would predict more negative (vs. positive) affective valuation of exercise, as reflected in experimentally induced HRV reactivity as well as in the affective valence compatibility–incompatibility effect in the FE task. It was furthermore expected that individuals with a more negative (vs. positive) automatic response to exercise would report exercising less (vs. more) often in their everyday lives.
Materials and methods
Participants and study sample
The authors sampled data from 107 individuals (Mage = 25.9 ± 4.5 years old, 64 female) that were recruited on a university campus and asked for participation (without applying preselection criteria) after full disclosure of the study aim and measurement procedures. All participants gave written informed consent. Measured data from 38 participants (Mage = 27.2 ± 4.9 years, 23 female) could not be used in our main analyses for various technical reasons: technical malfunction during video recording of the face (n = 1), errors in algorithmic identification of a face for facial action analysis (n = 5), more than 20% incorrect reactions in the facial expression task (n = 19), and technical malfunction during heartbeat recording (n = 13).
The data subjected to statistical analysis for hypothesis testing (study sample), therefore, included data from 69 young adults (Mage = 25.3 ± 4.2 years; 41 female). There were no significant differences between the excluded subsample and the study sample in any of the main study variables. However, the participants in the study sample were slightly younger than those in the excluded subsample, t(105) = 2.15, p = .034, d = 0.43.
Automatic associations with exercise
The participants’ automatic associations with exercise were assessed with a recoding-free (RF; Rothermund, Teige-Mocigemba, Gast, & Wentura, 2009) variant of the single-target IAT (STIAT; Bluemke & Friese, 2008). This test is the object of ongoing development in the authors’ working group, but a preliminary version of it was presented recently at a conference (Brand & Utesch, 2019). In the RF-STIAT, participants are asked to sort exercise-related target stimuli (exercise words: pulse, breathing, sweating, exertion, muscles, endurance) and attribute stimuli (positive words: pleasant, great, super, awesome, wonderful, gorgeous; negative words: unpleasant, annoying, bad, awful, horrible, terrible) as fast as possible to the target concept “exercise” (exercise-related stimuli), or “good” and “bad” (attribute stimuli). The sorting task consisted of pressing the keys “E” or “I” on the computer keyboard. RF variants of the IAT aim to eliminate recoding effects (Rothermund et al., 2009) by random trial-to-trial alteration of target-compatible and target-incompatible trials. In target compatible conditions, the target concept and “good” are on the one side, and the control concept and “bad” are to the other side, for those who have positive associations with the target concept; in target-incompatible trials the control concept and “good” are to the one side, and the target concept and “bad” are to the other side, for those who have positive associations with the target concept. The evaluative concepts remain constantly assigned to one side (e.g., “positive” is left or right; inter-individually randomized) whereas the target concept (here: “exercise”) randomly switches between left and right. The authors calculated and used the G‑score (Richetin, Costantini, Perugini, & Schönbrodt, 2015), which represents a robust, scale-invariant, nonparametric measure of automatic associations. This score relies on ordinal information (percentiles of the participants’ response latencies) and represents the difference between the means of the Gaussian rank latencies in the two test blocks. It drastically reduces the bias of outliers. Spearman-Brown corrected split-half reliability of this test score in the current sample was r = .57, and thus very similar to those reported in previous studies (e.g., Raccuia, 2016).Footnote 1
Fast responses in compatible pairings indicate a strong association between the concept of “exercise” and the evaluative attribute “good” and results in a high positive G‑score.
HRV reactivity
The procedure described in Schinkoeth et al. (2019) was used for the measurement of HRV reactivity. It consists of 14 exercise-related pictures and 14 affectively neutral control pictures from the IAPS (Lang et al., 1997; e.g., household items like an electric socket, a teaspoon, a chair). The exercise-related pictures depicted exercise of differing intensity, e.g., football, hip-hop dancing, and tennis (without showing faces or other obvious display of affect) and exercise equipment, such as a badminton racket with a shuttle or a high-jump landing pad. The pictures were taken from online stocks and selected according to recommendations of Rebar et al. (2016). Heartbeat and interbeat intervals were measured with a Polar RS800CX heart rate monitor and a chest belt sensor (Polar Electro Oy, Kempele, Finland). Data preprocessing and score calculation was performed with Kubios HRV software (Kubios Oy, v3.0.0, Kuopio, Finland). The root mean square of successive differences between normal heartbeats (RMSSD) was chosen as the HRV indicator (Schinkoeth et al., 2019). This is a time-domain index of HRV reflecting vagal tone (Thayer & Lane, 2000) and is a valid measure in ultra-short-term recordings, i.e., in periods of between 10 and 60 s (Shaffer & Ginsberg, 2017). HRV scores were natural log-transformed to account for skewed distributions prior to further analysis (Laborde, Mosley, & Thayer, 2017). HRV reactivity is the ratio of HRV during exercise-related stimuli to HRV during neutral stimuli (i.e., dividing HRV exercise by HRV neutral). Potential confounding variables for HRV (e.g., age, gender, smoking behavior, habitual levels of alcohol consumption, and disorders of the cardiovascular system) were assessed, as suggested by Laborde et al., (2017), and post-hoc analyses revealed that HRV remained unaffected by these variables.
