Abstract
Purpose
Objective of this study is the valorisation of mass rearing waste of fruit flies from sterile insect technique facilities by black soldier flies into high quality and quantity products as a sustainable utilisation concept.
Methods
Different ratios of medfly rearing waste in hen feed were used for rearing black solder fly (BSF) larvae. The growth rate and insect survival were determined. Moreover, the mass gains were determined and bioconversion rates were assessed. Furthermore, the nutritional compositions of BSF pupae were analysed to evaluate the product quality.
Results
The omnivorous BSF larvae were highly efficient at converting digested waste into body mass as compared to the control treatment (hen feed). High weight of pre-pupae was recorded for all experimental diets. The efficacy of larvae to reach pupae ranged from 92.5 to 98.5%, while the efficacy of pupae to reach adults ranged from 81.7 to 89.0%. All experimental diets exhibited high rates of prepupal weight, metabolism and efficiency of conversion of digested food. A nutritional analysis revealed that the protein and fat contents were high, while the bioaccumulation of heavy metals was low,
Conclusion
Medfly rearing waste is a potential feed ingredient for the production of BSF pre-pupae and could be applied to valorise this rearing waste into high-value feed.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Statement of Novelty
With the amassing of insect rearing waste due to the sterile insect technique for biological insect control, efficient valorisation concepts for this nutrient-rich substrate are urgently needed. While it has been shown that the omnivorous black soldier fly Hermetia illucens has the potential to convert and valorise food and other organic wastes including manure and faecal sludge, valorising insect rearing waste by black soldier fly is a novel approach. It was shown that black soldier fly could be reared on experimental diets containing medfly rearing waste and that it has potential for its valorisation into valuable feed.
Introduction
The black soldier fly (BSF), Hermetia illucens (L., 1758), is one of the beneficial edible insects that has been considered to be native in almost 80% of the world between latitudes in particular tropical and sub-tropical areas. It is indigenous to many regions, including America, Europe, India, Asia, Australia, and Africa [1, 2]. BSF larvae have a high potential for organic waste management and have been promising for bio-waste processing e.g., in landscaping in the recent decade [3]. Because they are polyphagous and contain a high quality of nutrients, BSF are excellent candidates for a range of applications [4]. In particular, BSF have the potential to contribute to solving two of modern agriculture's challenges: serving as an alternative protein source for animal feed and managing organic waste [5]. Moreover, BSF could provide novel antimicrobial compounds [6] and a source of chitin [7]. Furthermore, BSF have been proposed as a promising species for use in the entomo-remediation of heavy metal-polluted organic biomass [8, 9]. Generally, BSF waste is non-hazardous to the environment, moreover it has the potential to be an organic fertilizer for promoting plant growth because it contains several essential plant nutrients [10, 11], and it was also proposed as an interesting substrate for biogas generation due to its high biomethane potential [2]. It was shown that the BSF has the potential to convert various agricultural by-products associated with a significant reduction in organic matter volumes, such as coffee. [12], manure [13] and palm kernel meal [14] or organic waste materials like market waste, municipal organic waste [15] or dewatered faecal sludge [16]. These different organic wastes can be transformed by BSF larvae into high valuable nutrients [17]. Evidently, organic waste has been managed by BSF on a large scale in many regions around the world [18] Few studies have focused on the utilization of mass rearing insect waste from various species and application programs, particularly waste from fruit fly mass rearing facilities [19]. The use of cricket and locust rearing waste as a feed substrate for BSF with the goal to establish a circular food economy and improve the efficiency of insect farms has been investigated by Jucker et al. [20]. Moreover, BSF larvae had successfully converted silkworm pupae, a by-product of the sericulture industry, into safe and highly nutritious products [21]. They showed that cricket and locust as well as silkworm rearing waste could be used as feed substrate for BSF. This implies that the rearing waste of fruit flies produced from the insect facilities of sterile insect technique (SIT) programmes could also be recycled by BSF. SIT involves releasing high numbers of sterile flies in nature to induce sterile mating in a natural population that reduces its reproduction to a level below population maintenance. In order to achieve this, the production of sterile flies in sufficient numbers and of adequate quality is one of the principal requirements for the success of this technique. SIT programmes of Mediterranean fruit flies Ceratitis capitata (Wiedemann) (Medfly) are widely used in many countries around the world in different facilities, producing e.g., 6 million sterile males/week in South Africa [22], 130 million sterile males/week in Morocco [23] and 200 million sterile male per day in Guatemala [24]. The SIT programme in Queensland, Australia, producing the fruit fly, Bactrocera tryoni, was reported to result in the disposal of tonnes of rearing waste each year. However, the crude protein and fat contents were increased in the spent media as compared to fresh one which may due to the presence of larvae residues [19]. Accordingly, the valorisation of organic waste from rearing these large numbers of insects is a prerequisite that requires a sustainable solution. Attempt has been done to use the medfly rearing waste as ruminant feed whereas low levels of tannin and phenolic, good nutritional constitutes and degrades quickly [25]. However, a novel concept for the valorisation of fruit fly rearing waste is desperately needed. The purpose of our study was to determine the potential of BSF for the conversion of waste of Medfly rearing facilities and to assess the efficacy of this BSF-assisted waste management, while considering the nutritional benefits of BSF.
