Abstract
Recycling and reuse are a fundamental pillar for reducing global pollution. In this context, the use of waste generated by invasive species control actions, as a natural raw material for the exploitation of their bioactive compounds, provides a great interest for a more sustainable industry. In this study, phenolic compounds were determined from the different parts that compose the invasive species Arundo donax L. The experiments were carried out using response surface methodology (RSM) by the Box-Behnken method, allowing us to evaluate the factors (temperature, time and type of solvent) and the values that optimized the extraction. The highest concentration of total phenolic compounds equivalent in gallic acid (GAE) was obtained from extracts with distilled water, with the fraction of leaves (7.43 mg GAE /g) in first place, followed by inflorescences (5.15 mg GAE /g) and finally stems (2.73 mg GAE /g).These optimum values were obtained using conditions of 6 h and 45 °C for leaves and stems and 4 h and 35 °C for inflorescences.. Identification and quantification of phenolic acids and flavonoids were carried out by HPLC–MS/MS analytical technique. In general, stem extracts were the most abundant in phenolic acids with potential pharmacological properties. These studies provided promising results for the Arundo donax L. species as a source of polyphenols, making full use of its residue and aiming to boost the circular economy in important industry sectors.
Graphical Abstract

Similar content being viewed by others
Statement of Novelty
Our contribution deals about the potential of to reduce the environmental impact of invasive species through their reuse and obtaining bioproducts. A comprehensive management of these species is proposed, where once these are eliminated from the environment, they can be used for the extraction of other components and thus contribute to different specialized industries. This will contribute to the sustainable development goals.
Currently, the articles that study phenolic compounds from biowaste are oriented towards agro-industrial waste, but none have been found that focus on waste from invasive species. The current management methods for invasive species cause environmental impacts when they end up in landfills or in waste burning.
Introduction
Arundo donax L. is an herbaceous, rhizomatous perennial and invasive species in many warm regions (Oceania, Africa and America) belonging to the grass family (Poaceae). Has generally been considered as native to sub-tropical Eurasia, but the origin of invasive populations remains unknown [1]. The problems deriving from the naturalization, and currently invasion, of A. donax originate in their aggressive competition, adapting well to riparian systems where it spreads vegetatively. It tends to form large, continuous root masses, sometimes covering several hectares, making it impossible for native species to grow and increasing the risk of wildfire [2, 3]. In conclusion, we have the result of river corridors with low biological diversity dominated by reed groups.
A.donax has been considered for a long time as a species with a high economic potential, in contrast from an environmental protection point of view, being cultivated in some regions [4, 5]. Among the diverse uses, one of the most popular is as raw material for bioenergy or biochemistry, being applied in the production of biofuels by biological fermentation (biogas and bioethanol) and for the direct combustion of biomass [6]. In addition, extraction of its chemical compounds gives a wider application in industrial uses, and this is the focus of this work. In relation to these compounds, reviewing the literature has shown that the rhizomes of A. donax. are a potential source of antimicrobial agents and have been applied in medicinal uses [7]. Also, highly porous bioactive aerogels (materials with very interesting characteristics as biomaterial, especially in tissue engineering) have been produced by valorization of lignocellulosic biomass, from extractions realized with their stems and leaves [8].
Apart from an economic context, A. donax could be used as a soil and water pollution control tool by means of phytoremediation. Several studies coincide that A. donax may improve the fertility of soils that were previously industrially polluted, as well as improve the quality of polluted water masses [4]. This is an interesting application that makes A. donax a candidate for phytoremediation in line with the sustainable production of plant-based raw materials [9], making more sense to plant it in necessary areas that help the management and compensation of environmental impact.