Facial expression task
The FE task described in Brand and Ulrich (2019) was used for measuring the affective valence of the participants’ automatic affective valuation of exercise. However, in this study, the same 14 exercise-related pictures as in the HRV were employed. Another difference from the original procedure was that gray rectangles were used instead of pictures representing a comparison concept (physically inactive study work, in Brand & Ulrich, 2019). The reason for not choosing neutral pictures from the IAPS in the present study was that the authors wanted to avoid artifacts due to irritation (e.g., when participants are asked to respond with a smile to usually unvalenced objects like an electric socket; Lang et al., 1997). The authors assumed that gray rectangles provide a more appropriate baseline for the latency of showing an affective facial expression to affectively neutral stimuli in this case.
All exercise pictures and gray rectangles were presented twice in random order for 4 s each, with a 3‑s blank screen between them. All stimuli were 900 × 600 pixels in size, had a thin white frame (two pixels), and were presented centered against a black screen. The participants’ task was to produce either a smile or a frown as quickly as possible after stimulus presentation, depending on whether the picture was presented upright (symmetric to the borders of the monitor in one trial) or tilted (1 degree to the left or to the right) according to what they had learned in practice trials before the test. Facial responses (latencies in the generation of facial expressions, i.e., smile or frown, after stimulus presentation; error rates in producing the requested facial expression) were analyzed with automated FE coding software (Affectiva Affdex; McDuff et al., 2016; as implemented in the iMotions platform for biometric research). This software detects faces in videos and tracks the movements of 34 facial landmarks (e.g., brow furrow, nose wrinkle, lip pull corner). Data from these facial movements are analyzed and integrated to classify facial actions (here: smile vs. frown). The detection of positive and negative facial expressions by Affectiva Affdex has been shown to be significantly correlated with the results of EMG measures of corrugator and zygomaticus muscle activity (Kulke et al., 2018).
Following the recommendation by Brand and Ulrich (2019), two ratio scores were calculated. For those who exercise more, and who are therefore supposed to like exercise more, responding with a smile is the compatible response. Smile latency score is the ratio of average onset time for signs of positive valence in a participant’s FE after exercise-related stimuli to the average onset time for signs of positive valence after control stimuli. Scores smaller than 1 indicate that participants are comparably faster to respond with a requested smile after exercise pictures, which is considered indicative of a positive affective valuation of exercise. This applies conversely to frown latency score, which was calculated as the ratio of average onset time for signs of negative valence in one’s FE after exercise-related stimuli to average onset time for signs of negative valence after control stimuli. Scores smaller than 1 indicate a comparably faster response with a requested frown after exercise pictures. Those who exercise less, and who are therefore supposed to like exercise less, are meant to respond faster to exercise pictures with a frown, since according to their negative affective valuation of exercise, responding with a frown is the compatible reaction for them.
Calculation and analysis of both scores are essential (Brand & Ulrich, 2019) since it is known from earlier studies with emotional Stroop tasks that effects are mainly driven by reaction time delays in the incompatible condition (Pratto & John, 1991). Therefore, in heterogeneous study samples like here, when the rate of individuals with positive or negative affective valuations of exercise is unknown, both response modality scores (frown and smile latencies) must be analyzed.
Exercise behavior
Exercise behavior was assessed with one item from the International Physical Activity Questionnaire (Hagströmer, Oja, & Sjöström, 2006). Participants were asked how often in a week they would usually spend time for moderate or vigorous exercise. They indicated numbers of exercise sessions and average duration of sessions by typing their answers in two separate free-text fields presented on the computer screen. Number of exercise sessions was used for hypothesis testing since the authors have learned from participants in their own previous studies that this information can often be remembered more easily (and probably more accurately) than the exact session lengths.
Procedure
Tests were conducted under controlled conditions in the laboratory. After the participants gave full informed written consent for study participation, they were seated in front of a computer. Participants first completed the RF-STIAT. Then the chest belt sensor for testing HRV reactivity was attached. This test started with a rest measure, for which participants were asked to remain in a quiet sitting position for 5 min in front of the blank computer screen. Heartbeat and interbeat-interval (IBI) data recorded during this period were used to calculate average HR rest and HRV rest. The 28 pictures were then presented for 7 s each in four blocks (seven pictures randomly assigned to the blocks and in random sequence; block sequence was fixed to “exercise,” “neutral,” “exercise,” and “neutral” for all participants, and blocks were separated by 10‑s blank screens; see Schinkoeth et al., 2019). Participants were instructed in advance that they would see exercise-related pictures in order to elicit their personal thoughts and feelings about exercise. After the HRV-related part of the test procedure, the heartbeat recording was stopped. The FE task then followed. The participants finally gave their personal data (age, gender) and reported about their exercise behavior and potential HRV confounders (e.g., food intake and caffeine consumption).
