Abstract
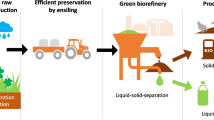
In a Green Biorefinery, grass silage can be a source for lactic acid, proteins, amino acids and fibres. Processing residues can be used for anaerobic digestion and methane production. But by changing the ensiling conditions, butyric acid fermentation can be achieved. That makes grass silage also a potential substrate for a combined butyric acid and methane production. The objective of this study was to determine the potential of butyric acid production at different ensiling conditions applied to grass and measuring the methane yield potential of solid residues after a separation step. The highest butyric acid concentration in the produced press juice was 20.1 ± 4.5 g kg−1 and was achieved by carbonated lime addition and a reduced dry matter content after 90 days at mesophilic storage conditions. This resulted in a theoretical butyric acid yield of 332 kg ha−1 a−1. For the fibrous leftover press cake, a theoretical methane production potential of 2778 m3CH4 ha−1 a−1 was reached. The results show that theoretically a combined production of butyric acid and methane can be realised in a Green Biorefinery concept.
Graphic Abstract

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Statement of Novelty
The specific objective of this research was to investigate whether the ensiling process is suitable for butyric acid production. Therefore, different ensiling conditions were evaluated for butyric acid production which will create additional value on the farm. The residues, which are not used for the production of butyric acid, will not be wasted but used as substrate for biogas production. The information we provide will help to open up new processes and alternative raw materials for butyric acid production und using the leftover residues for methane production through anaerobic digestion. The results are novel and significant to the combined fields of anaerobic digestion and Green Biorefineries and will be viewed by a scientific audience but it is also of great interest to the bioeconomic sector.
Introduction
In 2017, 5.38 billion hectares were considered as agricultural land and used for the production of food and goods worldwide. This includes cropland with 1.7 billion hectares and permanent meadows and pastures with 3.66 billion hectares, which means that 68% of the agricultural land worldwide is dominated by grassland [1]. This grassland can deliver a huge amount of biomass and contributes not only to the food production through animal husbandry, but also has ecological functions. Moreover, this high amount of biomass has a huge potential for energy production through anaerobic digestion.
However, grass can also be used for higher quality processing than energy production. According to bioeconomic approaches, the production of volatile fatty acid acids could lead to a higher added value on the farm. In this approaches, carboxylates formed during the anaerobic degradation of biomass could serve as intermediate platform chemicals [2]. The remaining biomass can be used to generate methane in an anaerobic digestion plant. Right now, the main focus for optimization of anaerobic digestion plants is on upgrading existing plants for demand oriented biogas production [3,4,5]. Another approach to increase the added value in the future could be the Green Biorefinery. In this concept, silages, which are one of the most common feedstock in agricultural biogas plants especially in Germany, could be used as a source for valuable chemicals like lactic acid or proteins [6,7,8]. Green Biorefineries are already recognised for the extraction of valuable proteins from grass and clover and making them available for the nutrition of monogastric animals and ruminants, as an alternative to the use of soybean [9]. Another option could be the generation of organic acids through ensiling [8]. In both cases, the extraction of the desired ingredients through a separation step is necessary. In which the used separation method and the silage quality have a great impact on the separation efficiency [10]. The ensiling process is a lactic acid fermentation, but by changing the ensiling conditions, the lactic acid fermentation can switch to a butyric acid fermentation during fermentation [11]. For the sole production of biogas or the use as animal feedstock, butyric acid formation during fermentation is undesirable. High energy losses occur during ensiling, and it is non-edible for animals due to the high butyric acid concentrations. But in the case of targeted production of butyric acid for use in the chemical industry, it could potentially be a low-cost alternative. Butyric acid is used in a wide range of products. It is a precursor for biofuel production, like biobutanol, in the pharmaceutical and chemical industries, e.g. for plastic production or butyric acid esters can be used in food and cosmetic industry as flavouring or fragrant agents [12]. Having a closer look on the fermentative production of butyric acid, one of the main challenges in butyric acid production is a self-inhibitory effect because of a decreasing pH-value. In addition, the formation of by-products such as acetic acid is of concern, as it is difficult to separate. Furthermore, low butyric acid concentrations followed by high product recovery costs and a limited productivity are major challenges, too. [13]. In research, biotechnological butyric acid production is widely based on glucose, but has also been performed with agricultural raw materials like wheat straw. However, these substrates need pre-treatment before fermentation, like steam explosion and hydrolysis to generate sugars like glucose or xylose for a butyric acid fermentation [13, 14]. Additionally, the usage of xylose e.g. by Clostridium tyrobutyricum is inhibited, but making the usage of xylose possible is part of approaches to optimize the butyric acid fermentation [15]. It is also mentioned, that the usage of glucose, starch or other agricultural derived products is not feasible, because of the high production costs and its competition with the usage for food production [16].