Phenolic compounds are synthesized in plants partly as a response to ecological and physiological pressures such as pathogen and insect attack, UV radiation and wounding [10]. Chemically, phenolic compounds vary widely, although their common characteristic is the presence of one (simple phenolics) or more (polyphenols) aromatic rings with bound hydroxyl groups in their structures. Depending on their structure, they can be grouped into phenolic acids, flavonoids, stilbenoids and lignans (Fig. 1) [11, 12]. In addition, phenolic compounds have been widely studied for their ability to counteract oxidative stress, as chronic oxidative stress is linked to several metabolic disorders and a variety of pathologies, such as obesity, diabetes and cardiovascular diseases [13]. The advantages of polyphenols as anticancer agents due to their high accessibility, low toxicity, specificity of response and diverse biological effects are also highlighted [14].
Phenolic acids with C6-C1 structure are widely distributed in the plant kingdom, with the exception of fungi and algae [12]. In general, phenolic acids are divided into hydroxybenzoic and hydroxycinnamic acids according to their structural characteristics. Polyphenols, which include flavonoids, have at least two phenolic subunits; compounds with three or more phenolic subunits are called tannins (hydrolysable and non-hydrolysable). The basic structure of flavonoids is C6-C3-C6 (Fig. 1), this basic structure allows a multitude of substitution patterns and variations on the "C" ring, giving rise to subclasses [15].
The extraction efficiency of phenolic compounds depends on various parameters [16]. First of all, the extraction technique and the solvents used is a fundamental point due the influence of innumerable chemical properties inherent to the plant material, being necessary the choice of different polarity solvents. The most common aqueous organic solvents used are water, acetone, ethyl acetate, alcohols (methanol, ethanol and propanol) and their possible mixtures [10]. Secondly, the time and temperature factors. An excess of both could result in the destruction of the compounds. By contrast, if they are limited, the solvent may not access to the plant tissue and consequently extract the phenolic compounds. That said, in this type of work, the best option is based on statistical tools that help us to study the combinations of factors in a wider range, providing optimal extractions and minimizing the energetic cost of the process [17]. Thus, the aim of this study is to obtain and extract the phenolic compounds of the invasive species A. Donax. The objective is to take advantage of the waste generated in the management programs of invasive species and introduce them into the circular economy. For this, the extraction process of phenolic compounds will be optimized through the response surface methodology.
Material and Methods
Sample Preparation and Extraction Method
The leaves, stems and inflorescences of A. donax were obtained from the town of Moaña, Galicia (Spain). The material was dried separately in an oven at a constant and controlled temperature, then crushed to minimize its volume. Extractions were performed in an incubator with orbital shaking at 100 rpm, mixing 2 g of plant (leaves, stem and inflorescences) with 200 ml of solvent (water, ethanol or hexane) in an Erlenmeyer flask. The analyses of the samples were carried out at the Centre for Scientific and Technological Support to Research (CACTI) of the Ourense and Vigo campuses.
All experiments were repeated in triplicate. The combination of the parameters time, temperature and solvent type for each experiment were determined by box-behnken design. At the end of the process, the solid extract was recovered through a mesh that was used to calculate the extraction yield from the following formula:
mo: weight of extracted pectin, m: weight of dried sunflower heads [18].
Solvent Selection
Exhaustive extractions with increasing polarity solvents, from a non-polar solvent (hexane) to a more polar solvent (distilled water), have been carried out in this study to ensure that a wide range of polarity of the compounds can be extracted [19]. The most important characteristics considered for the selection of these solvents are shown in Table 1 [20, 21].
Experimental Design
To evaluate the influences between factors and optimize the concentration of total phenols, the response surface methodology was used by means of the Box-Behnken experimental design. In the present study, a three-level Box-Behnken design was used and optimal conditions were determined by a minimum number of experiments compared to other designs [22]. The generalized second-order polynomial model used in the response surface analysis was explained by Eq. 2
where “Y” is the response variables; “Ao”, “Ai, “Aii” and “Aij” are the regression coefficients for intercept, linear, quadratic and interaction terms, respectively; “Xi” and “Xj” represent the independent variables (i ≠ j) [18]. The factors temperature (X1), extraction time (X2) and solvent type (X3) were studied on the response to optimise the concentration of total phenol (TPC) gallic acid equivalent (GAE). A total of 13 experiments with their respective combinations were run, one of them corresponding to the central point. Every factor consisted of three coded levels representing the minimum, mean and maximum values shown in Table 2.