Design, test scores, and statistical tests
A single-measurement study with within-subject experimental variation was conducted. Bivariate intercorrelations between all main study variables were analyzed with Pearson correlation coefficients. Means, standard deviations, and correlations between automatic associations, HR rest, log-transformed HRV scores (HRV rest and HRV reactivity), facial action scores (smile latency and frown latency), and exercise behavior (number of exercise sessions) are shown in Table 1. Two separate linear regression path-models with maximum likelihood estimation were calculated for hypothesis testing using the lavaan (0.6–4; Rosseel, 2012) package for R (R Core Team, 2019). Path model A tested relations between the variables automatic associations with exercise (exogenous variable), HRV reactivity, smile latency, and number of exercise sessions. Path model B contained the same variables, except that frown latency (instead of smile latency) was included in this model. The authors interpreted qualitative and descriptive model fit indicators (i.e., χ2, p-value, Tucker–Lewis Index [TLI], comparative fit index [CFI], root-mean-square error of approximation [RMSEA], standardized root mean square residual [SRMR], Akaike Information Criterion [AIC], and Bayesian Information Criterion [BIC]) for both models. Recommended thresholds of model fit indices, their interpretation, and use for the evaluation of a model fit are explained in Kline (2015). Following recommendations by Goodboy and Kline (2017, pp. 72–73), nonsignificant paths were not deleted.
The sequence of measurements in the current study reflects the sequence of processes as postulated in the ART (from automatic associations to affective valuation to behavior). To strengthen the argument that the type‑1 process under investigation can be considered in this sequence, two reversed models (models A′ and B′) were also fitted (i.e., with number of exercise sessions as the exogenous variable).
Results
The reported exercise volume per week ranged from 0–720 min per week (M = 166.36 min, SD = 167.38 min). More exercise sessions per week were correlated with lower HR rest and higher HRV rest. Further analyses revealed a high negative correlation between HR rest and HRV rest. Descriptive statistics, together with these and other correlations, are shown in Table 1.
Path analysis model A (smile latency score)
Model A (Fig. 1a) exhibited good fit to the data, χ2 (2) = 2.74, p = .254, TLI = 0.81, CFI = 0.94, RMSEA = 0.07 (90% confidence interval [CI]: .00, .26), and SRMR = 0.06. The AIC was 180.04, and the sample-size adjusted BIC was 173.63. More negative (vs. positive) automatic associations with exercise predicted an increase (vs. decrease) in HRV during the presentation of exercise-related stimuli (p = .044), and this increase (vs. decrease) statistically predicted lower (vs. higher) self-reported values of exercise (p = .013). This finding is in line with what the authors had expected. The regression weight to predict smile latency score by automatic associations remained too small for statistical significance (p = .078). The sign of the coefficient refers to the expected direction of the influence: more negative (vs. positive) automatic associations are related to delayed (vs. increased) response times for smiles. Aside from this, and contrary to the authors’ prediction, time-delayed (vs. accelerated) smile responses allow the statistically significant prediction of more (vs. less) exercise sessions per week (p = .031).
The reverse model A′ (Fig. 2a) exhibited insufficient model fit, χ2 (2) = 5.61, p = .060, TLI = 0.10, CFI = 0.70, RMSEA = 0.16 (90% CI: .00, .33), SRMR = 0.09, AIC = −61.39, and BIC = −67.80.
Path analyses model B (frown latency score)
Fit indices for model B were χ2 (2) = 1.756, p = .416, TLI = 1.09, CFI = 1.00, RMSEA = 0.00 (90% CI: .00, .23), and SRMR = 0.05, indicating a good fit to the data. The AIC was 177.86, and the sample-size adjusted BIC was 171.36. Again, increase (vs. decrease) in HRV while viewing exercise-related stimuli was statistically predicted by more negative (vs. positive) automatic associations with exercise (p = .042), and less (vs. more) self-reported exercise sessions were predicted by this increase (vs. decrease) in HRV (p = .048). Although the signs of all the regression weights here were as expected (positive vs. negative automatic associations predict slower vs. faster response times to frown, and these predict more vs. less exercise behavior), the paths between automatic associations and frown latencies after exercise-related pictures (p = .117), and reaction time to frown and number of exercise sessions per week (p = .344), remained nonsignificant (Fig. 1b).
The reversed model B’ (Fig. 2b) exhibited good global fit to the data, χ2 (2) = 1.88, p = .390, TLI = 1.05, CFI = 1.00, RMSEA = 0.00 (90% CI [.00, .23]), SRMR = 0.05, AIC = −63.26, and BIC = −69.76. However, in this reversed model only one path was significant, indicating that numbers of exercise sessions can predict HRV reactivity (p = .032).
Discussion
This study aimed to empirically test theoretical assumptions about the automatic and inherently affective type‑1 process as defined in the ART (Brand & Ekkekakis, 2018). Hence, the authors hypothesized that negative (vs. positive) automatic associations with exercise would predict a negative (vs. positive) affective valuation of exercise and expected that the two indices of affective valuation (i.e., changes in HRV as an expression of the somatic component, and affective valence as measured with the FE task) could be used to statistically predict the participants’ self-reported exercise behavior. Not all of the tested hypotheses found empirical support in the data. As hypothesized, automatic associations with exercise predicted HRV reactivity during the viewing of exercise-related pictures, and this HRV reactivity was associated with the number of self-reported exercise sessions per week. The regression weights for the paths between automatic associations and affective valuation as measured with the FE task were in the expected direction (negative in model A for smile latency; positive in model B for frown latency) but remained statistically insignificant. Frown latency did not significantly predict exercise behavior, and, in opposition to the authors’ hypothesis, faster (vs. slower) smiles on exercise-related pictures were correlated with less (vs. more) exercise behavior. The authors conclude that the measurement of the valence of the affective valuation with the FE task did not work well in this study.