Material and Methods
Insect Rearing
BSF were reared and maintained under laboratory conditions at 27 ± 2 °C and 65% R. H. Full grown pupae were supplied in the adult rearing screen cages (2 m × 2 m × 2 m) installed in an adult rearing room with a 14 L: 10 D photoperiod. The cages were provided by plastic cups containing pieces of cotton wool soaked with 10% sugar for drinking the emerged flies. Attractant substrate comprised of different organic waste that was placed inside the cage. Strips of wood were provided near the substrates to provide egg laying sites for the flies. The egg masses were collected daily and transferred to plastic containers (70 × 30 × 15 cm3) containing a medium comprised of hen feed (90% humidity) for feeding the newly hatched larvae. The containers were covered with muslin cloths for 5 days. Then, the young larvae were transferred to the larval rearing cages until pupation.
Feed Substrate
The hen feed was purchased from the local market. While medfly rearing waste was obtained from the mass rearing of Mediterranean fruit fly Ceratits capitate. The rearing diet composition per 1 kg diet was 300 g wheat bran as bulking agent, 70 g yeast, 130 g sucrose, 2 g sodium benzoate, 15 ml HCl and 500 ml water. The residual diet was collected after the last instar larvae (third instar) pupates within seven to eight days inside sand below the rearing trays. The larval rearing waste was collected which consists of wheat brain, sugar residue, yeast residue and residues of medfly larvae (dead larvae, molting residues, un-hatched eggs and residue of larval feed).
The waste was oven-dried at 40 °C for 24 h and then irradiated with a dose rate of 800 Gy using Cobalt 60 gamma cell at a dose rate of 1.0 Gy/min. Irradiation treatment is an effective phytosanitary treatment against different pathogens or unfavorable pests that could infect the waste with minimal adverse effects on the quality of most fresh products [26]. The dried waste was stored at room temperature until use.
The experimental diets were prepared using hen feed as a control diet and different ratios of medfly rearing waste as described in Table 1.
Bioassay
BSF larvae were fed on different diets at feeding rate average of 100 mg/larvae/day. The larvae were kept inside containers (20 × 10 × 15 cm), wrapped with a black plastic sheet at side and lid. Ten holes, with size of 15 mm each, were punctured at the lid to provide oxygen. For six days, 1.4 g larvae (about 200–220 larvae) were kept in the container. All treatments were carried out in triplicate and were repeated in two times. The daily amount of feed was adapted in accordance to the number of remaining larvae in each container. Every 3 days, the larvae were transferred to new containers filled with the appropriate amount of feed, which was prepared on the same day. At each transfer, the weight of the larvae was measured. Resultant pre-pupae from each treatment were collected daily, counted, weighted and stored in waxed paper cups on moist sawdust and covered with muslin cloth until pupation. The pupae were collected daily and placed inside plastic cups until adult emergence. The emerged flies were placed inside wooden cages (30 × 30 × 30 cm) to calculate the adult survival in each treatment. The larval growth, developmental time to pre-pupation, pupation, emergence and longevity were measured.
Larval growth was measured by biomass gain of larvae until developed into pre-pupae and expressed in growth rate (GR) following the Eq. 1:
The bioconversion rate (BR) was calculated using Eq. 2 [27], which is the ratio of larval weight gain (W) to the total amount of feed offered (F) minus the residue in the experimental container (R) (larval frass).
Feed reduction efficiency was measured by the reduction index (RI) (Eq. 3) [15] and was calculated using the overall degradation D (see Eq. 4) per duration t (in days) of larvae fed on the substrate.
The overall degradation D was calculated by the total feed offered (F) without the residue in the experimental container (R) divided by the total feed offered (F).
The amount of feed consumed that was used as energy by the larvae to maintain body homoeostatic balance is referred to Metabolism (M) that was calculated using Eq. 5 [28]. It is defined as ingested food that is calculated by the difference of the total feed offered (F) and the residue in the experimental container (R) (larval frass) divided by pre-pupal weight gain (PW).
The conversion efficiency of ingested food was determined by conversion efficiency index (ECI) that refers to the ratio of pre-pupal weight gain to the amount of ingested food and calculated using Eq. 6 [29]
The approximate digestibility (AD) is the difference between the weight of offered feed (F) and the residue of the offered feed (R) divided by the residue R and calculated using Eq. 7 [30, 31].
The ability of larvae to convert the waste into their body mass was calculated by efficiency of conversion of digested food (ECD) using Eq. 8 [32, 33].
Biochemical Analysis
Proximate analyses (Protein, fat, ash and carbohydrate) of Diet-H and Diet-W were conducted by the methods of AOAC [34]using 100 mg dry matter, respectively. The protein and fat contents of the experimental Pupae-H and Pupae-W were determined using 50 mg soft tissues from respective pupae. The samples were homogenised in 1 ml distilled water and centrifuged at 8000 rpm for 15 min at 2 °C using a refrigerated centrifuge; the resulted supernatants were stored at 3 °C to use for biochemical analysis for a few days (one week maximum). All experiments were conducted in triplicate and were repeated in two times. The crude protein content was analysed according to Horwitz [35], lipids were extracted and purified according to Christie [36]. The Ash and carbohydrate contents were analysed according to (AC, 1995). The nitrogen, carbon and trace elements contents were analysed by an Energy Dispersive X-ray analyzer (EDX, Jeol, Japan) supplied by a scanning electron microscope model HEOL-JSM 4500 (Jeol, Japan).