Spectrophotometric Determination of TPC
The total phenolic content in the samples/extracts was determined by the Folin–Ciocalteu colorimetric method using gallic acid as the standard [23]. The method relies on the transfer of electrons in alkaline medium from phenolic compounds to form a blue chromophore constituted by a phosphotungstic/phosphomolybdenum complex where the maximum absorption depends on the concentration of phenolic compounds [24]. An Agilent spectrophotometer, Cary-60 VIS-UV, as double internal beam with pulsing xenon lamp was used and absorbance was read at 760 nm. A calibration curve was building at five gallic acid concentrations (50, 25, 10, 5, 0) mg/L, (y = 0.0109×—0.001; R2 = 0.9998). The total phenolic content was expressed as mg of gallic acid equivalent (GAE) per g dry part plant.
Results were expressed as milligrams gallic acid equivalents (GAE) per gram of dry weight (TPC, mg GAE/g), calculated as follows Eq. (3):
where, “CGAE” is the concentration of total polyphenols (mg GAE/mL) at a given extraction time, “V” is the volume of the extract (mL) and “m” the dry part plant (g) [25].
HPLC–MS/MS Analysis Polyphenols Content
For quantitative identification of phenolic compounds in A.donax, the HPLC–MS/MS technique was used and compared with standard spectra of thirty-six compounds.
Analyses were carried out using liquid chromatography (Agilent technologies 1260 infinity) coupled to a triple quadrupole tandem mass spectrometer (MS/MS AB SCIEX Triple Quad 3500) with an electrospray ionisation (ESI) source. Chromatographic separation was performed on a Phenomenex Luna C18 column(150 mm × 2 mm; 3 μm particle diameter) as stationary phase. The mobile phase was composed of water (A, 0.1% formic acid) and acetonitrile (B, 0.1% formic acid), whose gradient programme was: 98% A (v/v) from 0 to 4.0 min, 98–80% A (v/v) from 4.0 to 7.0 min, 80–10% A (v/v) from 7.0 to 14.0 min, 10% A (v/v) from 14.0 to 15.0 min, 10–98% A (v/v) from 15.0 to 17.0 min. The sample injection volume was 5 μL and the mobile phase rate was 300 μL/min. For MS analysis of the samples, both negative and positive ion modes were operated and the following settings were used: ion sputtering voltage (IS), 4500 V; curtain gas, 25 psi; nebuliser gas (GS1), 55 psi; auxiliary gas (GS2), 55 psi; turbo temperature, 400 °C. The mass analyser operated in the multiple reaction monitoring (MRM) scanning mode, providing sensitive and accurate measurement. The exact mass and composition of precursor ions and fragments were analysed with the Analyst 1.6.2 software (AB Sciex, Foster City, CA) integrated in the instrument.
Statistical and Graphical Analysis
The adequacy of the models was determined by evaluating the p-value, besides to the coefficient of determination (R2) and the Fisher test value (F value) obtained from the analysis of variance (ANOVA). The statistical significance of the model parameters was determined at a 95% confidence level. The analysis of variance and the response surfaces was performed with Desing-Expert 13 (Trial Version, State-Ease Inc., Minneapolis, MN, USA) and Minitab 20.2 softwares.
It was necessary to adjust the values of the solvents to a numerical coding in which they were: ethanol (1), distilled water (2) and hexane (3).
Results and Discussion
Extraction Efficiency
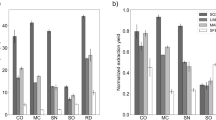
The overall extraction was highest for leaves and lowest for stems. The best extraction efficiency for leaves, inflorescences and stems was 22.5%, 24.75% and 15.75% respectively (Fig. 2). The extraction efficiency of water was very high compared to ethanol and hexane, which was very low for all parts. Regarding the conditions studied, the highest value with water in inflorescences and stems was at 6 h and 45 °C, indicating that these are favourable terms for this solvent. In leaves, the best value is also extracted at 6 h but at the lower temperature of 25 °C.