Automatic associations with exercise, HRV reactivity, and exercise behavior
Participants with negative automatic associations showed an increase in HRV in response to reminders of exercise, and this increase was linked to fewer exercise sessions per week. Vice versa, positive automatic associations with exercise were linked with a decrease in HRV while viewing exercise pictures, and more exercise sessions in consequence. First and foremost, the authors consider these findings to be preliminary evidence for the process postulated in the ART (Brand & Ekkekakis, 2018)—that reminders of exercise trigger exercise-related automatic associations (here: evaluative associations between the semantic concept of exercise and semantic representations of “good” and “bad”), which are interrelated with an instant psychophysiological reaction (see Damasio, 1996). In terms of the ART, the authors think that the psychophysiological reaction reflects the somatic core of the participants’ affective valuation of exercise (Schinkoeth et al., 2019). Nevertheless, given the correlational nature of the study, the directional relations between the variables must not be over-interpreted. However, results of the reversed models (i.e., insufficient data fit in model A′ and nonsignificant paths in model B′) indicate that the hypothesized sequence for this process in the ART may continue to be accepted. The significant relation between exercise sessions and HRV reactivity (in model B′) is also in accordance with the assumptions of the ART. According to Brand and Ekkekakis (2018), experiences with exercise leave mental traces in memory that form or strengthen automatic exercise associations and color the affective valuation of exercise.
However, the direction of HRV reactivity in this study was different from the one observed in Schinkoeth et al. (2019). In this earlier study, participants who reported less exercise, and were therefore supposed to like exercise less, reacted with a decrease in HRV while viewing exercise-related stimuli and not with an increase, as was observed in the study here (to the best of the authors’ knowledge, the two studies are the only ones to date that have examined HRV reactivity to exercise reminders). Of note, previous studies outside of exercise psychology, in which psychophysiological reactions to positively and negatively charged affective stimuli were investigated, have shown both an increase and a decrease in HRV (Kreibig, 2010 for a systematic review). Differences in study samples from the two exercise-related studies (Schinkoeth et al., 2019 and the one presented here) might have contributed to the divergent results: participants in the previous study (Schinkoeth et al., 2019) were on average 1.9 years younger (Mage = 23.4 ± 5.9 years; t[158] = −2.27, p = .025, d = −0.36) and reported on average 83.7 min more exercise per week (M = 250.1, SD = 184.7; t[158] = 2.97, p = .003, d = 0.48).
Aside from these sample differences, phasic changes in HRV have been interpreted in terms of a variety of affect self-regulation processes (e.g., Appelhans & Luecken, 2006; Holzman & Bridgett, 2017; Thayer et al., 2012). For example, increased HRV (as found here for individuals with a presumably negative affective valuation of the exercise stimuli, according to their automatic associations with exercise and self-reported exercise behavior) has been attributed to higher prefrontal control on the heart and facilitating effective emotional regulation (by either reappraisal or suppression; Butler, Wilhelm, & Gross, 2006; Thayer et al., 2012). Other authors have suggested that processes connected with attentional disengagement may have the same effect (Jennings, 1986; Thayer & Lane, 2000). On the other hand, decrease in HRV (as found here for the participants with a presumably positive affective valuation, according to their automatic associations with exercise and self-reported exercise behavior) has been associated with lower vagal impact on the heart (and perhaps a higher sympathetic activation; Thayer et al., 2012), and, for example, with states of approach and avoidance motivation (e.g., preparing for activity; Kreibig, 2010; Wu, Gu, Yang, & Luo, 2019). In the authors’ view, there should be no further speculation about this point here; instead, they posit that phasic HRV reactivity was measurable here and that more research is needed for a better understanding of the probably complex associations with affective self-regulation and motivational impulses.
Automatic associations with exercise, affective valence of the valuation, and exercise behavior
The results for the affective valence of the automatic valuation as measured with the FE task (i.e., latencies to produce a smile or frown in response to exercise pictures in comparison to control pictures) were not as hypothesized, as shown in a previous study (Brand & Ulrich, 2019). Although the directions of the correlations (regression weights) between automatic associations and frown and smile latency scores, and between frown latency and exercise behavior, were as expected, the coefficients remained too small to reach statistical significance. Above all, a highly counterintuitive correlation between smile latency and exercise behavior emerged (longer reaction times to produce a smile on exercise-related stimuli predicted more exercise episodes per week).
The authors would like to emphasize that this was only the second study that attempted to approach the affective valence of the automatic valuation of exercise in an FE task. The inconclusive results here underscore the previously reported result that this FE task might need further refinement (see Brand & Ulrich, 2019). These authors also had to exclude 31% of their data due to incorrect facial responses by the participants, and they concluded that this emotional Stroop task might be too demanding for many people. In the current study, the authors had to exclude 18% of the participants for the same reason. Other tests (for example, the AMP; Payne et al., 2005) may be more appropriate for future measurements of the affective valence of study participants’ automatic valuation of exercise.