The objective of this research was to determine the natural potential for butyric acid production from grass, further measuring the methane formation potential of the remaining residues and implementing the results into a Green Biorefinery concept.
Materials and Methods
Raw Material
The fresh grass was harvested on 23 May 2018 at the ‘Unterer Lindenhof’ (Eningen Unter Achalm, Germany), an agricultural research station of the University of Hohenheim. The grass was chopped by a forage harvester to 8 mm theoretical length of cut. During harvest no ensiling additives were added.
At ‘Unterer Lindenhof’ an average annual precipitation of 770 mm and an average temperature of 11.6 °C were measured for the last five years [17]. The grassland can be considered as intensively used grassland, as it is cut up to four times a year and fertilized with digestate from a nearby agricultural anaerobic digestion plant.
Fermentation Treatments
Glass jars with a volume of 1.5 L (Weck GmbH und Co. KG, Wehr-Oeflingen, Germany) were used for ensiling and closed with a sealing ring and a glass lid with 3 clips.
The conducted treatments are shown in Table 1. They consist of a control variant (C), a variant with carbonated lime (CaCO3) addition as a pH buffer (B), a variant with the addition of water (W) and a variant from the combination of water and carbonated lime (WB). Both the addition of CaCO3 and the addition of water are aimed at creating conditions to allow the formation of butyric acid. Furthermore, two different storage temperatures were tested. This means that all treatments were performed two times and stored at 22 ±1 °C (20) and at 37 ±1 °C (37).
For filling the jars with chopped grass, 20 kg of grass was mixed and each jar was filled with the feedstock including the additives. CaCO3 and distilled water were added directly into the jars. When filling the jars, there was no headspace left. For the control treatments (C20, C37) a density of 186 kgTS m-3 was achieved. Whereas for the other treatments the density differs according to the additives added (Table 1). The distilled water, CaCO3 and a combination of both was added to decrease the total solid (TS) content of the grass and to increase the buffering capacity. Jars without additives served as control treatment (Table 1).
For fermentation the jars were stored under two temperatures: (i) at 22±1 °C in an insulated storage room and (ii) at 37 ±1 °C in a water bath controlled by a circulation thermostat (Ecoline E200, Lauda Dr. R. Wobser GmbH & Co. KG., Lauda-Koenigshofen, Germany). Temperature of the water bath was measured continuously by four temperature sensors (B+B Immersion sensor 0625142911, B&B Thermotechnik GmbH, Donaueschingen, Germany) and recorded with a data logger (Ebro EBi 40, Xylem Analytics Germany Sales GmbH & Co. KG, Weilheim, Germany). While the temperature in the storage room was measured continuously with two temperature sensors (Testo TE Type K, Testo SE & Co.KGaA, Titisee-Neustadt, Germany) and recorded by a data logger (Testo 176T4, Testo SE & Co.KGaA, Titisee-Neustadt, Germany).
According to the DLG guidelines for ensiling and testing of ensiling additives [18, 19] jars were successively opened after 3, 14, 30 and 90 days of fermentation. Each of the eight treatments was carried out in triplicate, three jars per treatment were opened at each opening date requiring 96 jars in total.
Separation Procedure and Extraction Efficiency
After ninety days of fermentation, samples of the ensiled grass were taken and separated with a tincture press (HPH 2.5, HAPA Fertigungstechnik, Germany) at 20 MPa for 60 s into press cake and press juice.
Specific Methane Yield Determination by Hohenheim Biogas Yield Test (HBT)
The determination of the specific biogas yield (SBY) and the specific methane yield (SMY) was done with Hohenheim Biogas Yield Test (HBT), according to the VDI Guideline 4630 [20]. The gas yield was corrected for standard conditions (273.15 K, 1013 hPa) and expressed in m3 per kg of volatile solids (VS). Further in this article, the measured specific biogas and methane yields are mentioned in relation to m3 kg−1.
The HBT was performed with glass syringes, these were filled with a standardized inoculum and the substrate to be tested. The filled syringes were placed in a climate chamber and digested at 37 °C for 35 days. A detailed description of the HBT system is given by Helfrich and Oechsner [21]. All samples were done in triplicates.