Turning to the less effective solvents, the yield is highest in the inflorescence fraction with ethanol, followed by leaves and stem with hexane. But the values are not significant enough to be relevant.
Total Phenolic Contents (TPC) of Different Extracts
The results obtained for TPC by the Folin-Ciocalteu method are shown in Table 3, indicating that the highest levels were observed with distilled water. With this solvent, the highest levels were obtained in leaves extracts at 6 h of extraction and at temperatures of 45 °C and 25 °C , for the first and second highest respectively. Also, significant levels were observed in the inflorescence with an extract at medium extraction time and temperature conditions (4 h and 35 °C ). Lastly, the stem fraction recorded the lowest content of phenolic compounds with the aqueous extracts, with the best result being obtained at maximum times and temperatures (6 h and 45 °C ).
In order to assess and contrast the results presented in this study, Fontes-Candia et al. (2019) have carried out the recovery of polyphenols from A. donax waste biomass. Extractions were done by two methods, ultrasound and maceration in hot distilled water at 90 °C and with constant shaking during one hour. The reported results show that, in general, hot water treatment favoured the extraction of polysaccharides and polyphenols in the stems, resulting in a higher antioxidant capacity [26]. However, the leaves through the same method obtained less quantity. This contrasts with the results presented and may be due to differences in working conditions. It is likely that stem fraction was favoured by higher temperatures than those used (25–45 °C), depending on whether the phenolic compounds are free or linked to cell membranes/walls [27]. If they are linked to the cell wall, they could be released more easily by increasing the temperature, enhancing both the solubility of the solute and the diffusion coefficient, but this increase should be limited due to the instability of the compounds and possible denaturation [28]. However, studies confirm that around 100 °C the most labile would be catechin and epicatechin, while phenolic acids (the most studied) could withstand up to 150 °C , although attention should be paid to the combination of times [29].
Distilled water has a high polarity and has been confirmed to be highly effective for polar compounds, also if working temperature is increased it may be able to extract less polar compounds, since the intermolecular interactions involving hydrogen bonds become less pronounced, thereby favouring London's dispersion forces [30]. This could be the reason for a better extraction for this fraction.
Ethanol and hexane extracts had the lowest total phenolic content. Among them, hexane stands out with extracts in leaves and stems at 35 °C and 6 h of extraction, and in inflorescence at 45 °C and 4 h of extraction.
Polar solvents are frequently used for the recovery of polyphenols from a plant matrix [31]. But to extract different phenolic compounds from plants with a high degree of accuracy, several solvents of different polarity must be used [32]. Indeed results have been reported from A.donax rhizome through extractions based on the polarity index, from hexane (p' 0.1) to methanol (p' 5.1), where they coincided with the low amount of phenols found in hexane extracts (< 1%). In contrast, the higher content of the main constituent of all extracts, sterols, was higher for hexane extracts (52%), demonstrating their effectiveness for other components [33].
This research provides the extraction of compounds using the whole residue of the plant, which allowed us to investigate the inflorescence. This part seems to be interesting for their phenolic content [34], but there are no specific studies on this particular species. Certainly, it is the minority fraction among all the parts and it is not found in every season of the year (end of summer to autumn).
Statistical Analysis and Estimation of Adequacy of the Model
Tables 4, 5 and 6 summarize the results of the analysis of variance of the three models (for the three parts), were obtained using the TPC as response variable in the RSM.
In addition, pareto diagrams were added in order to detect the effects of the most important factors and interactions on the optimization, shown in Figs. 3, 4 and 5 for each part.
Statistical significance (p < 0.05) was observed for leaves in B (linear variables), and in CC (quadratic variables), which indicated that polyphenol extractability was influenced by these factors (Fig. 3). Interactions between the factors did not produce any significant influence. The coefficient of determination (R2) obtained from the variance of the quadratic regression model was 96.85%. This demonstrates the adequacy of the model to explain the total variance. Furthermore, the adjusted R2 value was 91.19, which showed that the model contained significance.