HRV reactivity and affective valence of the automatic valuation
Although this result should not be overinterpreted due to the difficulties of measuring affective valence with the described FE task, it should be noted that the somatic core of the affective valuation (HRV reactivity) and affective valence (smile and frown latency scores) were uncorrelated. In the authors’ view, this suggests that the two indicators indeed represent two different aspects of the automatic affective valuation of exercise; see Schinkoeth et al. (2019), who reported a similar result that valence ratings with the self-assessment manikin test and HRV reactivity were also uncorrelated. Whereas HRV reactivity might be indicative of (peripheral) psychophysiological activation due to cortical and subcortical processes, the valence of the core affective feeling associated with the affective valuation might be completely independent of it; see Kreibig (2010) for results indicating that affective feelings can be reported without concomitant autonomic changes.
Study limitations and recommendations for future studies
The number of cases with technical malfunctions was high in this study. As already noted, the FE task in particular might require further refinement or should be replaced by another test. The final sample consisted of data of only 69 participants. This small sample size may have affected statistical power and the models’ fit indices in the path analyses. Small sample sizes can lead to higher χ2 bias and RMSEAs (Jackson, 2003). In future studies, the findings here should be replicated using larger samples. Furthermore, exercise behavior was assessed by self-report. It is known that individuals tend to overrate their exercise behavior in self-assessments of this kind (Duncan, Sydeman, Perri, Limacher, & Martin, 2001). Future studies, especially those that will then focus even more on the links between type‑1 processes and exercise behavior, might employ a more objective test of exercise activity, for example, by using physical activity accelerometers. Last but not least, the participants in the present study were asked about their past exercise behavior. The ART, however, makes assumptions about the mental processes at the very moment when an exercise-related decision takes place. To stay even closer to the assumption of the ART, future studies might apply designs such that the automatic affective processes can be measured shortly before or right at the moment of an exercise-related decision.
Conclusion
This study showed that negative automatic associations with exercise were linked with a somatic reaction (increase in HRV) that was triggered by watching exercise-related pictures. The authors suggested understanding this reaction as one somatic aspect of what has been defined as the affective valuation of exercise in the ART (Brand & Ekkekakis, 2018). In their view, this study represents a first but successful attempt to disentangle the two components of the ART’s type‑1 process. The findings must not be overinterpreted as confirmation of the ART per se. It has been repeatedly pointed out (e.g., Brand & Cheval, 2019; Rhodes, McEwan, & Rebar, 2019) that a greater variety of theoretical and methodological approaches would be necessary to further develop our current understanding of exercise motivation. The authors hope that the findings presented here represent progress in this direction. Furthermore, through the combination of two reaction time-based measures with different response modalities, one physiological measure and self-reports, the authors have tried to avoid common-method bias. Therefore, the study might also offer an outlook on an exercise psychology that no longer predominantly relies on self-report data to examine its constructs.
Notes
Due to the smaller number of items, the reliability in single-target IATs is typically lower than in standard IATs. Cronbach-alpha coefficients for internal consistency as a measure of the reliability of IATs are usually significantly higher, and are occasionally reported in publications instead of the split-half reliability coefficient. It must be noted however that items (i.e., participants’ test responses) in the two IAT conditions (usually: test blocks) are not expected to covary so that internal consistency scores are inappropriate measures for determining the reliability of an IAT score.
References
Antoniewicz, F., & Brand, R. (2014). Automatic evaluations and exercise setting preference in frequent exercisers. Journal of Sport and Exercise Psychology, 36, 631–636. https://doi.org/10.1123/jsep.2014-0033.
Antoniewicz, F., & Brand, R. (2016). Learning to like exercising: evaluative conditioning changes automatic evaluations of exercising and influences subsequent exercising behavior. Journal of Sport and Exercise Psychology, 38, 138–148. https://doi.org/10.1123/jsep.2015-0125.
Appelhans, B. M., & Luecken, L. J. (2006). Heart rate variability as an index of regulated emotional responding. Review of General Psychology, 10(3), 229–240. https://doi.org/10.1037/1089-2680.10.3.229.
Berntson, G. G. (2019). Presidential address 2011: automatic modes of control and health. Psychophysiology, 56(1), 1–10. https://doi.org/10.1111/psyp.13306.
Berntson, G. G., Cacioppo, J. T., & Quigley, K. S. (1991). Autonomic determinism: the modes of autonomic control, the doctrine of autonomic space, and the laws of autonomic constraint. Psychological Review, 98(4), 459–487. https://doi.org/10.1037/0033-295X.98.4.459.
Bluemke, M., & Friese, M. (2008). Reliability and validity of the Single-Target IAT (ST-IAT): assessing automatic affect towards multiple attitude objects. European Journal of Social Psychology, 38, 977–997. https://doi.org/10.1002/ejsp.487.
Bradley, M. M., & Lang, P. J. (1994). Measuring emotion: the self-assessment manikin and the semantic differential. Journal of Behavior Therapy and Experimental Psychiatry, 25(1), 49–59. https://doi.org/10.1016/0005-7916(94)90063-9.
Brand, R., & Cheval, B. (2019). Theories to explain exercise motivation and physical inactivity: ways of expanding our current theoretical perspective. Frontiers in Psychology, 10, 1147. https://doi.org/10.3389/fpsyg.2019.01147.
Brand, R., & Ekkekakis, P. (2018). Affective–reflective theory of physical inactivity and exercise. German Journal of Exercise and Sport Research, 48(1), 48–58. https://doi.org/10.1007/s12662-017-0477-9.