The bioaccumulation of respective elements was assessed by the bioaccumulation factor (BF) according to Eq. 9 [37].
where CP is the concentrations of elements in BSF pupae and CF is the concentration of elements in the feed.
Data Analysis
Bioassay and biochemical analyses were evaluated statistically by one way analysis of variance (ANOVA) technique. Duncan multiple range test was used to analyze the means. The ANOVA statistics were defined as significant [38] when P < 0.01. The means and standard errors of feed performance, larval development, and longevtiy were calculated from original data. The data of pupation, adult emergence, bioconversion, and digested efficiency were transformed by arcsine tables.
Results and Discussion
Insect Development and Growth
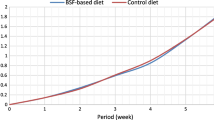
The mass yields of BSF larvae varied during their life span by different experimental diets (Fig. 1). At 9 days old, the mass yield of larvae fed on Diet-H75 was not significantly higher (16.8 ± 0.5 g) than fed on Diet-H50 and Diet-H (14.8 ± 0.7 and 14.75 ± 1.2 g), respectively, while it was significantly higher as compared to larvae fed on Diet-H25 and Diet-W (13 ± 1.2 and 12.2 ± 1.2 g), respectively (F(4,29) = 3.5, P = 0.02). The same trend was observed for the mass larval weight at 12 days old where the highest yield was recoded at the Diet-H75 (35.0 ± 1.0 g) as compared to a yield of 31.0 ± 2.4, 30.9 ± 0.6, 27.3 ± 2.5, 25.7 ± 2.4 g of Diet-H50 and Diet-H, Diet-H25 and Diet-W, respectively (F(4,29) = 3.4, P = 0.03). At 15 days old, the average of mass yield of larvae fed on Diet-H75 was significantly higher (39.7 ± 1.3 g) than fed on Diet-H50 and Diet-H and Diet-W (36.4 ± 1.4, 35.6 ± 1.2 and 34.2 ± 1.2 g), respectively (F(4,29) = 3.6, P = 0.01), respectively, while, it was not significantly higher than fed on Diet-H25 (36.8 ± 0.5). Additionally, the larval biomass was greatly increased during the sigmoidal growth phase from 6 to 12 days old as compared to 12 to 15 days. Our findings inferred that the BSF larvae were capable to metabolise medfly-rearing waste into high mass yield as compared to the control treatment (hen feed).
The duration of BSF larvae fed on Diet-W to reach pre-pupal stage was significantly prolonged (18.3 ± 0.2 days) as compared to Diet-H75 (16.6 ± 0.3), while, the difference was not significant as compared to Diet-H, Diet-H50 and Diet-H25 (17.9 ± 0.8, 17.6 ± 0.5 and 17.3 ± 0.6 days), respectively (F(4,29) = 3.0, P = 0.04) (Fig. 2). Moreover, the adult longevity produced on Diet-W was significantly lower (9.3 ± days) than on Diet-H75, Diet-H50 (10.2 ± 0.3 and 10.1 ± 0.4), respectively (F(4,29) = 6.4, P = 0.01). Our obtained results indicated that the larvae developed with 16.3–18.3 days faster than on dairy manure (28–30 days) [39] and on meat meal (33 days) [40]. Furthermore, the results of adult longevity (9.3–10.2 days) are in accordance with Bertinetti et al. [41] who determined a longevity of 8–11 days which was increased by 1 day by feeding adults on milk. This is contradictory to findings by Nakamura et al. [42] who found a longevity of 21–37 days and by Tomberlin et al. [43] who reported 14–17 days for adult longevity.
Figure 3 shows the pupal percentages derived from the larvae fed different experimental diets. The percentages of pupation in Diet-H (98.5%) were significantly higher as compared to of Diet-H75, Diet-H50, Diet-H25 and Diet-W (96.6, 94.7, 94.0 and 92.5%); (F = 9.7, P = 0.002), respectively. Similarly, the adult emergence after having fed on Diet-H was with 89.0% significantly the highest as compared to all other diets (F(4,29) = 12.7, P = 0.001) and the percentages decreased with increasing percentages of med fly rearing waste in the diet. It has been proposed that different diets during larval stage caused differences in larval and pupal mortalities, which also affected pupation and adult emergence, respectively [44,45,46]. The efficacy of larvae to reach pupae after being fed on medfly rearing waste (Diet-W) was in the current study with 92.5% higher as compared to 71.0–84.0% on dairy manure (Myer et al. 2008), 82.2% on chicken manure [31], and 40% on meat meal [40]. Moreover, the adult emergence of the present study was with 81.7–89.0% lower compared to 93.0–97.0% as determined by Holmes et al. [47]. However, adult emergence may be influenced by other factors as well such as moisture and type of pupation substrates [48, 49]. The BSF pupae in our study were not subjected to substrates or moisture.