Table 5 shows results of ANOVA, adequacy and fitness of the models for total phenols content in inflorescences. The values of the regression coefficients indicate that the linear variables A and B were significant (p < 0.05). The quadratic variables AA, BB and CC were also significant (Fig. 5). Here again, no significant influence was found between the interaction of the variables. The coefficient of determination (R2) obtained from the variance of the quadratic regression model was 96.67% and the adjusted R2 value was 90.70%, which also showed that the model showed significance.
In Table 6, for stems the quadratic models were not significant (p > 0.05) with the exception of the quadratic variable CC (Fig. 4). Furthermore, the regression model produced an R2 value of 78.5. This demonstrates the inadequacy of the model to explain the total variance. Also, the adjusted R2 value was 39.80%, which also showed that the model lacked significance.
The CV indicates the degree of precision with which the treatments are compared and is a good index of the reliability of the experiment. It expresses the experimental error as percentage of the mean; thus, the higher the cv value, the lower is the reliability of the experiment [35]. Large coefficients of variation (> 30%) are often associated with higher experimental variability [36], this is the case for stems with a CV of 32.65%. However, a lower CV value indicates a higher reliability of the experiment, as is the case for leaves 21.97% and inflorescences 20.30%.
Optimization of Extraction Condition
The graphs showed that high TPC values (Fig. 6a–c) could be obtained for leaves extracts using a low temperature with a long extraction time, or using high temperature values also with long extractions. This was achieved with water as solvent for both favourable conditions.
The effect of time, temperature and solvent on the stems is illustrated in Fig. 7. Here again, the effectiveness of water extractions and long durations with high temperatures are repeated as the most favourable combination, followed by long extractions with moderate temperatures.
The trend in the inflorescence was different from the rest of the parts where we located a very clear maximum point (Fig. 8b–c). The TPC increased gradually as the extraction temperature increased until it reached the mean values between 35 °C and 40 °C, after that point it decreased again (Fig. 7c). The same happened with the time that reached the maximum at 4–5 h of extraction (Fig. 7b). Both achieved this increase in TPC without the variation of the solvent, which always remained at the value corresponding to distilled water.
Quantification of Phenolic Acids and Flavonoids Using HPLC–MS/MS
Among the thirty-six standards used, a total of ten compounds were identified, eight phenolic acids and two flavonoids. All of them are shown in Table 7 for leaves, inflorescences and stems.
From these ten compounds, A. donax stems were the fraction that presented the best concentrations of the following acids. Syringic acid (59.80 µg/g) was shown as the main constituent, followed by ferulic acid (32.30 µg/g), p-coumaric (19.32 µg/g), protocatechuic (17.70 µg/g) and vanillic (16.88 µg/g). Sinapic acid, salicylic acid and syringaldehyde were found in much lower concentrations for the stems.
As for the leaves, higher amounts of vanillic (41.38 µg/g), syringic (25.32 µg/g), sinapic (22.53 µg/g) and ferulic (19.48 µg/g) acids were identified. Many coincide with those detected in the stems, but in smaller quantities, with the exception of vanillic acid, which has a greater presence in the leaves. Here also lower amounts of salicylic acid and syringaldehyde are shown, with the addition of p-coumaric acid and protocatechuic acid. Therefore, this study proved the presence of abundant phytochemicals where the stem was the part with the highest concentration of compounds. Finally, and contrasting with the leaves and stems, the inflorescence was the fraction where lower amounts of all detected compounds were observed.
Regarding the solubilities of the compounds in the solvents used, a preference for the more polar solvents, such as ethanol and distilled water, was perceived. This preference for the extraction solvent of each compound was exactly the same for the highest concentration fractions: leaves and stems. The lowest values for all acids were with hexane, with the exception of salicylic acid in inflorescences. Syringic, protocatechuic, vanillic and sinapic acids were more soluble in water, while ferulic, p-coumaric, protocatechuic, salicylic and syringaldehyde acids were more soluble in ethanol.