Brand, R., & Schweizer, G. (2015). Going to the gym or to the movies? Situated decisions as a functional link connecting reflective and automatic evaluations of exercise with exercising behavior. Journal of Sport and Exercise Psychology, 37(1), 63–73. https://doi.org/10.1123/jsep.2014–0018.
Brand, R., & Ulrich, L. (2019). I can see it in your face. Affective valuation of exercise in more or less physically active individuals. Frontiers in Psychology, 10, 2901. https://doi.org/10.3389/fpsyg.2019.02901.
Brand, R., & Utesch, T. (2019). Entwicklung eines robusten, Recoding-free Single Target Implicit Association Tests’ (RF-ST-IAT) [Abstract]. In A. Arampatzis, S. Braun, K. Schmitt & B. Wolfarth (Eds.), Sport im Öffentlichen Raum. Schriften der Deutschen Vereinigung für Sportwissenschaften, (Vol. 282, p. 313). Hamburg: Feldhaus.
Brouwer, A. M., van Wouwe, N., Mühl, C., van Erp, J., & Toet, A. (2013). Perceiving blocks of emotional pictures and sounds: effects on physiological variables. Frontiers in Human Neurosciences, 7, 1–10. https://doi.org/10.3389/fnhum.2013.00295.
Butler, E. A., Wilhelm, F. H., & Gross, J. J. (2006). Respiratory sinus arrhythmia, emotion, and emotion regulation during social interaction. Psychophysiology, 43, 612–622.
Cacioppo, J. T., Berntson, G. G., Larsen, J. T., Poehlmann, K. M., & Ito, T. A. (2000). The psychophysiology of emotion. In M. Lewis & J. M. Haviland-Jones (Eds.), Handbook of emotions (2nd edn., pp. 173–191). New York: Guilford.
Cheval, B., Radel, R., Neva, J. L., Boyd, L. A., Swinnen, S. P., Sander, D., et al. (2018). Behavioral and neural evidence of the rewarding value of exercise behaviors: a systematic review. Sports Medicine. https://doi.org/10.1007/s40279-018-0898-0.
Chevance, G., Bernard, P., Chamberland, P. E., & Rebar, A. (2019). The association between implicit attitudes toward physical activity and physical activity behaviour: a systematic review and correlational meta-analysis. Health Psychology Review, 13(3), 248–276. https://doi.org/10.1080/17437199.2019.1618726.
Conroy, D. E., & Berry, T. R. (2017). Automatic affective evaluations of physical activity. Exercise and Sport Sciences Reviews, 45, 230–237. https://doi.org/10.1249/JES.0000000000000120.
Damasio, A. R. (1996). The somatic marker hypothesis and the possible functions of the prefrontal cortex. Philosophical Transactions of the Royal Society London (Biological Sciences; Ser. B 351), 351, 1413–1420. https://doi.org/10.1098/rstb.1996.0125.
Duncan, G. E., Sydeman, S. J., Perri, M. G., Limacher, M. C., & Martin, A. D. (2001). Can sedentary adults accurately recall the intensity of their physical activity? Preventive Medicine, 33(1), 18–26. https://doi.org/10.1006/pmed.2001.0847.
Ekkekakis, P., & Brand, R. (2019). Affective responses to and automatic affective valuations of physical activity: fifty years of progress on the seminal question in exercise psychology. Psychology of Sport and Exercise, 42, 130–137. https://doi.org/10.1016/j.psychsport.2018.12.018.
Ekkekakis, P., Zenko, Z., Ladwig, M. A., & Hartman, M. E. (2018). Affect as a potential determinant of physical activity and exercise: critical appraisal of an emerging research field. In D. M. Williams, R. E. Rhodes & M. Conner (Eds.), Affective determinants of health behavior (pp. 237–261). New York: Oxford University Press.
Evans, J. S., & Stanovich, K. E. (2013). Dual-process theories of higher cognition: advancing the debate. Perspectives on Psychological Science, 8, 223–241. https://doi.org/10.1177/1745691612460685.
Eves, F. F., Scott, E. J., Hoppé, R., & French, D. P. (2007). Using the affective priming paradigm to explore the attitudes underlying walking behaviour. British Journal Of Health Psychology, 12, 571–585. https://doi.org/10.1348/135910706X153775.
Gawronski, B., & De Houwer, J. (2014). Implicit measures in social and personality psychology. In H. T. Reis & C. M. Judd (Eds.), Handbook of research methods in social and personality psychology (2nd edn., pp. 283–310). New York: Cambridge University Press.
Goodboy, A. K., & Kline, R. B. (2017). Statistical and practical concerns with published communication research featuring structural equation modeling. Communication Research Reports, 34(1), 66–77. https://doi.org/10.1080/08824096.2016.1214121.
Greenwald, A. G., McGee, D. E., & Schwartz, J. L. (1998). Measuring individual differences in implicit cognition: the implicit association test. Journal of Personality and Social Psychology, 74(6), 1464–1480. https://doi.org/10.1037/0022-3514.74.6.1464.
Greenwald, A. G., Poehlman, T. A., Uhlmann, E. L., & Banaji, M. R. (2009). Understanding and using the implicit association test: II. meta-analysis of predictive validity. Journal of Personality and Social Psychology, 97(1), 17–41. https://doi.org/10.1037/a0015575.