Analytical Methods
Total Solids and Volatile Solids Determination
The TS and VS of the samples were determined according to DIN EN 12,880, DIN EN 12,879. The samples were weighed and then dried overnight in a drying chamber at 65 °C (UF450, Memmert GmbH, Germany). Afterwards they were dried at 105 °C (UF450, Memmert GmbH, Germany) to constant weight and weighed again to determine the TS. The VS was determined by burning the dried samples at 550 °C in a muffle furnace for 6 h and weighing of the resulting ash. The TS was calculated from the resulting masses after drying at 105 °C, and for VS the resulting masses of ash were used to determine the volatile solids as difference between the dried amount and the ash.
The loss of volatile organic compounds during the drying process was considered according to Eq. (1) [22].
In which FA is the sum of fatty acids (FA, C2 – C6), LA is lactic acid (LA), PD is 1.2-propanediol, OA are alcohols (OA) and TSn is the measured value of dry matter, all in g kg−1. The measured VS values were recalculated according to the corrected TSc.
Chemical Composition of the Substrate
The chemical composition of the fresh cut grass was determined with Weender van Soest analysis at the Analytical Chemistry Unit of the Core Facility of the University of Hohenheim. The values are shown based on the TS content of the substrate. For simplification, in the entire results section, they are shown as g kg−1. The content of crude ash (CA), crude protein (CP), crude fat (CL) and crude fibre (CF) were performed according to the regulation of the Commission of the European Communities 152/2009 III [23]. Whereas neutral detergent fibre (NDF), acid detergent fibre (ADF) and acid detergent lignin (ADL) where determined according to the standard of the VDLUFA [24].
Determination of Volatile Fatty Acids with Capillary Gas Chromatography
The determination of volatile fatty acids (VFA) of the liquid and solid samples was performed by gas chromatography (GC) (GC2010plus, Shimadzu Corp., Japan) with an autoinjector (AOC-20i, Shimadzu Corp., Japan), and FID Detector and a capillary column (WCOT Fused Silica, Varian Medical Systems Inc., US). The concentrations of acetic acid, propionic acid, n- and iso-valeric acid, n- and iso-butyric and caproic acid were measured, after calibration with an appropriate standard solution. The concentrations of VFAs are shown based on the total solids content of the sample. For simplification, in the entire results section, they are shown as g kg−1.
Determination of Lactic Acid, Ethanol and 1.2-Propanediol with High Performance Liquid Chromatography (HPLC)
The determination of lactic acid, ethanol, 1.2-propanediol was done with HPLC (Bischoff Analysentechnik u. -ger. GmbH, Germany) after calibration with an appropriate standard. The HPLC is equipped with a RI-Detektor, an Aminex HPLC column (Aminex HPX-87 H, Bio-Rad, US) and a precolumn (HPX-87 H, Bio-Rad, US). The concentration of lactic acid, ethanol and 1.2-propanediol are, expressed based on the total solids content of the sample. For simplification, in the entire results section, these are only shown as g kg−1.
Chemical Oxygen Demand (COD)
The determination of the chemical oxygen demand was performed using a cuvette test (LCK 014, Hach Lange GmbH, Germany) with a measuring range between 1000 and 10,000 mg/L O2. Because of the high COD load of the samples, they were diluted 1:10 with distilled water. After pipetting the samples into the cuvettes, the pulping process was carried out at 170 °C in a high temperature thermostat (HT200S, Hach Lange GmbH, Germany). Later the COD of the samples was measured with a photometer (DR 3900, Hach Lange GmbH, Germany).
Measurement of the pH-Value
For liquid samples, the pH value was determined at room temperature without stirring with a laboratory pH meter (pH meter 211, HANNA Instruments, USA) using a single-rod glass electrode. The electrode was previously calibrated with pH 7 and pH 4 calibration solutions. The temperature effect was compensated based on temperature measurement (HI7669/2 W, HANNA Instruments, USA). For solid samples, 10 g of the sample materials was mixed with 100 ml distilled water and shaken for 1 h with an orbital shaker (KS-15, Edmund Br GmbH, Germany).
Statistical Methods
The data was tested for significant differences with the Kruskal-Wallis test. Subsequently an one-way ANOVA was done and the Tukey post hoc test was applied. Statistical analysis was done with the software R-Studio and the package agricolae [25, –27]. The figures were plotted with R-Studio and the package ggplot2 [26, 28].