The use of conventional solvents such as alcohols, often mixed with water, could better extract this type of phenolic acids, due to the high polarity of hydroxybenzoic and hydroxycinnamic acids [14]. Therefore, it should be considered to repeat these experiments by varying the solvents and their possible combinations, in order to look for higher extraction efficiency and, consequently, higher concentrations.
This research, along with other studies focused on the rhizomes of A. donax [33], demonstrates the potential of this species in the pharmacological sector, being able to fully exploit all the available residue.
Exploiting these compounds in an optimized way requires careful studies, which are able to provide a higher productivity in the extraction process, as well as a good adaptation to an industrial scale. This also means a reduction in production costs and an improvement in the quality of the compounds.
Furthermore, these practices help to minimize the environmental impact of the waste generated, as well as being possible substitutes for synthetic materials by alternative raw materials from biological resources.
Conclusion
The aim of this study was to analyze the effect of different extraction variables on extraction yield and total phenolic content. In addition, we also obtained the distribution of phenolic acids and flavonoids in the different parts that compose the A. donax species.
The optimum extraction conditions for the total phenolic content (TPC) were 6 h and 45 °C for leaves and stems, while for inflorescences they were 4 h and 35 °C. Consequently, values of 7.43 mg GAE /g, 5.15 mg GAE /g and 2.73 mg GAE /g were obtained for leaves, inflorescences and stems, respectively.
Identification and quantification of phenolic acids and flavonoids were carried out by HPLC–MS/MS analytical technique. In general, stem extracts were the most abundant in phenolic acids with potential pharmacological properties.
The high energy expenditure generated by long extraction times, the use of large volumes of solvents and the low concentrations of the compounds of interest obtained at the end of the process, have stimulated the search for more efficient and sustainable methods to extract phenols. In addition, this work gives priority to the use of waste from invasive species, which produce tonnes of waste from management measures for their eradication, which could be reinserted into the circular economy.
Data Availability
All data generated or analysed during this study are included in this published article.
References
Hardion, L., Verlaque, R., Saltonstall, K., Leriche, A., Vila, B.: Origin of the invasive Arundo donax (Poaceae): A trans-Asian expedition in herbaria. Ann. Bot. 114(3), 455–462 (2014). https://doi.org/10.1093/aob/mcu143
G. P. Bell, “Ecology and management of Arundo donax, and approaches to riparian habitat restoration in southern California,” Plant Invasions Stud. North Am. Eur. pp. 103–113, 1997.
Brooks, M.L., et al.: Effects of invasive alien plants on fire regimes. Bioscience 54(7), 677–688 (2004). https://doi.org/10.1641/0006-3568(2004)054[0677:EOIAPO]2.0.CO;2
Jámbor, A., Török, Á.: The economics of Arundo donax-A systematic literature review. Sustainability 11(15), 4225 (2019). https://doi.org/10.3390/su11154225
Nocentini, A., Field, J., Monti, A., Paustian, K.: Biofuel production and soil GHG emissions after land-use change to switchgrass and giant reed in the U.S. Southeast. Food Energy Secur. 7(1), e00125 (2018). https://doi.org/10.1002/fes3.125
Corno, L., Pilu, R., Adani, F.: Arundo donax L.: A non-food crop for bioenergy and bio-compound production. Biotechnol. Adv. 32(8), 1535–1549 (2014). https://doi.org/10.1016/j.biotechadv.2014.10.006
Licursi, D., et al.: Characterization of the Arundo Donax L. solid residue from hydrothermal conversion: Comparison with technical lignins and application perspectives. Ind. Crops Prod. 76, 1008–1024 (2015). https://doi.org/10.1016/j.indcrop.2015.08.007
C. Fontes, E. Erboz, A. Martínez-Abad, A. López-Rubio, and M. Martínez-Sanz, Superabsorbent food packaging bioactive cellulose based aerogels from Arundo donax waste biomass.