Guthold, R., Stevens, G. A., Riley, L. M., & Bull, F. C. (2018). Worldwide trends in insufficient physical activity from 2001 to 2016: a pooled analysis of 358 population-based surveys with 1·9 million participants. The Lancet Global Health, 6(10), e1077–e1086. https://doi.org/10.1016/S2214-109X(18)30454-6.
Hagströmer, M., Oja, P., & Sjöström, M. (2006). The international physical activity questionnaire (IPAQ): a study of concurrent and construct validity. Public Health Nutrition, 9(6), 755–762. https://doi.org/10.1079/PHN2005898.
Hofmann, W., Gawronski, B., Gschwendner, T., Le, H., & Schmitt, M. (2005). A meta-analysis on the correlation between the implicit association test and explicit self-report measures. Personality and Social Psychology Bulletin, 31(10), 1369–1385. https://doi.org/10.1177/0146167205275613.
Holzman, J. B., & Bridgett, D. J. (2017). Heart rate variability indices as bio-markers of top-down self-regulatory mechanisms: a meta-analytic review. Neuroscience & Biobehavioral Reviews, 74, 233–255. https://doi.org/10.1016/j.neubiorev.2016.12.032.
Jackson, D. L. (2003). Revisiting sample size and number of parameter estimates: some support for the N:q hypothesis. Structural Equation Modeling, 10, 128–141. https://doi.org/10.1207/S15328007SEM1001_6.
Jennings, J. R. (1986). Bodily changes during attention. In M. G. H. Coles, E. Donchin & S. W. Porges (Eds.), Psychophysiology: Systems, processes, and applications (pp. 268–289). New York: Guilford.
Kline, R. B. (2015). Principles and practice of structural equation modeling (4th edn.). New York: Guilford.
Koval, P., Ogrinz, B., Kuppens, P., Van den Bergh, O., Tuerlinckx, F., & Sütterlin, S. (2013). Affective instability in daily life is predicted by resting heart rate variability. PLoS One, 8(11), e81536. https://doi.org/10.1371/journal.pone.0081536.
Kreibig, S. D. (2010). Autonomic nervous system activity in emotion: a review. Biological Psychology, 84, 394–421. https://doi.org/10.1016/j.biopsycho.2010.03.010.
Kulke, L., Feyerabend, D., & Schacht, A. (2018). A comparison of the affectiva imotions facial expression analysis software with EMG for identifying facial expressions of emotion. https://doi.org/10.31234/osf.io/6c58y.
Laborde, S., Mosley, E., & Thayer, J. F. (2017). Heart rate variability and cardiac vagal tone in psychophysiological research–recommendations for experiment planning, data analysis, and data reporting. Frontiers in Psychology, 8, 213. https://doi.org/10.3389/fpsyg.2017.00213.
Lang, P. J., Bradley, M. M., & Cuthbert, B. N. (1997). International affective picture system (IAPS): technical manual and affective ratings. Gainesville: University of Florida, Center for Research in Psychophysiology.
Lang, P. J., Greenwald, M. K., Bradley, M. M., & Hamm, A. O. (1993). Looking at pictures: affective, facial, visceral, and behavioral reactions. Psychophysiology, 30, 261–273. https://doi.org/10.1111/j.1469-8986.1993.tb03352.x.
McDuff, D., Mahmoud, A., Mavadati, M., Amr, M., Turcot, J., & Kaliouby, R. E. (2016). AFFDEX SDK: A cross-platform real-time multi-face expression recognition toolkit. In Proceedings of the 2016 CHI conference extended abstracts on human factors in computing systems (pp. 3723–3726). New York: ACM. https://doi.org/10.1145/2851581.2890247.
Payne, B. K., Cheng, C. M., Govorun, O., & Stewart, B. D. (2005). An inkblot for attitudes: affect misattribution as implicit measurement. Journal of Personality and Social Psychology, 89, 277–293. https://doi.org/10.1037/0022-3514.89.3.277.
Podsakoff, P. M., MacKenzie, S. B., Lee, J., & Podsakoff, N. P. (2003). Common method bias in behavioral research: a critical review of the literature and recommended remedies. Journal of Applied Psychology, 5, 879–903. https://doi.org/10.1037/0021-9010.88.5.879.
Pratto, F., & John, O. P. (1991). Automatic vigilance: the attention-grabbing power of negative social information. Journal of Personality and Social Psychology, 61, 380–391. https://doi.org/10.1037/0022-3514.61.3.380.
Raccuia, L. (2016). Single-target implicit association tests (ST-IAT) predict voting behavior of decided and undecided voters in Swiss referendums. PLoS ONE, 11(10), e163872. https://doi.org/10.1371/journal.pone.0163872.
R Core Team (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Rebar, A. L., Schoeppe, S., Alley, S. J., Short, C. E., Dimmock, J. A., Jackson, B., et al. (2016). Automatic evaluation stimuli: the most frequently used words to describe physical activity and the pleasantness of physical activity. Frontiers in Psychology, 7, 1277. https://doi.org/10.3389/fpsyg.2016.01277.