Insect Performance and Bioconversion
Table 2 shows the weight averages of larvae (16–17 days old, mg/larvae). It was shown that the larval weight produced from Diet-H75 was significantly higher than from Diet-H, Diet-H50 and Diet-H25, while there was no significant difference as compared to Diet-W (F(4,29) = 2.6, P = 0.06). Similar results were recorded for the growth rate (mg/day). The growth rate of larvae fed on Diet-H75 was significantly higher than of larvae fed on Diet-H, Diet-H25 and Diet–W, while there was no significant difference as compared to on Diet-H50 (F(4,29) = 2.4, P = 0.08). Our results of larval weight (157.6–185.6 mg/larvae) and growth rate (2.8–3.6 mg/day) were in line with previous studies on different feed substrates where larval weight and growth rate ranged from 154–271 mg/larva and 2.3–3.7 mg/day, respectively [50,51,52]. The average weight of pre-pupae ranged from 116.2 ± 1.0–156.7 ± 7.1 mg/pupa) and it was clearly lower than larval weight. The lower pupal weight is due to the fact that the larvae stop feeding and rely on their own biomass to meet their energy requirements to develop into pupae [53, 54]. Data showed that the average pre-pupal weight was significantly higher for Diet-H75 as compared to the other diets (F(4,29) = 21.4, P < 0.001) (Table 2). The obtained pre-pupal weights in our study were high compared with previous findings in other studies on different diets [15, 55, 56]. The high weight of pre-pupae has a positive impact on the biological traits related to reproduction of adult flies that could increase the BSF mass rearing quality [40, 57].
As also shown in Table 2, the bioconversion rates (BR) of the larvae produced from Diet-H75 and Diet-H25 were significantly higher than Diet-H, Diet-H50 and Diet-W (F(4,29) = 4.7, P = 0.007). The BR ranged from 13.5 to 16.8%. These rates were higher in comparison to rates that had been found for BSF rearing on other types of organic waste [27, 53]. Data also showed that the bioconversion of medfly waste by BSF was high as compared to hen feed, and could be considered as a BSF substrate in the BSF industrial process. However, it was lower than the 16.0–22.0% reported by Banks et al. [56] for fresh human faeces. The high BRof Diet-W (14.3%) that has 100% medfly waste in our current study indicated that BSF larvae were converting the medfly rearing waste effectively. Results indicated that the combinations diets were highly efficient to be converted to body mass by BSF and the combinations of the medfly waste could make more suitable diets [30]. The diet efficiency of an insect species could be determined by its feed conversion rates. By-products with a lower monetary value than the main product are thought to have a lower environmental impact when considering economic allocation [31].
The reduction index (RI) ranged from 7.0 ± 0.1 to 8.8 ± 0.2%. The RI for Diet-W was significantly lower than for the other experimental diets, while the highest reduction index was recorded for Diet-H (F(4,29) = 17.8, P < 0.001). These results are in line with those found by [20] who found that the reduction index of BSF larvae fed on cricket and locust wastes were lower than the standard diet. Data of RI presented high rates of medfly waste reduction which were similar to other organic waste studied by [20, 27], these data indicated that it is possible to use BSF to reduce the amount of medfly waste. The data revealed that the BSF larvae were able to consume the medfly rearing waste without negative impact on feeding efficiencies, however, significant differences in bioconversion and feed reduction were observed between the diets containing waste in different ratios and the control diet.
The metabolized feed to energy (M) by BSF larvae ranged from 53.7 to 68.8% and it was significantly higher for Diet-H than the other experimental diets (F(4,29) = 13.7, P < 0.001).The obtained results of M were lower than the 77.3% reported by Mahmoud et al. for household biowaste [18]. However, the results are comparable with Gligorescu et al. who mentioned that BSF larvae can prioritise their energy allocation and adjust their metabolism to secure further development [54]. The high rates of both bioconversion and metabolization in the present study show the ability of BSF larvae to bioconvert medfly rearing waste effectively. From a waste management point of view, the higher proportion of metabolized waste and lower proportion of residue are beneficial, and both quality and quantity of feed have an impact on the metabolic rates, [28, 54].
The approximate digestibility of the different diets by BSF larvae expressed as assimilation efficiency (AD) was calculated to estimate the accurate quantification of actually digested food. AD ranged in the present study from 40.6 ± 1.3 to 74.1 ± 2.4%. The percentage of Diet-HW was significantly higher as compared to the other experimental diets (F(4,29) = 15.1, P < 0.001) (Table 2). The efficacy of larvae to convert the ingested diets to biomass (ECI) ranged from 13.5 ± 0.9 for Diet-H to 16.3 ± 0.8 for Diet-H75, whereas the ECI Diet-H75 and Diet-H25 were significantly higher as compared to Diet-H50 and Diet-W and Diet-H (F(4,29) = 4.5, P = 0.007). The obtained results revealed that the BSF larvae reared on medfly rearing waste had ECI compared to brewer waste [58], cropstraw waste [51] and animal manure [31], and was similar to household kitchen waste [18]. It has been reported that the ECI of most insects ranged from 11 to 17 [59].
Both AD and ECI were used to calculate ECD that refers to the ability of BSF larvae to convert the digested diet into biomass where many essential nutrients may not be absorbed into larval bodies. The ECD for Diet-W and Diet-H25 were significantly lower as compared to the other diets (F(4,29) = 5.2, P = 0.004). The ECD in the present study (18.3–35.5%) were high values as compared to 16.6% on household waste [18], 5–10% on Rice straw [60] and 5% on spent coffee ground [61]. Our findings revealed an inverse relationship between ECD and both M and AD, with the highest M and AD corresponding with the lowest ECD and vice versa. A higher ECD value indicates that the larva is more efficient at converting digested waste into body mass. The availability of sufficient concentrations of essential nutrients, as well as the preservation of homeostatic balance within larval bodies, have an impact on ECD values. [31, 59].