Results and Discussion
Raw Material Composition
The grass used for fermentation had a TS content of 389 g kg−1 and a VS content of 355 g kg−1. Both are in the typical range for grass used for ensiling [29]. Likewise, the contents of crude ash (CA) (88 g kg−1), crude fibre (CF) (255 g kg−1), crude protein (CP) (122 g kg−1) and crude fat (CL) (27 g kg−1) (Table 2). In the case of the sum of structural substances, specified as neutral detergent fibre (NDF), a content of 526 g kg−1 was measured and the amount of lignin, defined as acid detergent lignin (ADL), was 21 g kg−1. The fraction of acid detergent fibres (ADF) was measured at 279 g kg−1.
Chemical composition of the silage
The grass silage was stored according to the treatment at 22 ±1 °C or 37 ±1 °C for 3, 14, 30, and 90 days. Whereas Maillard reactions can occur in silages at temperatures between 35 and 40 °C [31, 32]. This can lead to energy losses during storage [33].
Table 3 shows the chemical composition, pH and VS-loss of the substrate after ninety days of fermentation. For all treatments stored at 37 °C, excluding treatment WB37, 1.2-propanediol formed during storage. Its concentration lies between 9.0 and 33.1 g kg−1. In the literature a degradation of lactic acid to acetic acid and 1.2-propanediol is reported, with an optimal temperature between 20 and 37 °C, which could be the reason for the accumulation of 1.2-propanediol during fermentation at 37 °C [36].
The development of pH and the concentrations of lactic acid, acetic acid, butyric acid and ethanol for all treatments are shown in Fig. 1. Considering the pH development of the control treatment (C20), it followed the typical course for ensiling, with a fast decrease of pH in the first three days to pH 4 [34]. This pH decrease is mainly caused by lactic acid production during fermentation. For the treatment C20, a final lactic acid concentration of 114.9 ±9.6 g kg−1 was achieved. While, a content of 56 g kg−1 was reached in another study (Table 3) [29]. But the concentration of lactic acid can differ widely according to raw material composition, buffering capacity and the dominant lactic acid bacteria species [35]. Overall, the highest concentration of lactic acid was reached by treatment B20 with 158.0 ±22.2 g kg−1. With regard to the untreated control treatment stored at 37 °C (C37), the lactic acid concentration (67.7 ±6.3 g kg−1) was 41% lower and the acetic acid concentration (49.2 ±3.4 g kg−1) was 148% higher when stored at 20 °C. Likewise, for all other treatments stored at 37 °C, a higher acetic acid concentration was observed compared to the same treatment stored at 20 °C. For silage storage temperatures at 40 °C, it is reported that due to a higher pH-values and a lower lactic to acetic acid ratio, a curtailed fermentation, increased proteolysis and secondary heterolactic fermentation takes place which are resulting in a reduced silage quality for animal feeding [31]. In terms of the ethanol concentration, the values differ widely between 4.2 and 20.9 g kg−1DM. The treatment WB20 showed the highest ethanol content and the treatment C37 showed the lowest amount of ethanol in the silage.
The highest VS-loss during fermentation and storage was measured for the treatments B20 (20.3%) and B37 (20.5%). Both are higher than the control treatments (12.5%; 13.1%). One reason for the high VS-losses could be the high generation and formation of organic acids, especially lactic acid and according to that the formation of CO2 through fermentation.
In the case of butyric acid concentration, only the treatments with a combination of low TS content and an increased buffering capacity formed the butyric acid during fermentation. Considering the fermentation durations, the formation of butyric acid began between days 3 and 14, but most of it was formed between days 30 and 90. The generation of butyric acid went hand in hand with a significantly higher VS-loss (18.7%, 18.9%) compared to the untreated control (12.5%) and an increase in pH between days 30 and 90. This might be caused by the higher pKa-value of butyric acid and degradation of lactic acid. The highest butyric acid concentration was achieved by treatment WB37 with 40.5 g kg−1 at a theoretical initial TS content of 0.222 g kg−1. McEniry et al. [37] achieved a concentration of 20.6 g kg−1 by the addition of 10 g kg−1 carbonated lime and an initial DM content of 265 g kg−1. In another study, no butyric acid was detected with an addition of 8.6 and 17.2 g kg−1 and an initial TS content of 374 g kg−1 [29].
Separation, Methane Yield Potential and Chemical Composition of the Press Juice
Separation
The silage was separated after ninety days of fermentation. Figure 2 shows the mass share and losses of the resulting press cake, press juice, and losses due to the separation process. As expected, all treatments with a reduced TS content show a higher share of press juice from 37 to 43% compared to the treatments with higher TS with 15%–24%. In case of the resulting press cake it is the other way around with 73%–82% (C20, B20, C37, B37) and 55%–61% (W20, WB20, W27, WB37). All together the losses lie between 1.5 and 3.8%. In general, a higher press juice (PJ) yield is preferred to increase the amount of extracted organic acids.