Rhodes, R. E., Janssen, I., Bredin, S. S. D., Warburton, D. E. R., & Bauman, A. (2017). Physical activity: health impact, prevalence, correlates and interventions. Journal of Psychology and Health, 32(8), 942–975. https://doi.org/10.1080/08870446.2017.1325486.
Rhodes, R. E., McEwan, D., & Rebar, A. L. (2019). Theories of physical activity and behavior change: a history and synthesis of approaches. Psychology of Sport and Exercise, 42, 100–109. https://doi.org/10.1016/j.psychsport.2018.11.010.
Richetin, J., Costantini, G., Perugini, M., & Schönbrodt, F. (2015). Should we stop looking for a better scoring algorithm for handling Implicit Association Test data? Test of the role of errors, extreme latencies treatment, scoring formula, and practice trials on reliability and validity. PLoS One, 10(6), e129601. https://doi.org/10.1371/journal.pone.0129601.
Rosseel, Y. (2012). lavaan: An R Package for Structural Equation Modeling. Journal of Statistical Software, 48(2), 1–36. http://www.jstatsoft.org/v48/i02/
Rothermund, K., Teige-Mocigemba, S., Gast, A., & Wentura, D. (2009). Minimizing the influence of recoding in the implicit association test: the recoding-free implicit association test (IAT-RF). The Quarterly Journal of Experimental Psychology, 62(1), 84–98. https://doi.org/10.1080/17470210701822975.
Russell, J. A. (1980). A circumplex model of affect. Journal of Personality and Social Psychology, 39, 1161. https://doi.org/10.1037/h0077714.
Russell, J. A. (2003). Core affect and the psychological construction of emotion. Psychological Review, 110(1), 145–172.
Russell, J. A., & Feldman Barrett, L. (2009). Core affect. In D. Sander & K. R. Scherer (Eds.), The Oxford companion to emotion and the affective sciences (p. 104). New York: Oxford University Press.
Schimmack, U. (2019). The implicit association test: a method in search of a construct. Perspectives on Psychological Sciences. https://doi.org/10.1177/1745691619863798.
Schinkoeth, M., & Antoniewicz, F. (2017). Automatic evaluations and exercising: systematic review and implications for future research. Frontiers in Psychology, 8, 2103. https://doi.org/10.3389/fpsyg.2017.02103.
Schinkoeth, M., Weymar, M., & Brand, R. (2019). Listening to the heart. Getting closer to the somatic core of affective valuation of exercise through heart rate variability analysis. Psychology of Sport and Exercise, 45, 101541. https://doi.org/10.1016/j.psychsport.2019.101541.
Shaffer, F., & Ginsberg, J. P. (2017). An overview of heart rate variability metrics and norms. Frontiers in Public Health, 5, 258. https://doi.org/10.3389/fpubh.2017.00258.
Simon, H. A. (1986). Rationality in psychology and economics. The Journal of Business, 59(4, Part 2), 209–224.
Thayer, J. F., & Lane, R. D. (2000). A model of neurovisceral integration in emotion regulation and dysregulation. Journal of Affective Disorders, 61(3), 201–216. https://doi.org/10.1016/S0165-0327(00)00338-4.
Thayer, J. F., Åhs, F., Fredrikson, M., Sollers 3rd, J. J., & Wager, T. D. (2012). A meta-analysis of heart rate variability and neuroimaging studies: implications for heart rate variability as a marker of stress and health. Neuroscience and Behavioral Reviews, 36(2), 747–756. https://doi.org/10.1016/j.neubiorev.2011.11.009.
Timme, S., & Brand, R. (2020). Affect and exertion during incremental physical exercise: examining changes using automated facial action analysis and experiential self-report. PLoS ONE, 15(2), e228739. https://doi.org/10.1371/journal.pone.0228739.
Warburton, D. E. R., Nicol, C. W., & Bredin, S. S. D. (2006). Health benefits of physical activity: the evidence. Canadian Medical Association Journal, 174(6), 801–809. https://doi.org/10.1503/cmaj.051351.
WHO (2010). Global recommendations on physical activity for health. Geneva: World Health Organization.
Wu, Y., Gu, R., Yang, Q., & Luo, Y.-J. (2019). How do amusement, anger and fear influence heart rate and heart rate variability? Frontiers of Neuroscience, 13, 1131. https://doi.org/10.3389/fnins.2019.01131.
Zenko, Z., & Ekkekakis, P. (2019). Critical review of measurement practices in the study of automatic associations of sedentary behavior, physical activity, and exercise. Journal of Sport and Exercise Psychology, 41(5), 271–288. https://doi.org/10.1123/jsep.2017-0349.
Funding
Open Access funding provided by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
MS conceived the study design with the help of RB. MS performed the statistical analyses and wrote the first draft of the manuscript. RB revised the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
M. Schinkoeth and R. Brand declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
For this article, no studies with animals were performed by any of the authors. All studies performed with human participants were in accordance with the ethical standards indicated in each case.
Rights and permissions
Open Access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schinkoeth, M., Brand, R. Automatic associations and the affective valuation of exercise: disentangling the type-1 process of the affective–reflective theory of physical inactivity and exercise. Ger J Exerc Sport Res 50, 366–376 (2020). https://doi.org/10.1007/s12662-020-00664-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12662-020-00664-9