Biochemical Analysis
The chemical compositions of the experimental feeding trials revealed that the fat (F(1,11) = 960.2, P < 0.001) and ash (F(1,11) = 23.2, P = 0.007) contents of Diet-W were significantly higher than of Diet-H (Fig. 4). While, the protein and carbohydrates contents of Diet-H were significantly higher (F(1,11) = 240.5, P < 0.001) and (F(1,11) = 6.7, P = 0.03), respectively. Although most of the nutrients of the diet should be consumed by med fly larvae during their life span to reach pupae, high nutrients contents were recorded in the rearing waste (see Fig. 4) as compared to hen feed. These high contents are caused by the presence of wheat bran (bulking agents) that was not consumed by med fly larvae and the presence of frass, un-hatched eggs and the larvae which were dead or developed late. It was found that the nutritional composition of wasted medfly diet was relatively similar to the initial diet, however a decrease in organic matter and digestibility of organic matter has been recorded [25], while Dominiak and Fanson [19] found that the wasted diet of Queensland fruit fly, Bactrocera tryoni larvae was higher in the crude fat and protein contents than the initial diet.
The chemical compositions of the two BSF pupae, Pupae-H and Pupae-W, previously fed as larvae on hen diet Diet-H and medfly rearing waste Diet-W, respectively are shown in Fig. 5. The protein content of Pupae-W was un-significantly higher than Pupae-H (F(1,11) = 0.88, P = 0.36). While, the fat content of Pupae-W was lower than Pupae-H (F(1,11) = 0.90, P = 0.37). In the same figure, the percentage of carbon (C) in Pupae-W was with 41.7% significantly higher than 38.2% of Pupae-H (F(1,11) = 19.2, P = 0.001). While the percentage of nitrogen in Pupae-W was with 29.3% significantly lower than 31.9% of Pupae-H (F(1,11) = 11.6, P = 0.007).
The concentrations of trace elements due to bioaccumulation by BSF pupae fed on the two tested diets are presented in Table 3. As could be expected, the differences in the bioaccumulation not only observed in the physiologically essential heavy, some metalloids and non-metals showed significant differences. The bioaccumulation factors (≥ 1) of the pupae-H were (1.0, 1.1, 1.4, 1.6, 1.6, and 7.0) in Hg < Ca < K < Cl = Cd < Zn, respectively, while they were (1.0, 1.1, 1.8, 2.6 and 6) of Fe < K < Zn < Ca < Mn, respectively of the pupae-W. Our results revealed that the concentration of elements contents of BSF pupae were varied depending on the larval diets which are in line with that found by [1] who analysed the mineral composition of BSF prepupae reared on different organic waste as larvae. The two tested diets in the current study exhibited different mineral accumulation of BSF pupae which are in line with findings of Purschke et al. [62] and Shumo et al. [63] who reported that the BSF larvae could accumulate heavy metals when fed with contaminated feeds.
The obtained result also indicated that BSF pupae had a low accumulation of non-essential elements, since their contents were lower in the pupae than those obtained from the two experimental treatments Diet-H and Diet-W. Our obtained results of bioaccumulation factor of Cd (1.6 and 0.9) of Pupae-H and Pupae-W, respectively are coincident with Proc et al. [64] who presented a value 1.74 in BSF pupae, while higher bioaccumulation factors of 2.32 and 2.94 and of 5.60–5.90 have been reported by Diener et al. [65] and Bulak [8], respectively.
Conclusions
High amounts of rearing waste are amassed daily due to insect mass rearing as a component of SIT for pest management such as the Mediterranean Fruit fly. Sustainable utilization concepts are required for this organic waste and its nutrients. It is suggested that the omnivorous BSF could be applied to valorise this rearing waste into high-value feed. In this study, it was shown that Medfly rearing waste is a potential feed ingredient for the production of BSF pre-pupae. Generally, BSF larvae were able to convert the medfly rearing waste without negative impact on feeding efficiencies. However, significant differences in bioconversion and feed reduction were observed between the diets containing Medfly rearing waste in different ratios and the control diet. At 15 days old, the mass yield was the highest for larvae fed on Diet-H75. And although percentages of pupation and emergence decreased with increasing medfly-rearing waste content in the experimental diets, pupation was above 90% and emergence above 80% for all diets. The bioconversion rates of BSF larvae on diets containing Medfly rearing waste were with up to 16.3% higher than on other organic wastes as seen in literature. A nutritional analysis revealed that the bioaccumulation of heavy metals was low, especially from the Diet-W derived from Medfly rearing waste.
More research is required to determine the optimum inclusion of Medfly rearing waste in diets of BSF. Moreover, chemical analyses with regard to nutritional profiles would complement the current study. In addition, legal as well as ethical issues need to be cleared before Medfly rearing waste can be used as a sustainable diet for the production of BSF larvae.
Data Availability
The datasets generated during and/or analysed during the current study are not publicly available but are available from the corresponding author on reasonable request.