Methane Yield Potential
After the separation procedure, the methane yield potential of the original silage and the produced press cake (PC) was determined. The resulting methane yield potential for the PC of the control treatment (C20) was 0.353 ± 0.002 m2 kg−1, which is comparable to other studies for grass silage e.g. 0.344-0.383 m2 kg−1[37] (Fig. 3). For the untreated control (C37), a specific methane yield of 0.371 ± 0.018 m2 kg−1 was achieved, but there was no significant difference to the other control treatment stored at room temperature. Altogether the lowest yield was achieved for the treatment B20 with 0.317 ±0.024 m2 kg−1, which is significantly lower than the treatments C37, W20, B37, and WB37. But, an effect of silage fermentation temperature on the specific methane yield could not be proved. It is reported that the ensiling fermentation characteristics have only a minor effect on the SMY. But high butyric acid and ethanol concentrations can lead to higher SMY’s. Nevertheless, due to high VS and energy losses through butyric acid fermentation, the higher SMY’s cannot compensate these losses [37].
When comparing the specific methane yield to the original silage before separation, significant differences were measured between press cake and silage of the treatments W20, W37 and B37 (Fig. 3). All other treatments did not show a significant difference in SMY before and after separation. But through the separation process, there was a considerable loss of mass and especially organic acids, which are dissolved in the produced press juice.
Specific methane yields of the investigated treatments with comparison between silage and press cake within the respective temperatures (A), within the fraction (B), and comparison between the different fractions of the same treatment (C). Different letters indicate significant differences between the treatments at the same temperature (A), between the treatments at the same fraction (B) and between the fraction at the same treatment (C) at p < 0.05
Chemical Composition of the Press Juice
Considering the number of organics in the press juice, expressed in the form of the COD value, it ranged from 115.4 to 186.5 g kg−1. There were two groups, which were significantly different from each other. All treatments with reduced TS content showed lower values (115.4–123.7 g kg−1) than the treatments without the addition of water (164.1–186.4 g kg−1). For the concentration of lactic acid, the highest value was 68.9 g kg−1 for the treatment C20 and the lowest value was measured for treatment WB37 with 2.4 g kg−1. Similarly, for day 90, as it was expected, only two treatments WB20 (10.7 g kg−1) and WB37 (20.1 g kg−1) showed considerable amounts of butyric acid. Compared to other studies and addition of microorganisms for butyric acid production, and the use of glucose as substrate, butyric acid concentrations from 6.3 to 62.8 g L−1 were achieved [38]. For agricultural raw materials like corn stalk and the addition of C. thermobutyricum, 15.82 g L−1 butyric acid concentration was achieved [13]. Furthermore, from hydrolysed wheat straw, 19-20 g L−1 are reported [14]. The resulting concentration of butyric acid in the separated fermentation broth in this study is comparable to other agricultural raw materials and processes. On one hand, the whole fermentation process is simpler, because the addition of special microorganisms and a pre-treatment of the substrate is not required. On the other hand, the resulting fermentation contains many impurities like other organic acids and suspended solids. Additionally, the recovery of the butyric acid from the produced press juice is challenging, like for other organic acids [39]. But to make the whole fermentation more controllable and repeatable, a pre-treatment of the raw material to get a homogeneous substrate and the selective addition of Clostridia should be considered for future experiments. When calculating the butyric acid yield (YBA), based on the used amount of VS for ensiling and the butyric acid concentration in the press juice, a yield of 9.2 (g kg−1) and 33.1 (g kg−1) was achieved. However, only 3.3% of the used VS was converted to butyric acid, so there is need for further improvement of the process to achieve a higher butyric acid yield (Table 4).
Upscaling, Production Scheme and Technical Obstacles
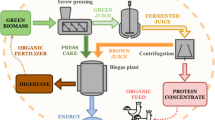
Following the production scheme of a Green Biorefinery assuming a grass yield per ha and year of 10 MgVS, and using the corresponding TS-losses during fermentation and separation, two treatments with the highest yield in butyric acid per VS (WB20, WB37) were selected (Fig. 4). In both cases, the total amount of organic acids in the silage was lower in comparison to the corresponding control treatment. But the amount of organic acids separated from the produced silage was higher in the treated silages, which is a result of the reduced TS content of the silage. Altogether an amount of 652 kg ha−1 a−1 of organic acids was produced for treatment WB37, which was 64% of the organic acids in the original silage. Considering the butyric acid yield, this treatment produced the highest amount per ha with 332 kg ha−1 a−1. The treatment WB20 also had high butyric acid concentrations in the press juice, but the amount produced per ha (93 kg ha−1 a−1) was considerably lower. The theoretical amount of butyric acid, which can be produced in this production scheme could contribute to the value-added chain of anaerobic digestion of agricultural substrates. However, to make the butyric acid available, further processing of the produced press juice is necessary to gain a product which can be used by the industry. One possible method could be a first filtration step by ceramic membranes to remove suspended particles, followed by a liquid-liquid extraction and distillation. Nonetheless, the main disadvantages are the cost, solvent’s toxicity and high energy consumption of the distillation [13]. A precipitation crystallization, also called as salting-out method is reported to be promising for the purification of fermentation broths and to separate butyric acid. But further research is necessary for the optimization and upscaling [13, 40].