Nsanganwimana, F., Marchand, L., Douay, F., Mench, M.: Arundo donax L., a candidate for phytomanaging water and soils contaminated by trace elements and producing plant-based feedstock. A review. Int. J. Phytoremediation 16(10), 982–1017 (2014). https://doi.org/10.1080/15226514.2013.810580
Khoddami, A., Wilkes, M.A., Roberts, T.H.: Techniques for analysis of plant phenolic compounds. Molecules 18(2), 2328–2375 (2013). https://doi.org/10.3390/molecules18022328
Šamec, D., Karalija, E., Šola, I., Vujčić Bok, V., Salopek-Sondi, B.: The role of polyphenols in abiotic stress response: The influence of molecular structure. Plants 10(1), 118 (2021)
Bravo, L.: Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 56(11), 317–333 (1998). https://doi.org/10.1111/j.1753-4887.1998.tb01670.x
Furukawa, S., et al.: Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest. 114(12), 1752–1761 (2017)
Brglez Mojzer, E., Hrnčič, M.K., Škerget, M., Knez, Ž, Bren, U.: Polyphenols: Extraction methods, antioxidative action, bioavailability and anticarcinogenic effects. Molecules 21(7), 901 (2016)
Stalikas, C.D.: Extraction, separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 30(18), 3268–3295 (2007). https://doi.org/10.1002/jssc.200700261
Ivanović, M., Razboršek, M.I., Kolar, M.: Innovative extraction techniques using deep eutectic solvents and analytical methods for the isolation and characterization of natural bioactive compounds from plant material. Plants 9(11), 1–29 (2020). https://doi.org/10.3390/plants9111428
Ross, K.A., Beta, T., Arntfield, S.D.: A comparative study on the phenolic acids identified and quantified in dry beans using HPLC as affected by different extraction and hydrolysis methods. Food Chem. 113(1), 336–344 (2009). https://doi.org/10.1016/j.foodchem.2008.07.064
Peng, X., Yang, G., Shi, Y., Zhou, Y., Zhang, M., Li, S.: Box–Behnken design based statistical modeling for the extraction and physicochemical properties of pectin from sunflower heads and the comparison with commercial low-methoxyl pectin. Sci. Rep. 10(1), 1–10 (2020). https://doi.org/10.1038/s41598-020-60339-1
Pandey, A., Tripathi, S., Pandey, C.A.: Concept of standardization, extraction and pre phytochemical screening strategies for herbal drug. J. Pharmacogn. Phytochem. JPP 115(25), 115–119 (2014)
Abubakar, A.R., Haque, M.: Preparation of medicinal plants: Basic extraction and fractionation procedures for experimental purposes. J. Pharm. Bioallied Sci. 12(1), 1–10 (2020). https://doi.org/10.4103/jpbs.JPBS_175_19
Das, K., Tiwari, R.K.S., Shrivastava, D.K.: Techniques for evaluation of medicinal plant products as antimicrobial agent: Current methods and future trends. J. Med. Plants Res. 4(2), 104–111 (2010). https://doi.org/10.5897/JMPR09.030
Pasma, S.A., Daik, R., Maskat, M.Y., Hassan, O.: Application of box-behnken design in optimization of glucose production from oil palm empty fruit bunch cellulose. Int. J. Polym. Sci. 2013, 1–8 (2013). https://doi.org/10.1155/2013/104502
Genwali, G.R., Acharya, P.P., Rajbhandari, M.: Isolation of Gallic acid and estimation of total phenolic content in some medicinal plants and their antioxidant activity. Nepal J. Sci. Technol. 14(1), 95–102 (2013). https://doi.org/10.3126/njst.v14i1.8928
Dvořáková, M., Hulín, P., Karabín, M., Dostálek, P.: Determination of polyphenols in beer by an effective method based on solid-phase extraction and high performance liquid chromatography with diode-array detection. Czech J. Food Sci. 25(4), 182–188 (2007). https://doi.org/10.17221/690-cjfs