References
Spranghers, T., et al.: Nutritional composition of black soldier fly (Hermetia illucens) prepupae reared on different organic waste substrates. J. Sci. Food Agric. 97, 2594–2600 (2017)
Bulak, P., et al.: Biogas generation from insects breeding post production wastes. J. Clean. Prod. 244, 118777 (2020). https://doi.org/10.1016/j.jclepro.2019.118777
Diener, S., et al.: Black soldier fly larvae for organic waste treatment: prospects and constraints. Proc. Waste Saf 2, 13–15 (2011)
Wang, Y.-S., Shelomi, M.: Review of black soldier fly (Hermetia illucens) as animal feed and human food. Foods 6, 91 (2017). https://doi.org/10.3390/foods6100091
Salomone, R., et al.: Environmental impact of food waste bioconversion by insects: application of life cycle assessment to process using Hermetia illucens. J. Clean. Prod. 140, 890–905 (2017). https://doi.org/10.1016/j.jclepro.2016.06.154
Zdybicka-Barabas, A., et al.: Immune response in the larvae of the black soldier fly Hermetia illucens. Invertebr. Surviv. J. 14, 9–17 (2017)
Waśko, A., et al.: The first report of the physicochemical structure of chitin isolated from Hermetia illucens. Int. J. Biol. Macromol. 92, 316–320 (2016). https://doi.org/10.1016/j.ijbiomac.2016.07.038
Bulak, P., et al.: Hermetia illucens as a new and promising species for use in entomoremediation. Sci. Total Environ. 633, 912–919 (2018). https://doi.org/10.1016/j.scitotenv.2018.03.252
Wang, X., et al.: Metallothionein in Hermetia illucens (Linnaeus, 1758) larvae (Diptera: Stratiomyidae), a potential biomarker for organic waste system. Environ. Sci. Pollut. Res. 25, 5379–5385 (2018). https://doi.org/10.1007/s11356-017-0856-7
Müller, A., Wolf, D., Gutzeit, H.O.: The black soldier fly, Hermetia illucens–a promising source for sustainable production of proteins, lipids and bioactive substances. Zeitschrift für Naturforschung C 72, 351–363 (2017). https://doi.org/10.1515/znc-2017-0030
Poveda, J.: Insect frass in the development of sustainable agriculture: a review. Agron. Sustain. Dev. 41, 1–10 (2021). https://doi.org/10.1007/s13593-020-00656-x
Lardé, G.: Recycling of coffee pulp by Hermetia illucens (Diptera: Stratiomyidae) larvae. Biol. Wastes 33, 307–310 (1990). https://doi.org/10.1016/0269-7483(90)90134-E
Myers, H.M., Tomberlin, J.K., Lambert, B.D., Kattes, D.: Development of black soldier fly (Diptera: Stratiomyidae) larvae fed dairy manure. Environ. Entomol. 37, 11–15 (2014). https://doi.org/10.1093/ee/37.1.11
Hem, S., Toure, S., Sagbla, C., Legendre, M.: Bioconversion of palm kernel meal for aquaculture: experiences from the forest region (Republic of Guinea). Afr. J. Biotechnol. 7, 1192–1198 (2008)
Diener, S., Zurbrügg, C., Tockner, K.: Conversion of organic material by black soldier fly larvae: establishing optimal feeding rates. Waste Manag. Res. 27, 603–610 (2009). https://doi.org/10.1177/2F0734242X09103838
Lalander, C., et al.: Faecal sludge management with the larvae of the black soldier fly (Hermetia illucens): from a hygiene aspect. Sci. Total Environ. 458, 312–318 (2013). https://doi.org/10.1016/j.scitotenv.2013.04.033
Van Huis, A.: Potential of insects as food and feed in assuring food security. Annu. Rev. Entomol. 58, 563–583 (2013). https://doi.org/10.1146/annurev-ento-120811-153704
Mahmood, S., Zurbrügg, C., Tabinda, A.B., Ali, A., Ashraf, A.: Sustainable waste management at household level with black soldier fly larvae (Hermetia illucens). Sustainability 13, 9722 (2021). https://doi.org/10.3390/su13179722
Dominiak, B.C., Fanson, B.G.: Assesing the proportion of nutrients removed from the larval diet by Queensland fruit fly ('Bactrocera tryoni’) at a mass rearing facility and possible uses of spent media. Gen. Appl. Entomol. 45, 71–75 (2017)
Jucker, C., Lupi, D., Moore, C.D., Leonardi, M.G., Savoldelli, S.: Nutrient recapture from insect farm waste: bioconversion with Hermetia illucens (L.)(Diptera: Stratiomyidae). Sustainability 12, 362 (2020). https://doi.org/10.3390/su12010362
Marco, A., Ramzy, R.R., Wang, D., Ji, H.: Sustainable management of Se-rich silkworm residuals by black soldier flies larvae to produce a high nutritional value and accumulate ω-3 PUFA. Waste Manag. 124, 72–81 (2021). https://doi.org/10.1016/j.wasman.2021.01.040
Barnes, B., Rosenberg, S., Arnolds, L., Johnson, J.: Production and quality assurance in the SIT Africa Mediterranean fruit fly (Diptera: Tephritidae) rearing facility in South Africa. Fla. Entomol. 90, 41–52 (2007). https://doi.org/10.1653/0015-4040(2007)90[41:PAQAIT]2.0.CO;2
Rachid, E., Ahmed, M.: Cur Tephritidae) control in Morocco. J. Entomol. 15, 47 (2018). https://doi.org/10.3923/je.2018.47.55