In the case of methane production, for the resulting press cake of the treatment 20 WB, an amount of 2567 m3CH4 ha−1 a−1 could be produced. This is a loss of 20% compared to the silage methane yield per ha of the control treatment C20 (3,193 m3 ha−1 a−1). Regarding the press cake of the treatment WB37 (2778 m3 ha−1 a−1), a loss of 19% was observed compared to the unseparated silage of the untreated control (C37) (3429 m3 ha−1 a−1). The overall production scheme with its methane and organic acid yields is highly dependent on the quality of the used raw material and thereby on the environmental as well as the weather conditions in the harvesting year and the cultivation conditions.
Conclusions
The ensiling process is basically a lactic acid fermentation, but when the ensiling conditions are changed, butyric acid can also be produced. Consequently, this process is also accompanied with higher TS losses during storage. When considering the final butyric acid concentration in the fermentation broth, this method can compete with other butyric acid production processes that use agricultural raw materials. It is simpler because pre-treating of the substrate is not necessary and it could be integrated into a farm with an anaerobic digestion plant, where synergy effects could be achieved. The main disadvantage of the whole process is the purification and extraction of the produced butyric acid, to make it available for the chemical industry and further utilization. However, further research is necessary to improve this process in the case of optimal TS and VS content and buffer addition for butyric acid production including the raw material composition and quality. Furthermore, the addition of selected butyric acid producing bacteria, microbial analysis and a pre-treatment step for hydrolysis of the biomass should be considered.
Availability of Data and Material
The collected data can be handed out upon request.
Code Availability (Software Application or Custom Ccode)
The program code for data analysis in R is not supplied. On the basis of the data, an evaluation can be carried out with the help of the functions mentioned in the method section.
References
Faostat: Land use. http://www.fao.org/faostat/en/#data/RL (2017). Accessed 26 July 2020
Agler, M.T., Wrenn, B.A., Zinder, S.H., Angenent, L.T.: Waste to bioproduct conversion with undefined mixed cultures: the carboxylate platform. Trends Biotechnol. (2011). https://doi.org/10.1016/j.tibtech.2010.11.006
Lemmer, A., Krl, J.: Demand-driven biogas production in anaerobic filters. Appl. Energy (2017). https://doi.org/10.1016/j.apenergy.2016.10.073
Hahn, H., Krautkremer, B., Hartmann, K., Wachendorf, M.: Review of concepts for a demand-driven biogas supply for flexible power generation. Renew. Sustain. Energy Rev. (2014). https://doi.org/10.1016/j.rser.2013.08.085
Mauky, E., Jacobi, H.F., Liebetrau, J., Nelles, M.: Flexible biogas production for demand-driven energy supply-feeding strategies and types of substrates. Bioresour. Technol. (2015). https://doi.org/10.1016/j.biortech.2014.08.123
Kamm, B., Schke, P., Hille, C.: Green biorefinery - industrial implementation. Food Chem. (2016). https://doi.org/10.1016/j.foodchem.2015.11.088
Kamm, B., Kamm, M.: Principles of biorefineries. Appl. Microbiol. Biotechnol. (2004). https://doi.org/10.1007/s00253-003-1537-7
Thang, V.H., Koschuh, W., Kulbe, K.D., Kromus, S., Krotscheck, C., Novalin, S.: Desalination of high salt content mixture by two-stage electrodialysis as the first step of separating valuable substances from grass silage. Desalination (2004). https://doi.org/10.1016/S0011-9164(04)00068-2
Santamaría-Fernández, M., Molinuevo-Salces, B., Kiel, P., Steenfeldt, S., Uellendahl, H., Lk, M.: Lactic acid fermentation for refining proteins from green crops and obtaining a high quality feed product for monogastric animals. J. Clean. Prod. (2017). https://doi.org/10.1016/j.jclepro.2017.06.115