Barjoveanu, G., Pătrăuțanu, O.A., Teodosiu, C., Volf, I.: Life cycle assessment of polyphenols extraction processes from waste biomass. Sci. Rep. 10(1), 1–12 (2020). https://doi.org/10.1038/s41598-020-70587-w
Fontes-Candia, C., Erboz, E., Martínez-Abad, A., López-Rubio, A., Martínez-Sanz, M.: Superabsorbent food packaging bioactive cellulose-based aerogels from Arundo donax waste biomass. Food Hydrocoll. 96, 151–160 (2019). https://doi.org/10.1016/j.foodhyd.2019.05.011
I. O. Minatel, C. V. Borges, M. I. Ferreira, H. A. G. Gomez, C.-Y. O. Chen, and G. P. P. Lima, “Phenolic compounds: functional properties, impact of processing and bioavailability.” In Phenolic compounds—biological activity, M. Soto-Hernndez, M. Palma-Tenango, M.R. del Garcia-Mateos (Eds.). InTech, pp. 1–24, 2017
Pinelo, M., Arnous, A., Meyer, A.S.: Upgrading of grape skins: Significance of plant cell-wall structural components and extraction techniques for phenol release. Trends Food Sci. Technol. 17(11), 579–590 (2006). https://doi.org/10.1016/j.tifs.2006.05.003
Herrero, M., Plaza, M., Cifuentes, A., Ibáñez, E.: Extraction techniques for the determination of phenolic compounds in food. Compr. Sampl. Sample Prep. 4, 159–180 (2012). https://doi.org/10.1016/B978-0-12-381373-2.00132-0
Plaza, M., Turner, C.: Pressurized hot water extraction of bioactives. TrAC—Trends Anal. Chem. 71, 39–54 (2015). https://doi.org/10.1016/j.trac.2015.02.022
Sultana, B., Anwar, F., Ashraf, M.: Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules 14(6), 2167–2180 (2009). https://doi.org/10.3390/molecules14062167
Altemimi, A., Lakhssassi, N., Baharlouei, A., Watson, D.G., Lightfoot, D.A.: Phytochemicals: Extraction, isolation, and identification of bioactive compounds from plant extracts. Plants 6(4), 42 (2017). https://doi.org/10.3390/plants6040042
Pansuksan, K., Sukprasert, S., Karaket, N.: Phytochemical Compounds in Arundo donax L. Rhizome and antimicrobial activities. Pharmacogn. J. 12(2), 287–292 (2020). https://doi.org/10.5530/pj.2020.12.45
Sytar, O.: Phenolic acids in the inflorescences of different varieties of buckwheat and their antioxidant activity. J. King Saud Univ.—Sci. 27(2), 136–142 (2015). https://doi.org/10.1016/j.jksus.2014.07.001
Gomez, K.A., Gomez, A.A.: Statistical procedures for agricultural research, vol. 6. Wiley, New York (1984)
Taylor, S.L., Payton, M.E., Raun, W.R.: Relationship between mean yield, coefficient of variation, mean square error, and plot size in wheat field experiments. Commun. Soil Sci. Plant Anal. 30(9–10), 1439–1447 (1999). https://doi.org/10.1080/00103629909370298
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
AC: conceptualization, methodology, investigation, supervision, project administration, funding acquisition, writing—review & editing. XA: software, validation, formal analysis, visualization, supervision. CM: methodology, investigation, data curation, writing-original draft. AS: conceptualization, resources, project administration, funding acquisition.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical Approval
The present material has not been published, simultaneously submitted, or accepted elsewhere, and it has been approved by the co-authors.
Consent to Participate
The co-authors consent to participate.
Consent to Publication
The co-authors consent to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visithttp://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Míguez, C., Cancela, Á., Sánchez, Á. et al. Possibilities for Exploitation of Invasive Species, Arundo donax L., as a Source of Phenol Compounds. Waste Biomass Valor 13, 4253–4265 (2022). https://doi.org/10.1007/s12649-022-01764-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-022-01764-3