Dyck, V.A., Hendrichs, J., Robinson, A.S.: Sterile Insect Technique: Principles and Practice in Area-Wide Integrated Pest Management. CRC Press, Boca Raton (2005)
Mastrangelo, T., Silva, J., Abdalla, A., Peçanha, M., Walder, J.: Potential use of larval diet disposal from medfly mass-rearing as alternative livestock feed. Livest. Res. Rural Dev. 22, 58 (2010)
Organization, W. H.: Food irradiation: a technique for preserving and improving the safety of food. https://apps.who.int/iris/handle/10665/38544 (1988)
Liu, Z., Minor, M., Morel, P.C., Najar-Rodriguez, A.J.: Bioconversion of three organic wastes by black soldier fly (Diptera: Stratiomyidae) larvae. Environ. Entomol. 47, 1609–1617 (2018). https://doi.org/10.1093/ee/nvy141
Kinasih, I., et al.: Growth performance of black soldier fly larvae (Hermetia illucens) fed on some plant based organic wastes. HAYATI J. Biosci. 25, 79–79 (2018). https://doi.org/10.4308/hjb.25.2.79
Hemati, S., Naseri, B., Nouri Ganbalani, G., Rafiee Dastjerdi, H., Golizadeh, A.: Effect of different host plants on nutritional indices of the pod borer, Helicoverpa armigera. J. Insect Sci. 12, 55 (2012). https://doi.org/10.1673/031.012.5501
Scriber, J., Slansky, F., Jr.: The nutritional ecology of immature insects. Annu. Rev. Entomol. 26, 183–211 (1981). https://doi.org/10.1146/annurev.en.26.010181.001151
Oonincx, D., Van Huis, A., Van Loon, J.: Nutrient utilisation by black soldier flies fed with chicken, pig, or cow manure. J. Insects Food Feed. 1, 131–139 (2015). https://doi.org/10.3920/JIFF2014.0023
Abduh, M.Y., Nadia, M.H., Manurung, R., Putra, R.E.: Factors affecting the bioconversion of Philippine tung seed by black soldier fly larvae for the production of protein and oil-rich biomass. J. Asia-Pac. Entomol. 21, 836–842 (2018). https://doi.org/10.1016/j.aspen.2018.06.007
Gold, M., et al.: Biowaste treatment with black soldier fly larvae: increasing performance through the formulation of biowastes based on protein and carbohydrates. Waste Manag. 102, 319–329 (2020). https://doi.org/10.1016/j.wasman.2019.10.036
AOAC International: Official Methods of Analysis 16 Association of Official Analytical Chemists. AOAC International, Washington (1995)
Horwitz, W.: Official methods of analysis of AOAC International. In: Horwitz, W. (ed.) Agricultural Chemicals, Contaminants, Drugs, Vol 1. AOAC International, Gaithersburg (1997)
Christie, W.W.: Lipid Analysis, vol. 207. Pergamon Press, Oxford (1982)
Walker, C.: Kinetic models to predict bioaccumulation of pollutants. Funct. Ecol. 4, 295–330 (1990)
Carpenter, R.: Principles and procedures of statistics, with special reference to the biological sciences. Eugen. Rev. 52, 172 (1960)
Li, Q., et al.: Bioconversion of dairy manure by black soldier fly (Diptera: Stratiomyidae) for biodiesel and sugar production. Waste Manag. 31, 1316–1320 (2011). https://doi.org/10.1016/j.wasman.2011.01.005
Gobbi, P., Martinez-Sanchez, A., Rojo, S.: The effects of larval diet on adult life-history traits of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). Eur. J. Entomol. 110, 461 (2013)
Bertinetti, C., Samayoa, A.C., Hwang, S.-Y.: Effects of feeding adults of Hermetia illucens (Diptera: Stratiomyidae) on longevity, oviposition, and egg hatchability: insights into optimizing egg production. J. Insect Sci. 19, 19 (2019). https://doi.org/10.1093/jisesa/iez001
Nakamura, S., Ichiki, R.T., Shimoda, M., Morioka, S.: Small-scale rearing of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae), in the laboratory: low-cost and year-round rearing. Appl. Entomol. Zool. 51, 161–166 (2016). https://doi.org/10.1007/s13355-015-0376-1
Tomberlin, J.K., Adler, P.H., Myers, H.M.: Development of the black soldier fly (Diptera: Stratiomyidae) in relation to temperature. Environ. Entomol. 38, 930–934 (2009). https://doi.org/10.1603/022.038.0347
Medina, R.F., Barbosa, P., Waddell, K.: Parasitism levels in Orgyia leucostigma feeding on two tree species: implications for the slow-growth-high-mortality hypothesis. Entomol. Exp. Appl. 115, 193–197 (2005). https://doi.org/10.1111/j.1570-7458.2005.00243.x
Sayed, W.A., El-Helaly, A., Jamal, Z.A., El-Bendary, H.: Effect of a low cost diet on the cotton leaf worm, Spodoptera littoralis nucleopolyhedrosis virus pathogenicity and sterile insect technique. Egypt. J. Biol. Pest Control 31, 1–8 (2021). https://doi.org/10.1186/s41938-021-00464-9
Benrey, B., Denno, R.F.: The slow-growth–high-mortality hypothesis: a test using the cabbage butterfly. Ecology 78, 987–999 (1997). https://doi.org/10.1890/0012-9658(1997)078[0987:TSGHMH]2.0.CO;2