Franco, M., Hurme, T., Winquist, E., Rinne, M.: Grass silage for biorefinery—a meta-analysis of silage factors affecting liquid–solid separation. Grass Forage Sci. (2019). https://doi.org/10.1111/gfs.12421
Steinbrenner, J., Nägele, H.-J., Buschmann, A., Hmann, B., Oechsner, H.: Testing different ensiling parameters to increase butyric acid concentration for maize silage, followed by silage separation and methane yield potential of separated solids residues. Bioresour. Technol. Rep. (2019). https://doi.org/10.1016/j.biteb.2019.100193
Dwidar, M., Park, J.-Y., Mitchell, R.J., Sang, B.-I.: The future of butyric acid in industry. Sci. World J. (2012). https://doi.org/10.1100/2012/471417
Jha, A.K., Li, J., Yuan, Y., Baral, N., Ai, B., Ai, B.: A review on bio-butyric acid production and its optimization. Int. J. Agric. Biol. 16, 1019 (2014)
Baroi, G.N., Baumann, I., Westermann, P., Gavala, H.N.: Butyric acid fermentation from pretreated and hydrolysed wheat straw by an adapted Clostridium tyrobutyricum strain. Microb. Biotechnol. (2015). https://doi.org/10.1111/1751-7915.12304
Fu, H., Le Yu, Lin, M., Wang, J., Xiu, Z., Yang, S.-T.: Metabolic engineering of Clostridium tyrobutyricum for enhanced butyric acid production from glucose and xylose. Metab. Eng. (2017). https://doi.org/10.1016/j.ymben.2016.12.014
Luo, H., Yang, R., Zhao, Y., Wang, Z., Liu, Z., Huang, M., Zeng, Q.: Recent advances and strategies in process and strain engineering for the production of butyric acid by microbial fermentation. Bioresour. Technol. (2018). https://doi.org/10.1016/j.biortech.2018.01.007
Agrarmeteorologie Baden-Wemberg: Wetterstation Unterer Lindenhof. https://www.wetter-bw.de/Internet/AM/NotesBwAM.nsf/bwweb/7b6dc2ff140f6662c1257db2002d5120?OpenDocument&TableRow=3.1.3,3.6#3.1. (2021)
German Agricultural Society: Guideline for Testing of Silage Additives on DLG Quality Mark Capability (2013)
Auerbach, H., Eise, M., Hng, K., Ler, C., Nußbaum, H., Ostertag, J., Pahlow, G., Pauly, T., Rahn, S., Richter, W., R, G., Rubenschuh, U., Spiekers, H., Staudacher, W., Sum, K.-H., Thaysen, J., Tr, A., Wyss, U.: DLF Testing Guidelines. for the award and use of the DLG Quality Mark for ensiling agents. DLG TestService GmbH, 64823 Groß-Umstadt, Germany. https://www.dlg.org/fileadmin/downloads/tests/guetezeichen/DLG_Pruefrichtlinien_Siliermittel_ENG_kurz_2018.pdf (2018). Accessed 3 December 2018
Association of German Engineers: Fermentation of organic materials: Characterisation of the substrate, sampling, collection of material data, fermentation tests. Beuth Verlag GmbH, Berlin (2006)
Helffrich, D., Oechsner, H.: The Hohenheim biogas yield test. Comparison of different laboratory techniques for the digestion of biomass. Agrartechnische Forschung 9, 27–30 (2003)
Weißbach, F., Strubelt, C.: Correcting the Dry Matter Content of GRass Silages as a Substrate for Biogas Production, pp. 210–211. Landtechnik (2008)
The Commission of the European Communities: COMMISSION REGULATION (EC) No 152/2009 of 27 January 2009 laying down the methods of sampling and analysis for the official control of feed (2009)
Federation of German Agricultural Investigation and Research Institutes (VDLUFA): The Chemical Analysis for Feedstuffs. Method Book III. VDLUFA, Darmstadt (2012)
R Core Team: R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna (2018)
RStudio Team: RStudio: Integrated Development Environment for R. RStudio Inc., Boston (2016)
de Mendiburu, F.: agricolae: Statistical Procedures for Agricultural Research (2017)
Wickham, H. (ed.): ggplot2: elegant graphics for data analysis. Springer, New York (2016)
Haag, N.L., Grumaz, C., Wiese, F., Kirstahler, P., Merkle, W., Nägele, H.-J., Sohn, K., Jungbluth, T., Oechsner, H.: Advanced green biorefining. Effects of ensiling treatments on lactic acid production, microbial activity and supplementary methane formation of grass and rye. Biomass Conv. Bioref. (2016). https://doi.org/10.1007/s13399-015-0178-2
LUFA Nord-West. https://www.lufa-nord-west.de/index.cfm/action/downloadcenter?file=0DEFC3DA (2020). Accessed 30 Oct 2020