Holmes, L., Vanlaerhoven, S., Tomberlin, J.: Substrate effects on pupation and adult emergence of Hermetia illucens (Diptera: Stratiomyidae). Environ. Entomol. 42, 370–374 (2013). https://doi.org/10.1603/EN12255
Ellis, J.D., Jr., Hepburn, R., Luckman, B., Elzen, P.J.: Effects of soil type, moisture, and density on pupation success of Aethina tumida (Coleoptera: Nitidulidae). Environ. Entomol. 33, 794–798 (2004). https://doi.org/10.1603/0046-225X-33.4.794
Chen, M., Shelton, A.M.: Impact of soil type, moisture, and depth on swede midge (Diptera: Cecidomyiidae) pupation and emergence. Environ. Entomol. 36, 1349–1355 (2014). https://doi.org/10.1093/ee/36.6.1349
Kim, C.-H., et al.: Use of black soldier fly larvae for food waste treatment and energy production in Asian countries: a review. Processes 9, 161 (2021). https://doi.org/10.3390/pr9010161
Liu, C., Wang, C., Yao, H.: Comprehensive resource utilization of waste using the black soldier fly (Hermetia illucens (L.))(Diptera: Stratiomyidae). Animals 9, 349 (2019). https://doi.org/10.3390/ani9060349
Pastor, B., Velasquez, Y., Gobbi, P., Rojo, S.: Conversion of organic wastes into fly larval biomass: bottlenecks and challenges. J. Insects Food Feed. 1, 179–193 (2015). https://doi.org/10.3920/JIFF2014.0024
Laganaro, M., Bahrndorff, S., Eriksen, N.T.: Growth and metabolic performance of black soldier fly larvae grown on low and high-quality substrates. Waste Manag. 121, 198–205 (2021). https://doi.org/10.1016/j.wasman.2020.12.009
Gligorescu, A., Toft, S., Hauggaard-Nielsen, H., Axelsen, J.A., Nielsen, S.A.: Development, growth and metabolic rate of Hermetia illucens larvae. J. Appl. Entomol. 143, 875–881 (2019). https://doi.org/10.1111/jen.12653
Sheppard, D.C., Tomberlin, J.K., Joyce, J.A., Kiser, B.C., Sumner, S.M.: Rearing methods for the black soldier fly (Diptera: Stratiomyidae). J. Med. Entomol. 39, 695–698 (2002). https://doi.org/10.1603/0022-2585-39.4.695
Banks, I.J., Gibson, W.T., Cameron, M.M.: Growth rates of black soldier fly larvae fed on fresh human faeces and their implication for improving sanitation. Trop. Med. Int. Health 19, 14–22 (2014). https://doi.org/10.1111/tmi.12228
Roper, C., Pignatelli, P., Partridge, L.: Evolutionary responses of Drosophila melanogaster life history to differences in larval density. J. Evol. Biol. 9, 609–622 (1996)
Meneguz, M., et al.: Effect of rearing substrate on growth performance, waste reduction efficiency and chemical composition of black soldier fly (Hermetia illucens) larvae. J. Sci. Food Agric. 98, 5776–5784 (2018)
Waldbauer, G.: Advances in Insect Physiology, vol. 5, pp. 229–288. Elsevier, Amsterdam (1968)
Manurung, R., Supriatna, A., Esyanthi, R.R., Putra, R.E.: Bioconversion of rice straw waste by black soldier fly larvae (Hermetia illucens L.): optimal feed rate for biomass production. J. Entomol. Zool. Stud. 4, 1036–1041 (2016)
Permana, A.D., Putra, J.E.N.R.E.: IOP Conference Series: Earth and Environmental Science, p. 012070. IOP Publishing, Bristol (2019)
Purschke, B., Scheibelberger, R., Axmann, S., Adler, A., Jäger, H.: Impact of substrate contamination with mycotoxins, heavy metals and pesticides on the growth performance and composition of black soldier fly larvae (Hermetia illucens) for use in the feed and food value chain. Food Addit. Contam. Part A 34, 1410–1420 (2017). https://doi.org/10.1080/19440049.2017.1299946
Shumo, M., et al.: The nutritive value of black soldier fly larvae reared on common organic waste streams in Kenya. Sci. Rep. 9, 1–13 (2019). https://doi.org/10.1038/s41598-019-46603-z
Proc, K., Bulak, P., Wiącek, D., Bieganowski, A.: Hermetia illucens exhibits bioaccumulative potential for 15 different elements–Implications for feed and food production. Sci. Total Environ. 723, 138125 (2020). https://doi.org/10.1016/j.scitotenv.2020.138125
Diener, S., Zurbrügg, C., Tockner, K.: Bioaccumulation of heavy metals in the black soldier fly, Hermetia illucens and effects on its life cycle. J. Insects Food Feed. 1, 261–270 (2015). https://doi.org/10.3920/JIFF2015.0030
Funding
Open Access funding enabled and organized by Projekt DEAL. The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
WS, RH, THS and BR conceived and designed research. WS, THS and RH conducted bioassay experiments. MA and RH carried out biochemical experiments. THS, RH and WS analyzed data. THS, and WS and BR wrote the manuscript. WS and BR reviewed and edited. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Sayed, W.A.A., Alm-Eldin, M.M.S., Hassan, R.S. et al. Recycling of Mediterranean Fruit Fly Rearing Waste by Black Soldier Fly, Hermetia illucens. Waste Biomass Valor 14, 93–104 (2023). https://doi.org/10.1007/s12649-022-01847-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-022-01847-1