Kim, S.C., Adesogan, A.T.: Influence of ensiling temperature, simulated rainfall, and delayed sealing on fermentation characteristics and aerobic stability of corn silage. J. Dairy Sci. (2006). https://doi.org/10.3168/jds.S0022-0302(06)72586-3
Muck, R.E., Moser, L.: PITT, R.E.: Postharvest factors affecting ensiling. Agronomy 42, 251–304 (2003)
Coblentz, W., Hoffman, P., Undersander, D.: Heat damaged forages: effects on forage energy content. Focus Forage 13, 4 (2011)
Bolsen, K.K., Ashbell, G., Weinberg, Z.G.: Silage fermentation and silage additives - review Asian-Australas. J. Anim. Sci. (1996). https://doi.org/10.5713/ajas.1996.483
Driehuis, F., Oude Elferink, S.J.W.H., van Wikselaar, P.G.: Fermentation characteristics and aerobic stability of grass silage inoculated with Lactobacillus buchneri, with or without homofermentative lactic acid bacteria. Grass Forage Sci. (2001). https://doi.org/10.1046/j.1365-2494.2001.00282.x
Oude Elferink, S.J., Krooneman, J., Gottschal, J.C., Spoelstra, S.F., Faber, F., Driehuis, F.: Anaerobic conversion of lactic acid to acetic acid and 1,2-propanediol by Lactobacillus buchneri. Appl. Environ. Microbiol. (2001). https://doi.org/10.1128/AEM.67.1.125-132.2001
McEniry, J., Allen, E., Murphy, J.D., O’Kiely, P.: Grass for biogas production. The impact of silage fermentation characteristics on methane yield in two contrasting biomethane potential test systems. Renew. Energy (2014). https://doi.org/10.1016/j.renene.2013.09.052
Zhang, C., Yang, H., Yang, F., Ma, Y.: Current progress on butyric acid production by fermentation. Curr. Microbiol. (2009). https://doi.org/10.1007/s00284-009-9491-y
Li, Q.-Z., Jiang, X.-L., Feng, X.-J., Wang, J.-M., Sun, C., Zhang, H.-B., Xian, M., Liu, H.-Z.: Recovery processes of organic acids from fermentation broths in the biomass-based industry. J. Microbiol. Biotechnol. (2016). https://doi.org/10.4014/jmb.1505.05049
Wu, D., Chen, H., Jiang, L., Xu, C.A.I.J., CEN, Z.: Efficient separation of butyric acid by an aqueous two-phase system with calcium chloride. Chin. J. Chem. Eng. (2010). https://doi.org/10.1016/S1004-9541(10)60255-8
Acknowledgements
The work was funded by the Federal Ministry of Food and Agriculture (BMEL) within the scope of the project “Optigär- Development of efficient two-phase biogas plants via a coupled energetic and material use after separation of hydrolysis products” (FKZ 224-005-15). The authors acknowledge the support from our project partners Rainer Bolduan (European Institute for Energy Research), Sarah Bger, Sebastian Reinhardt, Wolfgang Becker (all Fraunhofer Institute for Chemical Technology) and Ralph Leistner (Lipp GmbH). Many thanks go to our colleagues Benedikt Hmann for his dedicated work on the Hohenheim Biogas Yield test, Padma Priya Ravi for the proofreading and English language correction, and Annette Buschmann and Jacqueline Kindermann for analysing the samples and their precise work in the analytical laboratory. Furthermore, the authors would like to thank the student assistants Celina Spangenberger and Nathalie Brucker for their help carrying out the experiments.
Funding
Open Access funding enabled and organized by Projekt DEAL. The work was funded by the Federal Ministry of Food and Agriculture (BMEL) of Germany within the scope of the project “Optigär- Development of efficient two-phase biogas plants via a coupled energetic and material use after separation of hydrolysis products” (FKZ 224-005-15).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The manuscript is the original work of the authors and was not previously submitted to Waste and Biomass Valorization. All authors declare no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Steinbrenner, J., Mueller, J. & Oechsner, H. Combined Butyric Acid and Methane Production from Grass Silage in a Novel Green Biorefinery Concept. Waste Biomass Valor 13, 1873–1884 (2022). https://doi.org/10.1007/s12649-021-01626-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-021-01626-4